Abstract
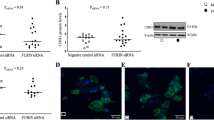
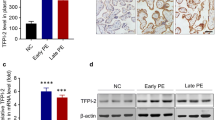
The production of soluble fms-like tyrosine kinase 1 (sFLT1) by exogenous chymotrypsin in trophoblast cells through protease-activated receptor (PAR) 2 was investigated to identify the role of a chymotrypsin-like serine protease in preeclampsia (PE) pathogenesis. We evaluated the expression of chymotrypsin, FLT1, and sFLT1 in monolayers of immortalized human trophoblast cells derived from placenta (TCL-1 cells). To investigate whether chymotrypsin enhances the production and release of sFLT1 through PAR-2, we examined changes in sFLT1 protein levels in conditioned medium by enzyme-linked immunosorbent assay and sFLT1 messenger RNA (mRNA) levels by real-time polymerase chain reaction in TCL-1 cells treated with exogenous chymotrypsin in the presence or absence of a PAR-2 antagonist or a chymotrypsin inhibitor (TPCK). We also examined changes in PAR-2 expression in TCL-1 cells treated with tumor necrosis factor (TNF) α in the presence or absence of a polyclonal anti-TNF-α antibody. Western blot analysis showed that TCL-1 trophoblast cells expressed chymotrypsin, FLT1, and sFLT1. Compared with the control cells, the sFLT1 level in the conditioned medium and sFLT1 mRNA level in cells were both significantly enhanced when treated with a PAR-2 agonist or chymotrypsin for 6 hours. In contrast, the sFLT1 level in the medium and sFLT1 mRNA level in cells treated with a PAR-2 agonist or chymotrypsin were suppressed in the presence of a PAR-2 antagonist or a chymotrypsin inhibitor. The PAR-2 expression was upregulated by TNF-α, which was suppressed in the presence of TNF-α antibodies. These results indicate that chymotrypsin-like serine protease enhances sFLT1 production through PAR-2 in trophoblast cells and thus plays an important additional role in PE pathogenesis.
Similar content being viewed by others
References
Abalos E, Cuesta C, Carroli G, et al. WHO multicountry survey on maternal and newborn health research network pre-eclampsia, eclampsia and adverse maternal and perinatal outcomes: a secondary analysis of the World Health Organization Multicountry Survey on Maternal and Newborn Health. Br J Obstet Gynecol. 2014;121(suppl 1):14–24.
Brosens I. A study of the spiral arteries of the decidua basalis in normotensive and hypertensive pregnancies. J Obstet Gynecol Br Common. 1964;71:222–230.
Gilbert JS, Babcock SA, Granger JP. Hypertension produced by reduced uterine perfusion in pregnant rats is associated with increased soluble fms-like tyrosine kinase-1 expression. Hypertension. 2007;50(6):1142–1147.
Nevo O, Soleymanlou N, Wu Y, et al. Increased expression of sFlt-1 in in vivo and in vitro models of human placental hypoxia is mediated by HIF-1. Am J Physiol Regul Integr Comp Physiol. 2006;291(4):R1085–R1093.
Cindrova-Davies T, Sanders DA, Burton GJ, Charnock-Jones DS. Soluble FLT1 sensitizes endothelial cells to inflammatory cytokines by antagonizing VEGF receptor-mediated signalling. Cardiovasc Res. 2011;89(3):671–679.
Maynard S, Epstein FH, Karumanchi SA. Preeclampsia and angiogenic imbalance. Annu Rev Med. 2008;59:61–78.
Maynard SE, Min JY, Merchan J, et al. Excess placental soluble FMS-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J Clin Invest. 2003;111(5):649–658.
Gu Y, Lewis DF, Wang Y. Placental productions and expressions of soluble endoglin, soluble fms-like tyrosine kinase receptor-1, and placental growth factor in normal and preeclamptic pregnancies. J Clin Endocrinol Metab. 2008;93(1):260–266.
Myatt L, Webster RP. Vascular biology of preeclampsia. J Thromb Haemost. 2009;7(3):375–384.
Lim KH, Zhou Y, Janatpour M, et al. Human cytotrophoblast differentiation/invasion is abnormal in pre-eclampsia. Am J Pathol. 1997;151(6):1809–1818.
Salafia CM, Pezzullo JC, Ghidini A, Lopez-Zeno JA, Whittington SS. Clinical correlations of patterns of placental pathology in preterm pre-eclampsia. Placenta. 1998;19(1):67–72.
Sugimura M, Kobayashi T, Shu F, Kanayama N, Terao T. Annexin V inhibits phosphatidylserine-induced intrauterine growth restriction in mice. Placenta. 1999;20(7):555–660.
