Abstract
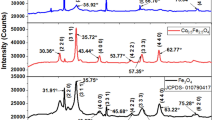
A nanocomposite material with an EDTA counterpart, ethylenediamine triacetate (ED3A), covalently fixed on the surface according to the core–shell technique is obtained in order to develop adsorbents for reversible analytical concentration of heavy-metal ions from solutions. The material is characterized using the methods of IR spectroscopy, X-ray phase analysis, elemental analysis, scanning electron microscopy, and energy-dispersive X-ray spectroscopy. It is shown that magnetite nanocrystallites with a size of 8–10 nm in Fe3O4@SiO2@ED3A are coated by a layer of silicon oxide that enhances 200- to 10-fold the stability of nanoparticles in the aqueous solutions in the range of pH 1–8. It is shown that the separation time of nanocomposites using a NbFeB magnet does not exceed 10 s. According to the data of elemental analysis, the concentration of functional groups on the nanocomposite surface is 0.32 mmol/g. Sorption isotherms of a number of metals on Fe3O4@SiO2@ED3A are studied. They indicate the chemical character of the metal adsorption processes due to their complexation with functional groups. The conditions of sorption concentration of the Fe(IIІ), Cu(II), Pb(II), Zn(II), and Cd(II) ions from aqueous solutions and their desorption are studied. The affinity of the obtained nanocomposite to heavy-metal ions decreases in the series of Fe(IIІ) > Cu(II) > Pb(II) > Cd(II) ~ Zn(II). It is shown that all the studied metals (except for Zn(II)) are quantitatively recovered on Fe3O4@SiO2@ED3A from weakly acidic aqueous solutions and are completely desorbed into the solution of 1.0 M HCl or HNO3.
Similar content being viewed by others
References
Fritz, J.S., Analytical Solid-Phase Extraction, New York: Wiley-VCH, 1999.
Kadirvelu, C., Faur-Brasquet, C., and Le Cloirec, P., Langmuir, 2000, vol. 16, p. 8404.
Hui, K.S., Chao, C.Y.H., and Kot, S.C., J. Hazard. Mater., 2005, vol. 127, p. 89.
Holubová, Z., Moos, M., and Sommer, L., Chem. Pap., 2012, vol. 66, no. 10, p. 899.
Lisichkin, G.V., Fadeev, A.Yu., Serdan, A.A., and Nesterenko, P.N., Khimiya privitykh poverkhnostnykh soedinenii (Chemistry of Grafted Surface Compounds), Lisichkin, G.V., Ed., Moscow: FIZMATLIT, 2003.
Cerdan, A.A. and Lisichkin, G.V., in Kontsentrirovanie sledov organicheskikh soedinenii (Concentrating of Organic Compounds’ Traces), Moscow: Nauka, 1990, vol. 10, p. 28.
Giakisikli, G. and Anthemidis, A.N., Anal. Chim. Acta, 2013, vol. 789, p. 1.
Zhang, L., Gao, X., Xiong, Zh., et al., RSC Adv., 2015, vol. 5, no. 72, p. 58873.
Samadi, A. and Amjadi, M., Microchim. Acta, 2015, vol. 182, nos. 1–2, p. 257.
Meng, J., Bu, J., and Deng, C., J. Chromatogr. A, 2011, vol. 1218, no. 12, p. 1585.
Mashhadizadeh, M.H. and Karami, Z., J. Hazard. Mater., 2011, vol. 190, nos. 1–3, p. 1023.
Egodawatte, S., Datt, A., Burns, E.A., and Larsen, S.C., Langmuir, 2015, vol. 31, no. 27, p. 7553.
Bagheri, H., Afkhami, A., and Saber-Tehrani, M., Talanta, 2012, vol. 97, p. 87.
Li, G., Liu, M., Zhang, Z., et al., J. Colloid Interface Sci., 2014, vol. 424, p. 124.
Zhang, F., Lan, J., Zhao, Z., et al., J. Colloid Interface Sci., 2012, vol. 387, no. 1, p. 205.
Ohshima, K., Watanabe, H., and Haraguchi, K., Anal. Sci., 1986, vol. 2, p. 131.
Stöber, W., Fink, A., and Bohn, E., J. Colloid Interface Sci., 1968, vol. 26, p. 62.
Oliveira-Silva, R., Pinto da Costa, J., Vitorino, R., and Daniel-da-Silva, A.L., J. Mater. Chem. B, 2015, vol. 3, p. 238.
Scherrer, P., Nachr. Ges. Wiss. Goettingen, Math.-Phys. Kl., 1918, vol. 26, p. 98.
Zhou, Y.T., Nie, H.L., White, C.B., et al., J. Colloid Interface Sci., 2009, vol. 330, p. 29.
Wang, H., Kang, J., Liu, H., and Qu, J., J. Environ. Sci., 2009, vol. 21, no. 11, p. 1473.
Addy, M., Losey, B., Mohseni, R., et al., Appl. Clay Sci., 2012, vols. 59–60, p. 115.
Amsheeva, A.A., Zh. Anal. Khim., 1978, vol. 78, p. 1055.
Dyatlova, N.M., Temkina, V.Ya., and Popov, K.I., Kompleksony i kompleksonaty metallov (Complexons and Complexonates of Metals), Moscow: Khimiya, 1988.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © N.G. Kobylinskaya, E.A. Khainakova, M.E. Diaz-Garcia, V.N. Zaitsev, 2017, published in Fizikokhimiya Poverkhnosti i Zashchita Materialov, 2017, Vol. 53, No. 4, pp. 408–417.
Rights and permissions
About this article
Cite this article
Kobylinskaya, N.G., Khainakova, E.A., Diaz-Garcia, M.E. et al. Nanocomposites based on magnetite modified by chelate groups for a solid-phase concentration of heavy-metal ions from aqueous solutions. Prot Met Phys Chem Surf 53, 675–684 (2017). https://doi.org/10.1134/S2070205117040104
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S2070205117040104