Abstract
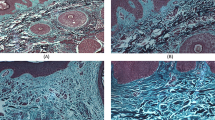
Immunohistochemical analysis was used to study depigmented areas, macular depigmentation, and perimacular areas of the skin of vitiligo patients. It has been shown that cells containing the melanocytic cell marker TRP1 are located in both macular and perimacular areas. Within the macula of depigmentation, all TRP1-positive cells are in close contact with the base membrane. In perimacular areas, many TRP1-positive cells that have lost contact with the base membrane are located in deep layers of epidermis. About 92% of TRP1-positive perimacular cells were also vimentin-positive. Vimentin-positive cells were numerous in perimacular areas, but missing in the macula of depigmentation. Dense groups of cells that were immunopositive for the transcription factor Snail, an inducer of epithelial mesenchymal transition, were localized in perimacular areas in close proximity to the macula of the depigmentation border. Within the macula of depigmentation, such cells were extremely rare. It seems reasonable that an intense process similar to the epithelial mesenchymal transition may be the cause of melanocyte death in the perimacular area and, thus, prevents repigmentation of depigmented areas.
Similar content being viewed by others
Abbreviations
- MD:
-
macular depigmentation
- EMT:
-
epithelial mesenchymal transition
- GAP43:
-
growth-associated protein, a marker of the peripheral nerve system
- Snail:
-
transcription factor and repressor of E-cadherin transcription
- TRP1:
-
tyrosinerelated protein 1
- SOX10:
-
transcription factor, a melanocytedifferentiation marker
References
Al’Abadie, M.S.K., Senior, H.J., Bleehen, S.S., and Gawkrodger, D.J., Neuropeptide and neuronal marker studies in vitiligo, Br. J. Dermatol., 1994, vol. 131, pp. 160–165.
Hann, S.K., Park, Y.K., Lee, K.G., Choi, E.H., and Im, S., Epidermal changes in active vitiligo, J. Dermatol., 1992, vol. 19, pp. 217–222.
Kalluri, R. and Neilson, E.G., Epithelial–mesenchymal transition and its implications for fibrosis, J. Clin. Invest., 2003, vol. 112, pp. 1776–1784.
Koshevenko, Yu.N., Vitiligo (Vitiligo), Moscow: Kosmetika i medicina, 2002.
Lauria, G., Cornblath, D.R., Johansson, O., McArthur, J.C., Mellgren, S.I., Nolano, M., Rosenberg, N., and Sommer, C., EFNS guidelines on the use of skin biopsy in the diagnosis of peripheral Neuropathy, Eur. J. Neurol., 2005, vol. 12, pp. 747–758.
Le Poole, I.C., van den Wijngaard, R.M., Westerhof, W., Dutrieux, R.P., and Das, P.K., Presence or absence of melanocytes in vitiligo lesions: an immunohistochemical investigation, J. Invest. Dermatol., 1993, vol. 100, pp. 816–822.
Lomonosov, K.M., Immunopathogenesis and therapy of vitiligo by means of immunomodulator neovir, Ross. Zh. Kozhn. Venerich. Bol., 2010, vol. 2, pp. 36–39.
Morohashi, M., Hashimoto, K., Goodman, T.F.Jr., Newton, D.E., and Rist, T., Ultrastructural studies of vitiligo, Vogt–Koyanagi syndrome, and incontinentia pigmenti achromians, Arch. Dermatol., 1977, vol. 113, pp. 755–766.
Nieto, M.A., The snail superfamily of zinc-finger transcription factors, Nat. Rev. Mol. Cell. Biol., 2002, vol. 23, pp. 155–166.
Panuncio, A.L. and Vignale, R., Ultrastructural studies in stable vitiligo, Am. J. Dermatopathol., 2003, vol. 25, pp. 16–20.
Plettenberg, A., Ballaun, C., Pammer, J., Mildner, M., Strunk, D., Weninger, W., and Tschachler, E., Human melanocytes and melanoma cells constitutively express the Bcl-2 proto-oncogene in situ and in cell culture, Am. J. Pathol., 1995, vol. 146, pp. 651–659.
Seleit, I., Bakry, O.A., Abdou, A.G., and Dawoud, N.M., Immunohistochemical study of melanocyte-melanocyte stem cell lineage in vitiligo; a clue to interfollicular melanocyte stem cell reservoir, Ultrastruct. Pathol., 2014, vol. 38, pp. 186–198.
Thiery, J.P., Epithelial–mesenchymal transitions in tumour progression, Nat. Rev. Cancer, 2002, vol. 2, pp. 442–454.
Tobin, D.J., Swanson, N.N., Pittelkow, M.R., Peters, E.M., and Schallreuter, K.U., Melanocytes are not absent in lesional skin of long duration vitiligo, J. Pathol., 2000, vol. 191, pp. 407–416.
Wagner, R.Y., Luciani, F., Cario-André, M., Rubod, A., Petit, V., Benzekri, L., Ezzedine, K., Lepreux, S., Steingrimsson, E., Taieb, A., Gauthier, Y., Larue, L., and Delmas, V., Altered E-cadherin levels and distribution in melanocytes precede clinical manifestations of vitiligo, J. Invest. Dermatol., 2015, vol. 135, pp. 1810–1819.
Xiao, D., Barry, S., Kmetz, D., Egger, M., Pan, J., Rai, S.N., Qu, J., McMasters, K.M., and Hao, H., Melanoma cell-derived exosomes promote epithelial-mesenchymal transition in primary melanocytes through paracrine/ autocrine signaling in the tumor microenvironment, Cancer Lett., 2016, vol. 376, pp. 318–327.
Yu, R., Huang, Y., Zhang, X., and Zhou, Y., Potential role of neurogenic inflammatory factors in the pathogenesis of vitiligo, J. Cutan. Med. Surg., 2012, vol. 16, pp. 230–244.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.V. Revishchin, D.Yu. Panteleev, L.G. Zaharova, K.M. Lomonosov, G.V. Pavlova, 2017, published in Tsitologiya, 2017, Vol. 59, No. 1, pp. 19–26.
Rights and permissions
About this article
Cite this article
Revishchin, A.V., Panteleev, D.Y., Zaharova, L.G. et al. An immunohistochemical study of depigmented skin of vitiligo patients. Cell Tiss. Biol. 11, 300–307 (2017). https://doi.org/10.1134/S1990519X17040101
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X17040101