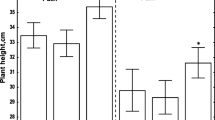
Abstract—A halo is a concentric zone (5–50 µm) of the altered surface of the leaf epidermis of wheat around the penetration site of the causative agent of powdery mildew Blumeria graminis. Halo variability was studied in a control and after treatment with metabolic inhibitors (3-amino-1,2,4-triazole, bisindole-maleimide hydrochloride, diphenyleniodionium chloride, 3-isobutyl-1-methylxanthine, catalase, lanthanum nitrate). After amido black was staining, the epidermis fragments were observed as monochromatic circular blue, or bicolor red and blue structures, or as a system of several purple-red, blue, or slightly colored rings. All treatments contributed to a significant reduction in the rate successful penetration of the pathogen into plant cells with the formation of haustoria, the marker of susceptibility at the tissue level in this pathosystem. Halo sizes were increased by treating with 3-amino-1,2,4-triazole, a peroxidase and catalase inhibitor, and also by treating catalase, an enzyme that degrades hydrogen peroxide. Treatment with lanthanum nitrate, which blocks calcium channels, led to a significant decrease in the halo diameter. Halo associated with haustoria, were 1.5 times smaller compared with those observed in the absence of successful development of the pathogen. The correlation between the sizes of such halos and the occurrence frequency of haustoria is shown for the first time. The average sizes of paired large and small halos for each variant, as well as the average distances between them, reliably correlated with each other and weakly correlated with the number of haustoria. The data indicate the possibility of using some halo parameters in diagnosing the local viability of individual infectious units of B. graminis, as well as in tests for the wheat plant resistance to powdery mildew.




Similar content being viewed by others
REFERENCES
Avetisyan, G.A. and Babosha, A.V., Role of oxidative stress in pathogenesis of wheat powdery mildew, Byull. Gl. Bot. Sada, 2011, vol. 196, pp. 157–164.
Babosha, A.V., Ryabchenko, A.S., and Avetisyan, T.V., Effect of exogenous cytokinins on dynamics of development and differentiation of infectious structures of the pathogen of wheat powdery mildew, Cell Tissue Biol., 2009, vol. 3, no. 4, pp. 387–396. https://doi.org/10.1134/S1990519X09040117
Beavo, J.A., Rogers, N.L., Crofford, O.B., Hardman, J.G., Sutherland, E.W., and Newman, E.V., Effects of xanthine derivatives on lipolysis and on adenosine 3',5'-monophosphate phosphodiesterase activity, Mol. Pharmacol., 1970, vol. 6, no. 6, pp. 597–603.
Cross, A.R. and Jones, O.T.G., The effect of the inhibitor diphenylene iodonium on the superoxide-generating system of neutrophils. Specific labelling of a component polypeptide of the oxidase, Biochem. J., 1986, vol. 237, no. 1, pp. 111–116. https://doi.org/10.1042/bj2370111
Demidchik, V., ROS-activated ion channels in plants: Biophysical characteristics, physiological functions and molecular nature, Int. J. Mol. Sci., 2018, vol. 19, no. 4, p. 1263. https://doi.org/10.3390/ijms19041263
Edwards, H.H., A basic staining material associated with the penetration process in resistant and susceptible powdery mildewed barley, New Phytol., 1970, vol. 69, pp. 299–301. https://doi.org/10.1111/j.1469-8137.1970.tb02429.x
Hückelhoven, R., Fodor, J., Preis, C., and Kogel, K.H., Hypersensitive cell death and papilla formation in barley attacked by the powdery mildew fungus are associated with hydrogen peroxide but not with salicylic acid accumulation, Plant Physiol., 1999, vol. 119, pp. 1251–1260. https://doi.org/10.1104/pp.119.4.1251
Kunoh, H., Ishizaki, H., and Nakaya, K., Cytological studies of early stages of powdery mildew in barley and wheat leaves: (II) significance of the primary germ tube of Erysiphe graminis on barley leaves, Physiol. Plant Pathol., 1977, p. 10, pp. 191–199. https://doi.org/10.1016/0048-4059(77)90022-4
Margoliash, E. and Novogrodsky, A., A study of the inhibition of catalase by 3-amino-1:2:4-triazole, Biochem. J., 1958, vol. 68, no. 3, pp. 468–475. https://doi.org/10.1042/bj0680468
Mellersh, D.G. and Heath, M.C., Plasma membrane— Cell wall adhesion is required for expression of plant defense responses during fungal penetration, Plant Cell, 2001, vol. 13, pp. 413–424. https://doi.org/10.1105/tpc.13.2.413
Mishina, G.N., Serezhkina, G.V., Avetisyan, T.V., Ryabchenko, A.S., and Andreev, L.N., Specific features of the formation of a halo during pathogenesis as a response of cereal epidermal cells to penetration of powdery mildew causative agents, Biol. Bull., 2001, vol. 28, pp. 354–360. https://doi.org/10.1023/A:1016666906210
Ouchi, S., Oku, H., and Hibino, C., Localization of induced resistance and susceptibility in barley leaves inoculated with the powdery mildew fungus, Phytopathology, 1976, vol. 66, pp. 901–905. https://doi.org/10.1094/Phyto-66-901
Pantoja, O., Gelli, A., and Blumwald, E., Voltage-dependent calcium channels in plant vacuoles, Science, 1992, vol. 255, no. 5051, pp. 1567–1570. https://doi.org/10.1126/science.255.5051.1567
Pottosin, I. and Zepeda-Jazo, I., Powering the plasma membrane Ca2+-ROS self-amplifying loop, J. Exp. Bot., 2018, vol. 69, no. 14, pp. 3317–3320. https://doi.org/10.1093/jxb/ery179
Sanin, S.S., Motovilin, A.A., Korneva, L.G., et al., Chemical protection of wheat from the diseases under the conditions of intensive grain production, Zashchita i Karantin Rastenii, 2011, vol. 8, pp. 3–10.
