Abstract
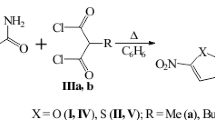
A preparative procedure has been developed for the synthesis of a series of new medicinally relevant 3-aryl-5-(aryloxymethyl)-1,2,4-oxadiazoles in 46–66% yields by alkylation of substituted phenols with 3-aryl-5-(chloromethyl)-1,2,4-oxadiazoles in the system K2CO3/KI/DMF. Poorly studied 3-R-4H-1,2,4-oxadiazin-5(6H)-ones were synthesized by a new method based on the reaction of methyl chloroacetate with amidoximes in the superbasic system t-BuONa/DMSO. The synthesized compounds at concentrations of up to 250 μg/mL showed no antibacterial activity against sensitive strains of Staphylococcus aureus, Bacillus subtilis, Escherichia coli, and Pseudomonas fluorescens, which supposedly suggests their low toxicity for human intestinal and mucous microflora.



Similar content being viewed by others
REFERENCES
Gudipati, R., Anreddy, R.N., and Manda, S., Saudi Pharm. J., 2011, vol. 19, p. 153. https://doi.org/10.1016/j.jsps.2011.03.002
Gan, X., Hu, D., Chen, Z., Wang, Y., and Song, B., Bioorg. Med. Chem. Lett., 2017, vol. 27, p. 4298. https://doi.org/10.1016/j.bmcl.2017.08.038
Rane, R.A., Gutte, S.D., and Sahu, N.U., Bioorg. Med. Chem. Lett., 2012, vol. 22, p. 6429. https://doi.org/10.1016/j.bmcl.2012.08.061
Rajak, H., Agarawal, A., Parmar, P., Thakur, B.S., Veerasamy, R., Sharma, P.C., and Kharya, M.D., Bioorg. Med. Chem. Lett., 2011, vol. 21, p. 5735. https://doi.org/10.1016/j.bmcl.2011.08.022
Vaidya, A., Jain, S., and Jain, P., Mini-Rev. Med. Chem., 2016, vol. 16, p. 825. https://doi.org/10.2174/1389557516666160211120835
Krasavin, M., Shetnev, A., Sharonova, T., Baykov, S., Kalinin, S., Nocentini, A., Sharoyko, V., Poli, G., Tuccinardi, T., Presnukhina, S., Tennikova, T., and Supuran, C., Eur. J. Med. Chem., 2019, vol. 164, p. 92. https://doi.org/10.1016/j.ejmech.2018.12.049
Krasavin, M., Shetnev, A., Sharonova, T., Baykov, S., Tuccinardi, T., Kalinin, S., and Supuran, C.T., Bioorg. Chem., 2018, vol. 76, p. 88. https://doi.org/10.1016/j.bioorg.2017.10.005
Lakshmithendrala, K., Saravanan, K., Elancheran, R., Archana, K., Manikandan, N., Arjun, H.A., Ramanathan, M., Lokanath, N.K., and Kabilan, S., Eur. J. Med. Chem., 2019, vol. 168, p. 1. https://doi.org/10.1016/j.ejmech.2019.02.033
Sun, H., Tawa, G., and Wallqvist, A., Drug Discovery Today, 2012, vol. 17, p. 310. https://doi.org/10.1016/j.drudis.2011.10.024
Amarasinghe Kande, K.D., Matthew, M.B., Srivastava, A., and Gray, J.L., Tetrahedron Lett., 2006, vol. 47, p. 3629. https://doi.org/10.1002/chin.200636130
Augustine, J.K., Akabote, V., Hegde, S.G., and Alagarsamy, P., J. Org. Chem., 2009, vol. 74, p. 5640. https://doi.org/10.1021/jo900818h
Deegan, T.L., Nitz, T.J., Cebzanov, D., Pufko, D.E., and Porco, J.A., Jr., Bioorg. Med. Chem. Lett., 1999, vol. 9, p. 209. https://doi.org/10.1016/s0960-894x(98)00712-4
Howe, R.K. and Schleppnik, F.M., J. Heterocycl. Chem., 1982, vol. 19, p. 721. https://doi.org/10.1002/jhet.5570190404
Nishiwaki, N., Kobiro, K., Hirao, S., Sawayama, J., Saigo, K., Ise, Y., Okajima, Y., and Ariga, M., Org. Biomol. Chem., 2011, vol. 9, p. 6750. https://doi.org/10.1039/C1OB05682D
Baykov, S., Sharonova, T., Osipyan, A., Rozhkov, S., Shetnev, A., and Smirnov, A., Tetrahedron Lett., 2016, vol. 26, p. 2898. https://doi.org/10.1016/j.tetlet.2016.05.071