Sugimura M, Ohashi R, Kobayashi T, Kanayama N. Intraplacental coagulation in intrauterine growth restriction: cause or result? Semin Thromb Hemost. 2001;27(7):107–113.
Lockwood CJ, Toti P, Arcuri F, et al. Thrombin regulates soluble fms-like tyrosine kinase-1 (sFlt-1) expression in first trimester decidua. Am J Pathol. 2007;170(4):1398–1405.
Zhao Y, Koga K, Osuga Y, et al. Thrombin enhances soluble Fms-like tyrosine kinase 1 expression in trophoblasts; possible involvement in the pathogenesis of preeclampsia. Fertil Steril. 2012;98(4):917–921.
Coughlin SR. Thrombin signalling and protease-activated receptors. Nature. 2000;407(6801):258–264.
Nakanishi-Matsui M, Zheng YW, Sulciner DJ, et al. PAR3 is a cofactor for PAR4 activation by thrombin. Nature. 2000;404(6778):609–613.
Nystedt S, Ramakrishnan V, Sundelin J. The proteinase-activated receptor 2 is induced by inflammatory mediators in human endothelial cells—comparison with the thrombin receptor. J Biol Chem. 1996;271(25):14910–1415.
O’Brien PJ, Prevost N, Molino M, et al. Thrombin responses in human endothelial cells. Contributions from receptors other than PAR1 include the transactivation of PAR2 by thrombin-cleaved PAR1. J Biol Chem. 2000;275(18):13502–13509.
Kong W, McConalogue K, Khitin LM, et al. Luminal trypsin may regulate enterocytes through proteinase-activated receptor 2. Proc Natl Acad Sci U S A. 1997;94(16):8884–8889.
Camerer E, Huang W, Coughlin SR. Tissue factor- and factor X-dependent activation of protease-activated receptor 2 by factor VIIa. Proc Natl Acad Sci U S A. 2000;97(10):5255–5260.
Rothmeier AS, Ruf W. Protease-activated receptor 2 signaling in inflammation. Semin Immunopathol. 2012;34(1):133–149.
Hutchinson ES, Brownbill P, Jones NW, et al. Utero-placental haemodynamics in the pathogenesis of pre-eclampsia. Placenta. 2009;30(7):634–641.
Lewis MP, Clements M, Takeda S, et al. Partial characterization of an immortalized human trophoblast cell-line, TCL-1, which possesses a CSF-1 autocrine loop. Placenta. 1996;17(2-3):137–46.
Kovats S, Main EK, Librach C, et al. A class I antigen, HLA-G, expressed in human trophoblasts. Science. 1990;248(4952):220–223.
Kabir-Salmani M, Shiokawa S, Akimoto Y, et al. Alphavbeta3 integrin signaling pathway is involved in insulin-like growth factor I-stimulated human extravillous trophoblast cell migration. Endocrinology. 2003;144(4):1620–1630.
Suga N, Sugimura M, Koshiishi T, et al. Heparin/heparan sulfate/CD44-v3 enhances cell migration in term placenta-derived immortalized human trophoblast cells. Biol Reprod. 2012;86(5):134, 1–8.
Wang Y, Gu Y, Zhang Y, Lewis DF, Alexander JS, Granger DN. Increased chymotrypsin-like protease (chymase) expression and activity in placentas from women with preeclampsia. Placenta. 2007;28(4):263–269.
Munaut C, Lorquet S, Pequeux C, et al. Hypoxia is responsible for soluble vascular endothelial growth factor receptor-1 (VEGFR-1) but not for soluble endoglin induction in villous trophoblast. Hum Reprod. 2008;23(6):1407–1415.
Zhao S, Gu Y, Groome LJ, Cooper D, Wang Y. Proteases and sFlt-1 release in the human placenta. Placenta. 2010;31(6):512–518.
Sokolova E, Aleshin S, Reiser G. Expression of protease-activated receptor (PAR)-2, but not other PARs, is regulated by inflammatory cytokines in rat astrocytes. Neurochem Int. 2012;60(3):276–275.
Al-Ani B, Hewett PW, Cudmore MJ, et al. Activation of proteinase-activated receptor 2 stimulates soluble vascular endothelial growth factor receptor 1 release via epidermal growth factor receptor transactivation in endothelial cells. Hypertension. 2010;55(3):689–697.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hirai, C., Sugimura, M., Makino, S. et al. Chymotrypsin Enhances Soluble Fms-Like Tyrosine Kinase 1 Production Through Protease-Activated Receptor 2 in Placenta-Derived Immortalized Human Trophoblast Cells. Reprod. Sci. 23, 1542–1550 (2016). https://doi.org/10.1177/1933719116646203
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719116646203