Abstract
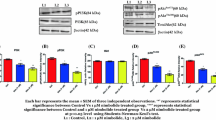
In the present study, 1-(3,5-dimethylphenyl)-6-methyl-1H-pyrazolo[4,3-c]pyridin-4 (5H)-one (DPMPP) was investigated as an antiproliferative agent for prostate cancer cells and the mechanism of its action was studied. Cell lines 22Rv1 and SGC‑7901 were used as in vitro models of prostate cancer. The DPMPP treatment inhibited proliferation of 22Rv1 and SGC‑7901 cells in dose-depended manner. The viability of 22Rv1 and SGC‑7901 cells was reduced to 21 and 19%, respectively after treatment with 32 µM DPMPP. In DPMPP treated (16 µM) 22Rv1 and SGC‑7901 cells apoptosis increased to 62.78 and 68.51%, respectively. Moreover, DPMPP treatment caused cell cycle arrest in S phase and inhibition of PI3K/AKT activation. In the same time ROS production showed elevation and MMP (Matrix MetalloProteinase) decreased in the cells. Apparently DPMPP induces cytotoxicity through induction of oxidative response and apoptosis in prostate cancer cells in vitro. The PI3K/Akt/ERK phosphorylation was inhibited, while p21 and p53, death receptor, expression was promoted by DPMPP treatment. Therefore, DPMPP has a potential to be used as a therapeutic agent for treatment of prostate cancer.








Similar content being viewed by others
REFERENCES
Jemal, A., Siegel, R., and Xu, J.E., Ward Cancer statistics, 2010, Cancer J. Clin., 2010, vol. 60, pp. 277–300.
Fedorov, A., Fluckiger, J., Ayers, G.D., Li, X., Gupta, S.N., Tempany, C., Mulkern, R., Yankeelov, T.E., and Fennessy, F.M., A comparison of two methods for estimating DCE-MRI parameters via individual and cohort based AIFs in prostate cancer: a step towards practical implementation, Magn. Reson. Imaging, 2014, vol. 32, pp. 321–329.
Yu, E.Y., Massard, C., Gross, M.E., Carducci, M.A., Culine, S., Hudes, G., Posadas, E.M., Sternberg, C.N., Wilding, G., Trudel, G.C., et al., Once-daily dasatinib: expansion of phase II study evaluating safety and efficacy of dasatinib in patients with metastatic castration-resistant prostate cancer, Urology, 2011, vol. 77, pp. 1166–1171.
Shetty AV, Thirugnanam S, Dakshinamoorthy G, et al: 18a-glycyrrhetinic acid targets prostate cancer cells by down-regulating inflammation-related genes, Int. J. Oncol., 2011, vol. 39, pp. 635–640.
Maciej, D., Vishal, C.K., Valeria, N., Roberto, F., Kenji, S., Leonidas, E., Dominik, L., et al., Structure–activity relationship in pyrazolo[4,3-c]pyridines, first inhibitors of PEX14–PEX5 protein–protein interaction with trypanocidal activity, J. Med. Chem., 2020, vol. 63, pp. 847–879.
Smyth, L.A., Matthews, T.P., Horton, P.N., Hursthouse, M.B., and Collins, I., Synthesis and reactivity of 3-amino-1H-pyrazolo[4,3-c]pyridin-4(5H)-ones: development of a novel kinase-focused library, Tetrahedron, 2010, vol. 66, no. 15, pp. 2843–2854.
Choi, Y.-M., Kim, H.-K., Shim, W., Anwar, M.A., Kwon, J.-W., Kwon, H.-K., et al., Mechanism of cisplatin-induced cytotoxicity is correlated to impaired metabolism due to mitochondrial ROS generation, PLoS One, 2015, vol. 10, no. 8. e0135083.
Hamid, S.M., Cicek, S., Karamil, S., Ozturk, M.B., Debelec-Butuner, B., Erbaykent-Tepedelen, B., Varisli, L., Gonen-Korkmaz, C., Yorukoglu, K., and Korkmaz, K.S., HOXB13 contributes to G1/S and G2/M checkpoint controls in prostate, Mol. Cell Endocrinol., 2014, vol. 383, pp. 38–47.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xue, J., Zhang, Z. & Hu, H. Prostate Cancer Growth Inhibition by 1-(3,5-Dimethylphenyl)–6-methyl-1H-pyrazolo[4,3-c]pyridin-4(5H)-one via Down-regulation of Phosphorylation PI3K/AKT and STA3/JAK2. Dokl Biochem Biophys 495, 347–353 (2020). https://doi.org/10.1134/S160767292006006X
Published:
Issue Date:
DOI: https://doi.org/10.1134/S160767292006006X