Abstract
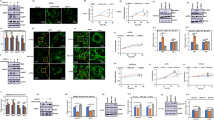
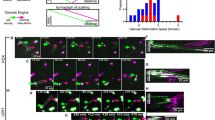
The dissemination of cancer cells is one of the main reasons for treatment failure. During the process of establishing distant metastases, cancer cells must migrate through narrow environments, such as intercellular junctions, extracellular matrix, and basement membranes. The deformability of the cell nucleus is a limiting factor for migration through narrow environments; therefore, as the largest and hardest organelle, the nucleus is a key factor in crossing restrictive spaces. Nesprin-1/2 provides mechanical linkage between the nucleus and the cytoskeleton, but the specific mechanism by Nesprin-1/2 regulating tumor cell nuclear deformation is unclear. Our study found that knocking down Nesprin-1/2 significantly weakens cell migration ability, and knocking down Nesprin-1/2 makes the nucleus becoming more easily deformed. Meanwhile, the knockdown of Nesprin-1/2 leads to a decrease in Lamin A/C levels. To explore whether Lamin A/C protein undergoes degradation, we treated cells with caspase-6 inhibitor Z-VEID-FMK, autophagy inhibitor 3-methyladenine (3-MA), or broad-spectrum proteasome inhibitor MG132, and found that knockdown of Nesprin-1/2 led to the degradation of Lamin A/C via the proteasome pathway. Through immunofluorescence experiments, we observed F-actin distribution in the process of pore migration, and found that knockdown of Nesprin-1/2 weakened the pushing force of the nuclear tail, making it unable to propel the nucleus forward. In conclusion, this study demonstrates that Nesprin-1/2 regulates nuclear deformation and reorganizes the cytoskeleton, which together affects cell pore migration and provides a theoretical reference for the study of nuclear deformation mechanisms.






Similar content being viewed by others
Data availability
Data will be made available on reasonable request.
References
A.J. Ridley, M.A. Schwartz, K. Burridge, R.A. Firtel, M.H. Ginsberg, G. Borisy, J.T. Parsons, A.R. Horwitz, Cell migration: integrating signals from front to back. Science 302, 1704–1709 (2003)
J.P. Campanale, D.J. Montell, Who’s really in charge: diverse follower cell behaviors in collective cell migration. Curr. Opin. Cell Biol. 81, 102160 (2023)
J. Chen, D. Yan, Y. Chen, Understanding the driving force for cell migration plasticity. Biophys J. 122, 1–7 (2023)
P. Friedl, Prespecification and plasticity: shifting mechanisms of cell migration. Curr. Opin. Cell Biol. 16, 14–23 (2004)
M. Krause, F.W. Yang, M. Te Lindert, P. Isermann, J. Schepens, R.J.A. Maas, C. Venkataraman, J. Lammerding, A. Madzvamuse, W. Hendriks, J. Te Riet, K. Wolf, Cell migration through three-dimensional confining pores: speed accelerations by deformation and recoil of the nucleus. Philos. Trans. R Soc. Lond. B Biol Sci. 374, 20180225 (2019)
A. Fruleux, R.J. Hawkins, Physical role for the nucleus in cell migration. J. Phys. Condens. Matter 28, 363002 (2016)
D.E. Jaalouk, J. Lammerding, Mechanotransduction gone awry. Nat. Rev. Mol. Cell Biol. 10, 63–73 (2009)
D. Feng, J.A. Nagy, K. Pyne, H.F. Dvorak, A.M. Dvorak, Neutrophils emigrate from venules by a transendothelial cell pathway in response to FMLP. J. Exp. Med. 187, 903–915 (1998)
M.M. Nava, Y.A. Miroshnikova, L.C. Biggs, D.B. Whitefield, F. Metge, J. Boucas, H. Vihinen, E. Jokitalo, X. Li, J.M. Garcia Arcos, B. Hoffmann, R. Merkel, C.M. Niessen, K.N. Dahl, S.A. Wickstrom, Heterochromatin-driven nuclear softening protects the genome against mechanical stress-induced damage. Cell 181, 800-817e822 (2020)
