Abstract
Objective
Fenretinide is a synthetic retinoid analogue that promotes apoptosis but has decreased toxicity when compared to other retinoids. We have previously shown that retinoic acid (RA) production in endometriotic tissue is decreased, resulting in reduced estrogen metabolism and apoptotic resistance. We hypothesize fenretinide may induce apoptosis in endometriotic cells and tissues, thereby reducing disease burden.
Materials and Methods
Primary endometriotic stromal cells were collected, isolated, cultured, and treated with fenretinide in doses from 0 to 20 p,mol/L. Cell count, viability, and immunoblots were per-formed to examine apoptosis. Quantitative reverse transcription-polymerase chain reaction from endometriotic cells treated with fenretinide was used to examine expression of genes involved in RA signaling including stimulated by RA 6 (STRA6), cellular RA binding protein 2 (CRABP2), and fatty acid binding protein 5 (FABP5). Endometriotic tissue was xenografted subcutaneously into the flanks of mice which were treated with fenretinide for 2 weeks, after which the mice were killed and lesion volumes calculated. Statistical analysis was performed using ttestand analysis ofvariance.
Results
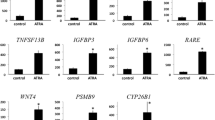
Treatment with fenretinide significantly decreased total cell count (doses 5-20 p,L) and viability (doses 10-20 p,mol/L). Fenretinide increased protein levels of the apoptotic marker poly (ADP ribose) polymerase (starting at 10 p,mol/L) and decreased proliferation marker proliferating cell nuclear antigen (10 μmol/L, starting at 8-day treatment). Examination of genes involved in retinoid uptake and action showed that treatment induced STRA6 expression while expression of CRABP2 and FABP5 remained unchanged. Fenretinide also significantly decreased the endometriotic lesion xenograft volume.
Conclusions
Fenretinide increases STRA6 expression thereby potentially reversing the pathological loss of retinoid availability. Treatment with this Compound induces apoptosis. In vivo treatments decrease lesion volume. Targeting the RA signaling pathway may be a promising novel treatment for women with endometriosis.
Similar content being viewed by others
References
Bulun SE. Endometriosis. N Engl J Med. 2009;360(3):268–279.
Giudice LC. Clinical practice. Endometriosis. N Engl J Med. 2010;362(25):2389–2398.
Lucidi RS, Witz CA, Chrisco M, Binkley PA, Shain SA, Schenken RS. A novel in vitro model of the early endometriotic lesion demonstrates that attachment of endometrial cells to mesothelial cells is dependent on the source of endometrial cells. Fertil Steril. 2005;84(1):16–21.
Giudice LC, Kao LC. Endometriosis. Lancet. 2004;364(9447):1789–1799.
Giudice LC, Teiles TL, Lobo S, Kao L. The molecular basis for implantation failure in endometriosis:on the road to discov-ery. Ann N Y Acad Sci. 2002;955:252–264; discussion 293-255, 396-406.
Bulun SE, Cheng YH, Pavone ME, et al.. Estrogen receptorbeta, estrogen receptoralpha, and progesterone resistance in endometriosis. Semin Reprod Med. 2010;28(1):36–43.
Pavone ME, Dyson M, Reirstad S, et al.. Endometriosis expresses a molecular pattern consistent with decreased retinoid uptake, metabolism and action. Hum Reprod. 2011;26(8):2157–2164.
Pavone ME, Reierstad S, Sun H, Milad M, Bulun SE, Cheng YH. Altered retinoid uptake and action contributes to cell survival in endometriosis. J Clin Endocrinol Metab. 2010;95(11):300–309.
Umesono K, Giguere V, Glass CK, Rosenfeld MG, Evans RM. Retinoic acid and thyroid hormone induce gene expression through a common responsive element. Nature. 1988; 336(6196):262–265.
Jones S, Meng L, Parsons DW, et al.. Somatic mutations in the chromatin remodeling gene ARID1A occur in several tumor types. Hum Mutat. 2012;33(1):100–103.
Nasu K, Yuge A, Tsuno A, Nishida M, Narahara H. Involvement of resistance to apoptosis in the pathogenesis of endometriosis. Histol Histopathol. 2009;24(9):1181–1192.
