Abstract
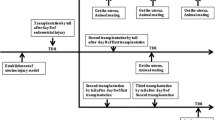
Bone marrow mesenchymal stem cells (BMSCs) show multidirectional differentiation and possess immunoregulatory properties. Although transplantation of BMSCs has a therapeutic effect on many diseases, it is unclear whether BMSC transplantation can be used as a therapy for a thin endometrium. To explore whether transplantation of BMSCs directly into the uterine cavity can improve endometrium thickness, a thin endometrium rat model was established by infusing ethanol into the uterine cavity. In all, 48 rats with thin endometrium and 24 normal rats were divided into 3 groups: (1) normal group, (2) experimental group transplanted with BMSCs into uterine cavity, and (3) control group transplanted with saline into the uterine cavity. The morphology of the endometrium, the regeneration and receptivity of the endometrium, and the mechanisms involved in BMSC therapy were subsequently analyzed by hematoxylin and eosin staining, Western blot analysis, and reverse transcription-polymerase chain reaction throughout an observation period of 3 estrus cycles. The rats in the experimental group had a significantly thicker endometrial lining and exhibited higher expression of cytokeratin, vimentin, integrin αγβ3, and leukaemia inhibitor factor (LIF) than that of the control group (P < .05). Bromodeoxyuridine -positive cells were detected in the endometrium after BMSC transplantation. Some proinflammatory cytokines, such as tumor necrosis factor α messenger RNA (mRNA) and interleukin (IL)-1βmRNA, were significantly downregulated, and anti-inflammatory cytokines, such as basic fibroblast growth factor (bFGF) mRNA and IL-6mRNA, were significantly upregulated in the experimental group compared to the control group (P < .05). In conclusion, BMSCs improved endometrium thickness, probably via their migration and immunomodulatory properties. Uterine perfusion with BMSCs represents a promising new tool for the currently intractable problem of an inadequate, thin endometrium.
Similar content being viewed by others
References
Friedler S, Schenker JG, Herman A, Lewin A. The role of ultrasonography in the evaluation of endometrial receptivity following assisted reproductive treatments: a critical review. Hum Reprod Update. 1996;2(4):323–335.
Al-Ghamdi A, Coskun S, Al-Hassan S, Al-Rejjal R, Awartani K. The correlation between endometrial thickness and outcome of in vitro fertilization and embryo transfer (IVF-ET) outcome. Reprod Biol Endocinol. 2008;6:37.
Senturk LM, Erel CT. Thin endometrium in assisted reproductive technology. Curr Opin Obstet Gynecol. 2008;20(3):221–228.
Grove JE, Bruscia E, Krause DS. Plasticity of bone marrow-derived stem cells. Stem Cells. 2004;22(4):487–500.
Du H, Taylor HS. Contribution of bone marrow-derived stem cells to endometrium and endometriosis. Stem Cells. 2007;25(8):2082–2086.
Yagi H, Soto-Gutierrez A, Parekkadan B, et al. Mesenchymal stem cells: mechanisms of immunomodulation and homing. Cells Transplant. 2010;19(6):667–679.
Kinnaird T, Stabile E, Burnett MS, et al. Local delivery of marrow derived stromal cells augments collateral perfusion through paracrine mechanisms. Circulation. 2004;109(12):1543–1549.
Giordano A, Galderisi U, Marino IR. From the laboratory bench to the patient’s bedside: An update on clinical trials with mesenchymal stem cells. J Cell Physiol. 2007;211(1):27–35.
Du H, Naqvi H, Taylor HS. Ischemia/reproduction injury promotes and Granulocyte-Colony Stimulating Factor inhibits migration of bone marrow-derived stem cells to endometrium. Stem Cells Dev. 2012;21(18):3324–3331.
Taylor HS. Endometrial cells derived from donor stem cells in bone marrow transplant recipients. JAMA 2004;292(1):81–85.
Ikoma T, Kyo S, Maida Y, et al. Bone marrow-derived cells from male donors can compose endometrial glands in female transplant recipients. Am J Obstet Gynecol. 2009;201(6):608.e1–e8.
Bratincsak A, Brownstein MJ, Cassiani-Ingoni R, et al. CD45-positive blood cells give rise to uterine epithelial cells in mice. Stem Cells. 2007;25(11):2820–2826.