A. Tajik, Y. Zhang, F. Wei, J. Sun, Q. Jia, W. Zhou, R. Singh, N. Khanna, A.S. Belmont, N. Wang, Transcription upregulation via force-induced direct stretching of chromatin. Nat. Mater. 15, 1287–1296 (2016)
J. Swift, I.L. Ivanovska, A. Buxboim, T. Harada, P.C. Dingal, J. Pinter, J.D. Pajerowski, K.R. Spinler, J.W. Shin, M. Tewari, F. Rehfeldt, D.W. Speicher, D.E. Discher, Nuclear lamin-A scales with tissue stiffness and enhances matrix-directed differentiation. Science 341, 1240104 (2013)
V. Venturini, F. Pezzano, F. Catala Castro, H.M. Hakkinen, S. Jimenez-Delgado, M. Colomer-Rosell, M. Marro, Q. Tolosa-Ramon, S. Paz-Lopez, M.A. Valverde, J. Weghuber, P. Loza-Alvarez, M. Krieg, S. Wieser, V. Ruprecht, The nucleus measures shape changes for cellular proprioception to control dynamic cell behavior. Science 370, eaba2644 (2020)
A.J. Lomakin, C.J. Cattin, D. Cuvelier, Z. Alraies, M. Molina, G.P.F. Nader, N. Srivastava, P.J. Saez, J.M. Garcia-Arcos, I.Y. Zhitnyak, A. Bhargava, M.K. Driscoll, E.S. Welf, R. Fiolka, R.J. Petrie, N.S. De Silva, J.M. Gonzalez-Granado, N. Manel, A.M. Lennon-Dumenil, D.J. Muller, M. Piel, The nucleus acts as a ruler tailoring cell responses to spatial constraints. Science 370, eaba2894 (2020)
B.D. Hoffman, C. Grashoff, M.A. Schwartz, Dynamic molecular processes mediate cellular mechanotransduction. Nature 475, 316–323 (2011)
A. Katiyar, J. Zhang, J.D. Antani, Y. Yu, K.L. Scott, P.P. Lele, C.A. Reinhart-King, N.J. Sniadecki, K.J. Roux, R.B. Dickinson, T.P. Lele, The nucleus bypasses obstacles by deforming like a drop with surface tension mediated by Lamin A/C. Adv. Sci. (Weinh) 9, e2201248 (2022)
N.L. Ovsiannikova, S.V. Lavrushkina, A.V. Ivanova, L.M. Mazina, O.A. Zhironkina, Kireev, II, Lamin A as a determinant of mechanical properties of the cell nucleus in health and disease. Biochemistry (Mosc) 86, 1288–1300 (2021)
G. Lee, S.B. Han, D.H. Kim, Cell-ECM contact-guided intracellular polarization is mediated via lamin A/C dependent nucleus-cytoskeletal connection. Biomaterials 268, 120548 (2021)
J. Lammerding, P.C. Schulze, T. Takahashi, S. Kozlov, T. Sullivan, R.D. Kamm, C.L. Stewart, R.T. Lee, Lamin A/C deficiency causes defective nuclear mechanics and mechanotransduction. J Clin Invest 113, 370–378 (2004)
F. Roncato, O. Regev, S.W. Feigelson, S.K. Yadav, L. Kaczmarczyk, N. Levi, D. Drago-Garcia, S. Ovadia, M. Kizner, Y. Addadi, J.C. Sabino, Y. Ovadya, S.F. de Almeida, E. Feldmesser, G. Gerlitz, R. Alon, Reduced Lamin A/C does not facilitate cancer cell transendothelial migration but compromises lung metastasis. Cancers (Basel) 13, 2383 (2021)
G.E. Morris, K.N. Randles, Nesprin isoforms: are they inside or outside the nucleus? Biochem. Soc. Trans. 38, 278–280 (2010)
D. Rajgor, C.M. Shanahan, Nesprins: from the nuclear envelope and beyond. Expert Rev. Mol. Med. 15, e5 (2013)
E. Antmen, U. Demirci, V. Hasirci, Micropatterned surfaces expose the coupling between actin cytoskeleton-Lamin/Nesprin and nuclear deformability of breast cancer cells with different malignancies. Adv. Biol. (Weinh) 5, e2000048 (2021)
A. Matsumoto, M. Hieda, Y. Yokoyama, Y. Nishioka, K. Yoshidome, M. Tsujimoto, N. Matsuura, Global loss of a nuclear lamina component, lamin A/C, and LINC complex components SUN1, SUN2, and nesprin-2 in breast cancer. Cancer Med. 4, 1547–1557 (2015)
I. Dupin, S. Etienne-Manneville, Nuclear positioning: mechanisms and functions. Int. J. Biochem. Cell Biol. 43, 1698–1707 (2011)
M. Almonacid, M.E. Terret, M.H. Verlhac, Nuclear positioning as an integrator of cell fate. Curr. Opin. Cell Biol. 56, 122–129 (2019)