Formelli F, Barua AB, Olson JA. Bioactivities of N-(4-hydroxy-phenyl) retinamide and retinoyl beta-glucuronide. Faseb J. 1996; 10(9):1014–1024.
Broaddus RR, Xie S, Hsu CJ, Wang J, Zhang S, Zou C. The chemopreventive agents 4-HPR and DFMO inhibit growth and induce apoptosis in uterine leiomyomas. Am J Obstet Gynecol. 2004;190(3):686–692.
Ciolino HP, Dai Z, Nair V. Retinol inhibits aromatase activity and expression in vitro. J Nutr Biochem. 2011;22(6):522–526.
Ciolino HP, Wang TT, Sathyamoorthy N. Inhibition of aromatase activity and expression in MCF-7 cells by the chemopreventive retinoid N-(4-hydroxy-phenyl)-retinamide. Br J Cancer. 2000; 83(3):333–337.
Pavone ME, Bulun SE. Aromatase inhibitors for the treatment of endometriosis. Fertil Steril. 2012;98(6):1370–1379.
Ryan IP, Schriock ED, Taylor RN. Isolation, characterization, and comparison of human endometrial and endometriosis cells in vitro. J Clin Endocrinol Metab. 1994;78(3):642–649.
Noble LS, Takayama K, Zeitoun KM, et al.. Prostaglandin E2 stimulates aromatase expression in endometriosis-derived stromal cells. J Clin Endocrinol Metab. 1997;82(2):600–606.
Dyson MT, Roqueiro D, Monsivais D, et al.. Genome-wide DNA methylation analysis predicts an epigenetic switch for GATA factor expression in endometriosis. PLoS Genet. 2014;10(3):e1004158.
Biorad. Bio-Rad TC20 Automated Cell Counter Description. Cell Counter Description. Web site. http://www.bio-rad.com/en-us/product/tc20-automated-cell-counter?tab=Description.
Dyson MT, Jones JK, Kowalewski MP, et al.. Mitochondrial Akinase anchoring protein 121 binds type II protein kinase A and enhances steroidogenic acute regulatory protein-mediated steroi-dogenesis in MA-10 mouse leydig tumor cells. Biol Reprod. 2008; 78(2):267–277.
Bruner KL, Eisenberg E, Gorstein F, Osteen KG. Progesterone and transforming growth factor-beta coordinately regulate sup-pression of endometrial matrix metalloproteinases in a model of experimental endometriosis. Steroids. 1999;64(9):648–653.
Bruner KL, Matrisian LM, Rodgers WH, Gorstein F, Osteen KG. Suppression of matrix metalloproteinases inhibits establishment of ectopic lesions by human endometrium in nude mice. J Clin Invest. 1997;99(12):2851–2857.
Eaton JL, Unno K, Caraveo M, Lu Z, Kim JJ. Increased AKT or MEK1/2 activity influences progesterone receptor levels and localization in endometriosis. J Clin Endocrinol Metab. 2013; 98(12):e1871–e1879.
Formelli F, Cleris L. Therapeutic effects of the combination of fenretinide and all-trans-retinoic acid and of the two retinoids with cisplatin in a human ovarian Carcinoma xenograft and in a cisplatin-resistant sub-line. Eur J Cancer. 2000;36(18):2411–2419.
Carrera S, Cuadrado-Castano S, Samuel J, et al.. Stra6, a retinoic acid-responsive gene, participates in p53-induced apoptosis after DNA damage. Cell Death Differ. 2013;20(7):910–919.
Decensi A, Robertson C, Guerrieri-Gonzaga A, et al.. Randomized double-blind 2x2 trial of low-dose tamoxifen and fenretinide for breast Cancer prevention in high-risk premenopausal women. J Clin Oncol. 2009;27(23):3749–3756.
Villablanca JG, London WB, Naranjo A, et al.. Phase II study of oral capsular 4-hydroxyphenylretinamide (4-HPR/fenretinide) in pediatric patients with refractory or recurrent neuroblastoma:a report from the Children’s Oncology Group. Clin Cancer Res. 2011;17(21):6858–6866.
Maurer BJ, Kang MH, Villablanca JG, et al.. Phase I trial of fenretinide delivered orally in a novel organized lipid complex in patients with relapsed/refractory neuroblastoma:a report from the New Approaches to Neuroblastoma Therapy (NANT) consor-tium. PediatrBlood Cancer. 2013;60(11):1801–1808.
