Abstract
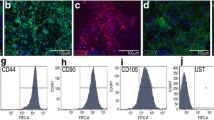
The distribution of bone-marrow stromal cells (BMSC) was studied in 3D polylactide scaffolds. Seeding of cells into the scaffold by the dynamic method (with the aid of a peristaltic pump) has been shown to provide distribution of cells throughout the entire scaffold volume, unlike the static method of seeding, in which the cell suspension is applied onto the scaffold surface. Unlike the cells seeded into the scaffold by the dynamic method, the cells seeded by the static method practically completely migrate from the scaffold on the dish for the first several days. It is revealed that BMSCs cultivated in 3D polylactide scaffolds modified by fibrin form colonies, whereas BMSCs cultivated inside scaffolds modified by collagen type 1 distribute all over the scaffold volume in the form of individual cells.
Similar content being viewed by others
Abbreviations
- BMSC:
-
bone-marrow stromal cell
References
Bauwens, C.L., Peerani, R., Niebruegge, S., Woodhouse, K.A., Kumacheva, E., Husain, M., and Zandstra, P.W., Control of human embryonic stem cell colony and aggregate size heterogeneity influences differentiation trajectories, Stem Cells, 2008, vol. 26, pp. 2300–2310.
Boyce, S.T. and Warden, G.D., Principles and practices for treatment of cutaneous wounds with cultured skin substitutes, Amer. J. Surgery, 2002, vol. 183, pp. 445–456.
Capes, J.S., Ando, H.Y., and Camron, R.E., Fabrication of polymeric scaffolds with a controlled distribution of pores, J. Mater. Sci., Mater. Med., 2005, vol. 16, pp. 1069–1075.
Carolyn, S.A., Reed, M.W.R., and Brown, N.J., A critical analysis of current in vitro and in vivo angiogenesis assays, Int. J. of Exper. Pathol., 2009, vol. 90, pp. 195–221.
Chandrakasan, G., Torchia, D.A., and Piez, K.A., Preparation of intact monomeric collagen from rat tail tendon and skin and the structure of the nonhelical ends in solution, J. Biol. Chem., 1967, vol. 251, pp. 6062–6067.
Choi, S-W., Xie, J., and Xia, Y., Chitosan-based inverse opals: three-dimensional scaffolds with uniform pore structures for cell culture, Adv. Mat., 2009, vol. 21, pp. 2997–3001.
Dai, N.T., Williamson, M.R., Khammo, N., Adams, E.F., and Coombes, A.G., Composite cell support membranes based on collagen and polycaprolactone for tissue engineering of skin, Biomaterials, 2004, vol. 25, pp. 4263–4271.
Dar, A., Shachar, M., Leor, J., and Cohen, S., Optimization of cardiac cell seeding and distribution in 3D porous alginate scaffolds, Biotechnol. Bioeng., 2002, vol. 80, pp. 305–312.
Dvorak, H.F., Harvey, V.S., Estrella, P., Brown, L.F., McDonagh, J., and Dvorak, A.M., Fibrin containing gels induce angiogenesis. Implications for tumor stroma generation and wound healing, Lab. Invest., 1987, vol. 57, pp. 673–686.
Hou, Q.P., Grijpma, D.W., and Feijen, J., Porous polymeric structures for tissue engineering prepared by a coagulation, compression moulding and salt leaching technique, Biomaterials, 2003, vol. 24, pp. 1937–1947.
Khademhosseini, A., Langer, R., Borenstein, J., and Vacanti, J.P., Microscale technologies for tissue engineering and biology, Proc. Natl. Acad. Sci. USA, 2006, vol. 103, pp. 2480–2487.
Khetani, S.R. and Bhatia, S.N., Engineering tissues for in vitro applications, Curr. Opin. Biotechnol., 2005, vol. 17, pp. 524–531.
Mikos, A.G., Thorsen, A.J., Czerwonka, L.A., Bao, Y., Langer, R., Winslow, D.N., and Vacanti, J.P., Preparation and characterization of poly(L-lactic acid) foams, Polymer, 1994, vol. 35, pp. 1068–1077.