C. Bruno, Nuclear positioning: a matter of life. Semin. Cell Dev. Biol. 82, 1–2 (2018)
D.G. Thomas, A. Yenepalli, C.M. Denais, A. Rape, J.R. Beach, Y.L. Wang, W.P. Schiemann, H. Baskaran, J. Lammerding, T.T. Egelhoff, Non-muscle myosin IIB is critical for nuclear translocation during 3D invasion. J. Cell Biol. 210, 583–594 (2015)
B. Cadot, V. Gache, E.R. Gomes, Moving and positioning the nucleus in skeletal muscle - one step at a time. Nucleus 6, 373–381 (2015)
B. Burke, Chain reaction: LINC complexes and nuclear positioning. F1000Res 8, 136 (2019)
A. Jayo, M. Malboubi, S. Antoku, W. Chang, E. Ortiz-Zapater, C. Groen, K. Pfisterer, T. Tootle, G. Charras, G.G. Gundersen, M. Parsons, Fascin regulates nuclear movement and deformation in migrating cells. Dev. Cell 38, 371–383 (2016)
K. Pfisterer, A. Jayo, M. Parsons, Control of nuclear organization by F-actin binding proteins. Nucleus 8, 126–133 (2017)
P.M. Davidson, A. Battistella, T. Dejardin, T. Betz, J. Plastino, N. Borghi, B. Cadot, C. Sykes, Nesprin-2 accumulates at the front of the nucleus during confined cell migration. EMBO Rep. 21, e49910 (2020)
R. Zhu, C. Liu, G.G. Gundersen, Nuclear positioning in migrating fibroblasts. Semin. Cell Dev. Biol. 82, 41–50 (2018)
T.J. Chancellor, J. Lee, C.K. Thodeti, T. Lele, Actomyosin tension exerted on the nucleus through nesprin-1 connections influences endothelial cell adhesion, migration, and cyclic strain-induced reorientation. Biophys. J. 99, 115–123 (2010)
T. Lammermann, B.L. Bader, S.J. Monkley, T. Worbs, R. Wedlich-Soldner, K. Hirsch, M. Keller, R. Forster, D.R. Critchley, R. Fassler, M. Sixt, Rapid leukocyte migration by integrin-independent flowing and squeezing. Nature 453, 51–55 (2008)
J.K. Kim, A. Louhghalam, G. Lee, B.W. Schafer, D. Wirtz, D.H. Kim, Nuclear lamin A/C harnesses the perinuclear apical actin cables to protect nuclear morphology. Nat. Commun. 8, 2123 (2017)
S.J. Heo, K.H. Song, S. Thakur, L.M. Miller, X. Cao, A.P. Peredo, B.N. Seiber, F. Qu, T.P. Driscoll, V.B. Shenoy, M. Lakadamyali, J.A. Burdick, R.L. Mauck, Nuclear softening expedites interstitial cell migration in fibrous networks and dense connective tissues. Sci. Adv. 6, eaax5083 (2020)
T. Fischer, A. Hayn, C.T. Mierke, Effect of nuclear stiffness on cell mechanics and migration of human breast cancer cells. Front. Cell Dev. Biol. 8, 393 (2020)
T. Harada, J. Swift, J. Irianto, J.W. Shin, K.R. Spinler, A. Athirasala, R. Diegmiller, P.C. Dingal, I.L. Ivanovska, D.E. Discher, Nuclear lamin stiffness is a barrier to 3D migration, but softness can limit survival. J. Cell Biol. 204, 669–682 (2014)
A. Das, A. Barai, M. Monteiro, S. Kumar, S. Sen, Nuclear softening is essential for protease-independent migration. Matrix Biol. 82, 4–19 (2019)
Acknowledgements
This work is supported, in part or in whole, by the National Natural Science Foundation of China (32071304, U19A2006, 12132004, 11972111, 32171309, 12272086), the China Postdoctoral Science Foundation (2019T120821), the Sichuan Science and Technology Program (2023NSFSC1233, 2022NSFSC0048, 2022NSFSC0686, 2023YFSY0038), and the Joint Funds of Center for Engineering Medicine (ZYGX2021YGLH017).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare no potential conflicts of interest.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qin, X., Chen, K., Wang, M. et al. Nesprin-1/2 facilitates breast cancer cell pore migration via nucleus deformation. Eur. Phys. J. Spec. Top. 232, 2739–2749 (2023). https://doi.org/10.1140/epjs/s11734-023-00930-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1140/epjs/s11734-023-00930-5