Mints M, Jansson M, Sadeghi B, et al. Endometrial endothelial cells are derived from donor stem cells in a bone marrow transplant recipient. Hum Reprod. 2008;23(1):139–143.
Munoz-Elias G, Woodbury D, Black IB. Marrow stromal cells, mitosis, and neuronal differentiation: stem cell and precursor functions. Stem Cells. 2003;21(4):437–448.
Zhao J, Gao H, Li YP. Development of an animal model for thin endometrium using 95% ethanol. J Fert In Vitro. 2012;2(4):109.
Morigi M, Introna M, Imberti B, et al. Human bone marrow mesenchymal stem cells accelerate recovery of acute renal injury and prolong survival in mice. Stem Cells. 2008;26(8):2075–2082.
Ortiz LA, DuTreil M, Fattman C, et al. Interleukin 1 receptor antagonist mediates the anti-inflammatory and anti-fibrotic effect of mesenchymal stem cells during lung injury. Proc Natl Acad Sci U S A. 2007;104:11002–11007.
Kunter U, Rong S, Djuric Z, et al. Transplanted mesenchymal stem cells accelerate glomerular healing in experimental glomerulonephritis. J Am Soc Nephrol. 2006;17(8):2202–2212.
Lee RH, Seo MJ, Reger RL, et al. Multipotent stromal cells from human marrow home to and promote repair of pancreatic islets and renal glomeruli in diabetic pancreatic islets and renal glomeruli in diabetic NOD/scid mice. Proc Natl Acad Sci U S A. 2006;103(46):17438–17443.
Ringden O, Uzunel M, Rasmusson L, et al. Mesenchymal stem cells for the treatment of therapy-resistant graft-versus-host disease. Transplantation. 2006;81(10):1390–1397.
Minguell JJ, Erices A. Mesenchymal stem cells and the treatment of cardiac disease. Exp Biol Med (Maywood). 2006;231(1):39–49.
Achache H, Revel A. Endometrial receptivity markers, the journey to successful embryo implantation. Hum Reprod Update. 2006;12(6):731–746.
Nagori CB, Panchal SY, Patel H. endometrial regeneration using autologous adult stem cells followed by conception by in vitro fertilization in a patient of severe Asherman’s syndrome. J Hum Reprod Sci. 2011;4(1):43–48.
Hou M, Yang KM, Zhang H, et al. Transplantation of mesenchymal stem cells from human bone marrow improves damaged heart function in rats. Int J Cardiol. 2007;115(2):220–228.
Deng J, Petersen BE, Steindler DA, Jorgensen M, Laywell ED. Mesenchymal stem cells spontaneously express neural proteins in culture and are neurogenic after transplantation. Stem Cells. 2006;24(4):1054–1064.
Wislet-Gendebien S, Hans G, Leprince P, Rigo JM, Moonen G, Rogister B. Plasticity of cultured mesenchymal stem cells: switch from nestin-positive to excitable neuron-like phenotype. Stem Cells. 2005;23(3):392–402.
Mao F, Xu WR, Qian H, et al. Immunosuppressive effects of mesenchymal stem cells in collagen induced mouse arthritis. Inflamm Res. 2010;59(3):219–225.
Bai L, Lennon DP, Eaton V, et al. Human bone marrow-derived mesenchymal stem cells induce Th2-polarized immune response and promote endogenous repair in animal models of multiple sclerosis. Glia. 2009;57(11):1192–1203.
Imanishi Y, Saito A, Komoda H, et al. Allogenic mesenchymal stem cell transplantation has a therapeutic effect in acute myocardial infarction in rats. J Mol Cell Cardiol. 2008;44(4):662–671.
Yagi H, Soto-Gutierrez A, Parekkadan B, et al. Mesenchymal stem cells: mechanisms of immunomodulation and homing. Cell Transplant. 2010;19(6):667–679.
JY Oh, KM Kim, MS Shin, et al. The anti-inflammatory and anti-angiogenic role of mesenchymal stem cells in corneal wound healing following chemical injury. Stem Cells. 2008;26(4):1047–1055.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, J., Zhang, Q., Wang, Y. et al. Uterine Infusion With Bone Marrow Mesenchymal Stem Cells Improves Endometrium Thickness in a Rat Model of Thin Endometrium. Reprod. Sci. 22, 181–188 (2015). https://doi.org/10.1177/1933719114537715
Published:
Issue Date:
DOI: https://doi.org/10.1177/1933719114537715