Nikolaenko, N.S., Tsypkina, N.V., Pinaev, G.P., Dulaev, A.K., Deev, R.V., and Gololobov, V.G., Isolation and culture of bone marrow stromal cells with a view to their use in treating bone defects, Transplantologiya, 2003, vol. 4, no. 1, pp. 169–171.
Ruszczak, Z.B., Modern aspects of wound healing: an update, Dermatol. Surg., 2000, vol. 26, pp. 219–229.
Rybarczyk, B.J., Lawrence, S.O., and Simpson-Haidaris, P.J., Matrix-fibrinogen enhances wound closure by increasing both cell proliferation and migration, Blood, 2003, vol. 102, pp. 4035–4043.
Shi, G.X., Wang, S.G., and Bei, J.Z., Preparation of porous cell scaffolds of poly(L-lactic acid) and poly(L-lactic-go-glycolic acid) and measurement of their porosity, J. Funct. Polymer., 2001, vol. 14, pp. 7–11.
Shved, Yu.A., Kukhareva, L.B., Zorin, I.M., Solovyov, A.Yu., Blinova, M.I., Bilibin, A.Yu., and Pinaev, G.P., Cultivation of human dermal fibroblasts on the polylactide polymer films, Tsitologiia, 2006, vol. 48, no. 2, pp. 161–168.
Shved, Yu.A., Zorin, I.M., Bilibin, A.Yu., Kukhareva, L.B., Blinova, M.I., and Pinaev, G.P., Interaction of cultured skin cells with the polylactide matrix coved with different collagen structural isoforms, Cell Tissue Biol., 2007, vol. 1, pp. 89–95.
Takei, A., Tashiro, Y., Nakashima, Y., and Sueishi, K., Effects of fibrin on the angiogenesis in vitro of bovine endothelial cells in collagen gel, In Vitro Cell. Devel. Biol. Animal., 1995, vol. 31, pp. 467–472.
Vunjak-Novakovic, G., Obradovic, B., Martin, I., Bursac, P.M., Langer, R., and Freed, L.E., Dynamic cell seeding of polymer scaffolds for cartilage tissue engineering, Biotechnol. Prog., 1998, vol. 14, pp. 193–202.
Whang, K., Healy, K.E., Elenz, D.R., Nam, E.K., Tsai, D.C., Thomas, C.H., Nuber, G.W., Glorieux, F.H., Travers, R., and Sprague, S.M., Engineering bone regeneration with bioabsorbable scaffolds with novel microarchitecture, Tis. Eng., 1999, vol. 5, pp. 35–51.
Yu, D.G., Lin, W.C., and Yang, M.C., Surface modification of poly(L-lactic acid) membrane via layer-by-layer assembly of silver nanoparticle-embedded polyelectrolyte multilayer, Bioconjugate Chem., 2007, vol. 18, pp. 1521–1529.
Zhu, H., Ji, J., and Shen, J., Surface engineering of poly(D,L-lactic acid) by entrapment of biomacromolecules, Macromol. Rapid Comm., 2002, vol. 23, pp. 819–823.
Zhu, X.H., Arifin, D.Y., Khoo, B.H., Hua, J., and Wang, C-H., Study of cell seeding on porous poly(D,L-lactic-co-glycolic acid) sponge and growth in a Couette-Taylor bioreactor, Chem. Eng. Science, 2010, vol. 65, pp. 2108–2117.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © Yu.A. Nashchekina, P.O. Nikonov, V.M. Mikhailov, G.P. Pinaev, 2014, published in Tsitologiya, 2014, Vol. 56, No. 4, pp. 283–290.
Rights and permissions
About this article
Cite this article
Nashchekina, Y.A., Nikonov, P.O., Mikhailov, V.M. et al. Distribution of bone-marrow stromal cells in a 3D scaffold depending on the seeding method and the scaffold inside a surface modification. Cell Tiss. Biol. 8, 313–320 (2014). https://doi.org/10.1134/S1990519X14040075
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X14040075