Ratko TA, Detrisac CJ, Dinger NM, Thomas CF, Kelloff GJ, Moon RC. Chemopreventive efficacy of combined retinoid and tamoxifen treatment following surgical excision of a primary mammary Cancer in female rats. Cancer Res. 1989;49(16):4472–4476.
Mittal N, Malpani S, Dyson M, et al.. Fenretinide:a novel treatment for endometrial Cancer. PLoS One. 2014;9(10):e110410.
Gopal AK, Pagel JM, Hedin N, Press OW. Fenretinide enhances rituximab-induced cytotoxicity against B-cell lymphoma xeno-grafts through a caspase-dependent mechanism. Blood. 2004; 103(9):3516–3520.
Mukherjee N, Reuland SN, Lu Y, et al.. Combining a BCL2 inhi-bitor with the retinoid derivative fenretinide targets melanoma cells Including melanoma initiating cells. J Invest Dermatol. 2015;135(3):842–850.
Cheng YH, Yin P, Xue Q, Yilmaz B, Dawson MI, Bulun SE. Retinoic acid (RA) regulates 17beta-hydroxySteroid dehydrogenase type 2 expression in endometrium:interaction of RA receptors with specificity protein (SP) 1/SP3 for estradiol metabolism. J Clin Endocrinol Metab. 2008;93(5):1915–1923.
Berry DC, O’Byrne SM, Vreeland AC, Blaner WS, Noy N. Cross talk between signaling and vitamin A Transport by the retinol-binding protein receptor STRA6. Mol Cell Biol. 2012;32(15):3164–3175.
Sokalska A, Anderson M, Villanueva J, et al.. Effects of simvastatin on retinoic acid System in primary human endometrial stro-mal cells and in a chimeric model of human endometriosis. J Clin Endocrinol Metab. 2013;98(3):e463–e471.
Veronesi U, De Palo G, Marubini E, et al.. Randomized trial of fenretinide to prevent second breast malignancy in women with early breast Cancer. J Natl Cancer Inst. 1999;91(21):1847–1856.
Veronesi U, Mariani L, Decensi A, et al.. Fifteen-year results of a randomized phase III trial of fenretinide to prevent second breast Cancer. Ann Oncol. 2006;17(7):1065–1071.
Torrisi R, Parodi S, Fontana V, et al.. Effect of fenretinide on plasma IGF-I and IGFBP-3 in early breast Cancer patients. Int J Cancer. 1998;76(6):787–790.
Koay DC, Zerillo C, Narayan M, Harris LN, DiGiovanna MP. Anti-tumor effects of retinoids combined with trastuzumab or tamoxifen in breast Cancer cells:induction of apoptosis by retinoid/trastuzumab combinations. Breast Cancer Res. 2010;12(4):r62.
Durante S, Orienti I, Teti G, et al.. Antitumor activity of fenretinide complexed with human serum albumin in lung Cancer xenograft mouse model. Oncotarget. 2014;5(13):4811–4820.
Mata NL, Lichter JB, Vogel R, Han Y, Bui TV, Singerman LJ. Investigation of oral fenretinide for treatment of geographic atro-phy in age-related macular degeneration. Retina. 2013;33(3):498–507.
Holpuch AS, Phelps MP, Desai K-GH, et al.. Evaluation of a mucoadhesive fenretinide patch for local intraoral delivery:a strategy to reintroduce fenretinide for oral Cancer chemoprevention. Carcinogenesis. 2012;33(5):1098–1105.
Costa A, Malone W, Perloff M, et al.. Tolerability of the synthetic retinoid fenretinide® (HPR). Eur J Canc Clin Oncol. 1989;25(5):805–808.
Camerini T, Mariani L, De Palo G, et al.. Safety of the synthetic retinoid fenretinide:long-term results from a controlled clinical trial for the prevention of contralateral breast Cancer. J Clin Oncol. 2001; 19(6):1664–1670.
Soprano DR, Soprano KJ. Retinoids as teratogens. Annu Rev Nutr. 1995;15(1):111–132.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pavone, M.E., Malpani, S.S., Dyson, M. et al. Fenretinide:A Potential Treatment for Endometriosis. Reprod. Sci. 23, 1139–1147 (2016). https://doi.org/10.1177/1933719116632920
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719116632920