Serezhkina, G.V., Mishina, G.N., Andreev, L.N., Avetisyan, T.V., and Lapochkina, I.F., Cytophysiological characteristics of resistance of Aegilops speltoides Tausch, and disomic addition wheat—Aegilops lines (2N = 44) at the stage of seedlings against powdery mildew, Biol. Bull., 1999, vol. 26, pp. 344–350.
Shetty, N.P., Jørgensen, H.J.L., Jensen, J.D., Collinge, D.B., and Shetty, H.S., Roles of reactive oxygen species in interactions between plants and pathogens, Eur. J. Plant Pathol., 2008, vol. 121, pp. 267–280. https://doi.org/10.1007/s10658-008-9302-5
Sugai, K., Masaoka, H., Penjore, K., Hanboonsong, Y., Nishiguchi, M., and Yamaoka, N., The time and spatial strategy of Blumeria graminis f. sp. hordei for surviving after failure of first infection, Physiol. Mol. Plant Pathol., 2010, vol. 74, no. 5, pp. 346–350. https://doi.org/10.1016/j.pmpp.2010.05.005
Tarchevskii, I.G., Signal’nye sistemy kletok rastenii (Signal Systems of Plant Cells), Moscow: Nauka, 2002.
Thordal-Christensen, H., Zhang, Z., Wei, Y., and Collinge, D.B., Subcellular localization of H2O2 in plants. H2O2 accumulation in papillae and hypersensitive response during the barley–powdery mildew interaction, Plant J., 1997, vol. 11, pp. 1187–1194. https://doi.org/10.1046/j.1365-313X.1997.11061187.x
Toullec, D., Pianetti, P., Coste, H., Bellevergue, P., Grand-Perret, T., Ajakane, M., Baudet, V., Boissin, P., Boursier, E., Loriolle, F., Duhamel, L., Charon, D., and Kirilovsky, J., The bisindolylmaleimide GF 109203X is a potent and selective inhibitor of protein kinase C, J. Biol. Chem., 1991, vol. 266, no. 24, pp. 15771–15781.
Trujillo, M., Kogel, K.-H., and Hückelhoven, R., Superoxide and hydrogen peroxide play different roles in the nonhost interaction of barley and wheat with inappropriate formae speciales of Blumeria graminis, Mol. Plant-Microbe Interact., 2004, vol. 17, pp. 304–312. https://doi.org/10.1094/MPMI.2004.17.3.304
Xing, Y., Jia, W., and Zhang, J., AtMKK1 mediates ABA-induced CAT1 expression and H2O2 production via AtMPK6-coupled signaling in Arabidopsis, Plant J., 2008, vol. 54, pp. 440–451. https://doi.org/10.1111/j.1365-313X.2008.03433.x
Yang, T. and Poovaiah, B.W., Hydrogen peroxide homeostasis: Activation of plant catalase by calcium/calmodulin, Proc. Natl. Acad. Sci. U. S. A., 2002, vol. 99, pp. 4097–4102. https://doi.org/10.1073/pnas.052564899
Funding
The study was carried out within the state task of the Tsitsin Main Botanical Garden RAS (topic no. 118021490111-5).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This work does not contain any studies involving human and animal subjects.
CONFLICT OF INTEREST
The authors of this work declare that they have no conflicts of interest.
Additional information
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Babosha, A.V., Avetisyan, T.V. Effect of Metabolic Inhibitors on the Size and Structure of Halos Induced by the Powdery Mildew Pathogen Blumeria graminis f. sp. tritici on Wheat Leaves. Biol Bull Rev 13 (Suppl 1), S123–S132 (2023). https://doi.org/10.1134/S2079086423070022
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2079086423070022