Abstract
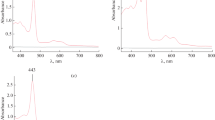
5-Mononitro-2,3,7,8,12,13,17,18-octaethylporphyrin, 5,10,15-trinitro-2,3,7,8,12,13,17,18-octaethylporphyrin, and their Cd(II) complexes were synthesized. The coordination reactions of 2,3,7,8,12,13,17,18-octaethylporphyrin and its mono- and trinitro-substituted derivatives and the metal-exchange reactions of the synthesized Cd(II) octaethylporphyrin complexes with Mn(II) chloride in DMF were investigated by spectrophotometry. The corresponding Mn(II) and Mn(III) porphyrins were obtained and identified. The kinetic parameters of the studied reactions were determined.





Similar content being viewed by others
REFERENCES
Li, L.-L. and Diau, E.W.-G., Chem. Soc. Rev., 2013, vol. 42, p. 291. https://doi.org/10.1039/C2CS35257E
Stuzhin, P.A., Goryachev, M.Yu., Ivanova, S.S, Nazarova, A., Pimkov, I., and Koifman, O.I., J. Porphyr. Phthalocyanines, 2013, vol. 17, p. 905. https://doi.org/10.1142/S1088424613500892
Luguya, R., Jaquinod, L., Fronczek, F.R., Vicente, M.G.H., and Smith, K.M., Tetrahedron, 2004, vol. 60, p. 2757. https://doi.org/10.1016/j.tet.2004.01.080
Lyubimtsev, A., Semeikin, A., Zheglova, N., Sheinin, V., Kulikova, O., and Syrbu, S., Macroheterocycles, 2018, vol. 11, p. 103. https://doi.org/10.6060/mhc171151
Gong, L.Ch. and Dolphin, D., Can. J. Chem., 1985, vol. 63, p. 401. https://doi.org/10.1139/v85-066
Watanabe, E., Nichimura, S., Ogoshi, H., and Yoshida, Z., Tetrahedron, 1975, vol. 31, p. 1385. https://doi.org/10.1016/0040-4020(75)87068-2
Yatsimirskii, K.B., Vvedenie v bioneorganicheskuyu khimiyu (Introduction to Bioinorganic Chemistry), Kiev: Naukova Dumka, 1976.
da Silva, V.S., Teixeira, L.I., do Nascimentoa, E., Idemoria, Y.M., and De Freitas-Silva, G., Appl. Catal. A: Gen., 2014, vol. 469, p. 124. https://doi.org/10.1016/j.apcata.2013.09.033
Chizhova, N.V., Zvezdina, S.V., and Kataleva, Y.S., Russ. J. Gen. Chem., 2015, vol. 85, p. 1132. https://doi.org/10.1134/S1070363215050217
Chizhova, N.V., Maltceva, O.V., Zvezdina, S.V., Mamardashvili, N.Z., and Koifman, O.I., J. Coord. Chem., 2018, vol. 71, p. 3222. https://doi.org/10.1080/00958972.2018.1519186
Cheng, B., Cukiernik, F., Fries, P.H., Marchon, J.C., and Scheidt, W.R., Inorg. Chem., 1995, vol. 34, p. 4627. https://doi.org/10.1021/ic00122a020
Berezin, B.D., Koordinatsionnye soedineniya porfirinov i ftalotsianina (Coordination Compounds of Porphyrins and Phthalocyanine), Moscow: Nauka, 1978.
Mamardashvili, G.M., Mamardashvili, N.Zh., and Koifman, O.I.,Russ. Chem. Rev., 2008, vol. 77, p. 59. https://doi.org/10.1070/RC2008v077n01ABEH003743
Berezin, B.D. and Enikolopyan, N.S., Metalloporfiriny (Metalloporphyrins), Moscow: Nauka, 1988.