Shetnev, A.A., Pankratieva, V.E., Kunichkina, A.S., Vlasov, A.S., Proskurina, I.K., Kotov, A.D., and Korsakov, M.K., Russ. J. Org. Chem., 2020, vol. 56, p. 1181. https://doi.org/10.31857/S0514749220070095
Sharonova, T., Pankrat’eva, V., Savko, P., Baykov, S., and Shetnev, A., Tetrahedron Lett., 2018, vol. 59, p. 2824. https://doi.org/10.1016/j.tetlet.2018.06.019
Trofimov, B.A., Schmidt, E.Yu., Vasil’tsov, A.M., Mikhaleva, A.I., Zaitsev, A.B., Morozova, L.V., Gorshkov, A.G., Henkelmann, J., and Arndt, J.-D., Synthesis, 2001, vol. 16, p. 2427. https://doi.org/10.1055/s-2001-18721
Shetnev, A., Baykov, S., Kalinin, S., Belova, A., Sharoyko, V., Rozhkov, A., and Krasavin, M., Int. J. Mol. Sci., 2019, vol. 20, p. 1699. https://doi.org/10.3390/ijms20071699
Shetnev, A., Osipyan, A., Baykov, S., Sapegin, A., Chirkova, Z., Korsakov, M., Petzer, A., Engelbrecht, I., and Petzer, J.P., Bioorg. Med. Chem. Lett., 2019, vol. 29, p. 40. https://doi.org/10.1016/j.bmcl.2018.11.018
Tarasenko, M., Sidneva, V., Belova, A., Romanycheva, A., Sharonova, T., Baykov, S., Shetnev, A., and Kuznetsov, M., Arkivoc, 2018, vol. 2018, part (vii), p. 458. https://doi.org/10.24820/ark.5550190.p010.760
Shetnev, A.A. and Zubkov, F.I., Chem. Heterocycl. Compd., 2017, vol. 53, p. 495. https://doi.org/10.1007/s10593-017-2081-1
Ke, S., Cao, X., Liang, Y., Wang, K., and Yang, Z., Mini-Rev. Med. Chem., 2011, vol. 11, p. 642. https://doi.org/10.2174/138955711796268769
Dürüst, Y., Karakus, H., Kaiser, M., and Tasdemir, D., Eur. J. Med. Chem., 2012, vol. 48, p. 296. https://doi.org/10.1016/j.ejmech.2011.12.028
Poplavskaya, I.A. and Kurmangalieva, R.G., Khimiya amidoksimov (Chemistry of Amidoximes). Alma-Ata: Nauka, 1988.
Poroikov, V.V., Filimonov, D.A., Gloriozova, T.A., Lagunin, A.A., Druzhilovskiy, D.S., Rudik, A.V., Stolbov, L.A., Dmitriev, A.V., Tarasova, O.A., Ivanov, S.M., and Pogodina, P.V., Russ. Chem. Bull., Int. Ed., 2019, vol. 68, p. 2143. https://doi.org/10.1007/s11172-019-2683-0
Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically, Approved Standard, 9th edn. CLSI document M07-A9, Clinical and Laboratory Standards Institute, USA, 2012.
Hussein, Q.A., Heterocycles, 1987, vol. 26, p. 163. https://doi.org/10.3987/r-1987-01-0163
Funding
Studies in the field of synthesis of the target compounds were performed under financial support by the Russian Foundation for Basic Research (project no. 19-33-600-64). Biological activity of the synthesized compounds was studied under financial support by the Ministry of Education of the Russian Federation (project no. 073-00077-21-02, “Development of an Innovative Drug for the Treatment of Open-Angle Glaucoma via Selective Inhibition of Carbonic Anhydrase II”; state registry no. 730000F.99.1.BV10AA00006).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare the absence of conflict of interest.
Additional information
Translated from Zhurnal Organicheskoi Khimii, 2022, Vol. 58, No. 3, pp. 293–302 https://doi.org/10.31857/S0514749222030077.
Rights and permissions
About this article
Cite this article
Shetnev, A.A., Vasilieva, E.A., Proskurina, I.K. et al. Synthesis and Biological Activity of 3-Aryl-5-(aryloxymethyl)-1,2,4-oxadiazoles. Russ J Org Chem 58, 306–314 (2022). https://doi.org/10.1134/S1070428022030071
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428022030071