Berezin, B.D., Rumyantseva, S.V., and Berezin, M.B., Russ. J. Coord. Chem., 2004, vol. 30, p. 291. https://doi.org/10.1023/B:RUCO.0000022806.85114.93
Boucher, L.J., Coord. Chem. Rev., 1972, vol. 7, p. 289. https://doi.org/10.1016/S0010-8545(00)80024-7
Doro, F.G., Lindsay Smith, J.R., Ferreira, A.G., and Assis, M.D.,J. Mol. Catal. Chem., 2000, vol. 164, p. 97. https://doi.org/10.1016/S1381-1169(00)00352-6
Berezin, B.D., Karmanova, T.V., Gromova, T.V., Syrbu, S.A., and Semeikin, A.S., Russ. J. Coord. Chem., 2002, vol. 28, p. 608. https://doi.org/10.1023/A:1020030714257
Hambright, R., The Porphyrin Handbook, Chemistry of Water Soluble Porphyrins. Kadish, K.M., Smith, K.M., and Guilard, R., Eds., New York: Academic Press, 2000, vol. 3, p. 129.
Yatismirskii, K.B. and Lampeka, Ya.D., Fizikokhimiya kompleksov metallov s makrotsilicheskimi ligandami (Physicochemistry of Metal Complexes with Macrocyclic Ligands), Kiev: Naukova Dumka, 1985.
Basolo, F. and Pearson, R.G., Mechanisms of Inorganic Reactions a Study of Metal Complexes in Solution, New York: John Wiley and Sons, Inc., 1967.
Gubin, S.P., Khimiya klasterov. Osnovy klassifikatsii i stroeniya (Cluster chemistry. Basics of Classification and Structure), Moscow: Nauka, 1987.
Entelis, S.G., Kinetika reaktsii v zhidkoi faze (Kinetics of Reactions in the Liquid Phase), Entelis, S.G. and Tiger, R.P., Eds., Moscow: Khimiya, 1973, p. 416.
Chizhova, N.V., Khelevina, J.G., and Berezin, B.D., Russ. J. Gen. Chem., 2003, vol. 73, p. 1645. https://doi.org/10.1023/B:RUGC.0000016039.52090.d2
Linstead, R.P. and Weiss, F.T., J. Chem. Soc., 1950, p. 2975. https://doi.org/10.1039/JR9500002975
Cook, A.H. and Linstead, R.P., J. Chem. Soc., 1937, p. 929. https://doi.org/10.1039/JR9370000929
Ali, B.B., Belkhiria, M.S., Giorgi, M., and Nasri, H., Open J. Inorg. Chem., 2011, p. 39. https://doi.org/10.4236/ojic.2011.13006
Zvezdina, S.V., Berezin, M.B., and Berezin, B.D., Russ. J. Inorg. Chem., 2007, vol. 52, p. 1269. https://doi.org/10.1134/S0036023607080177
Zvezdina, S.V. and Mamardashvili, N.Zh., Russ. J. Coord. Chem., 2012, vol. 38, p. 323. https://doi.org/10.1134/S1070328412050120
ACKNOWLEDGMENTS
The work was performed using the equipment of the “Upper Vologa Regional Center for Physical and Chemical Research” Center for Collective Use.
Funding
The work was financially supported by the Russian Foundation for Basic Research: project no. 19-03-00078_A [synthesis of porphyrin ligands and their Cd(II), Mn(II), and Mn(III) complexes] and project no. 1843370001_r-a [study of the metal-exchange reaction of the Cd(II) complexes of the synthesized porphyrins with Mn(II) chloride].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Supplementary material
Rights and permissions
About this article
Cite this article
Zvezdina, S.V., Chizhova, N.V. & Mamardashvili, N.Z. Synthesis and Spectral Properties of Unsymmetrically Substituted Mn(II) and Mn(III) Octaethylporphyrins. Russ J Org Chem 56, 1374–1382 (2020). https://doi.org/10.1134/S1070428020080072
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428020080072