Abstract
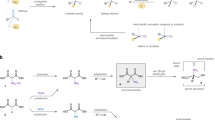
A novel and highly efficient synthetic approach has been developed for the synthesis of deuterium-labeled olopatadine-d6 using inexpensive and commercially available dimethyl sulfate-d6 at the stage of alkylation of primary amine intermediate. The proposed synthetic path makes it possible to avoid the use of expensive labeled reagents such as dimethyl amine-d6 used as labeled precursor in the traditional synthetic route. The structure of the obtained olapatadine-d6 has been confirmed by 1H NMR and mass spectral data.
Similar content being viewed by others
References
Elison, E., Rapoport, H., Laursen, R., and Elliott, H.W., Science, 1961, vol. 134, p. 1078. doi 10.3390/ molecules22101592
Isotope Effects on Enzyme-Catalyzed Reactions (Proc. Sixth Annual Harry Steenbock Symposium, Madison, Wisconsin, June 4 and 5, 1976), Cleland, W.W., O’Leary, M.H., and Northrop, D.B., Eds., Baltimore: University Park Press, 1977.
Gillette, J.R., Darbyshire, J.F., and Sugiyama, Y., Biochemistry, 1994, vol. 33, p. 2927. doi 10.1021/ bi00176a024
Wenzel, M., J. Labelled Compd. Radiopharm., 1989, vol.27, p. 1143. doi 10.1002/jlcr.2580271006
Baker, M.T., Ronnenberg, W.C., Ruzicka, J.A., Chiang, C.K., and Tinker, J.H., Drug Metab. Dispos., 1993, vol. 21, p.1170. PMID 7905401.
Kushner, D.J., Baker, A., and Dunstall, T.G., Can. J. Physiol. Pharmacol., 1999, vol. 77, p. 79. doi 10.1139/ y99-005
Jones, A.D., Zelle, R.E., and Silverman, I.R., US Patent Appl. Pub. no. 2014336387, 2014.
Harbeson, S.L., and Tung, R.D., Int. Patent Appl. Pub. no. WO2008156632, 2008
Gant, T.G., Sarshar, S., and Woo, S.H., US Patent Appl. Pub. no. 20080234257.
Ohshima, E., Otaki, S., Sato, H., Kumazawa, T., Obase, H., Ishii, A., Ishii, H., Ohmori, K., and Hira- yama, N., J. Med. Chem., 1992, vol. 35, no. 11, p. 2074. doi 10.1021/jm00089a020
Abelson, M.B. and Spitalny, L., Am. J. Ophthalmol., 1998, vol. 98, p. 797. doi 10.1016/S0002-9394(98) 00044-0
Kateluris, C.H., Ciprandi, G., Missotten, L., Turner, E.D., Bertin, D., and Berdeaux, G., Clin. Ther., 2002, vol. 24, p. 1561. doi 10.1016/S0149-2918(02) 80060-1
Spangler, D.L., Bensch, G., Berdy, G.J., and Clinic, A., Clin. Ther., 2001 vol. 8, p. 1072. doi 10.1016/S0149-2918(01)80106-5
Ohmori, K., Hayashi, K., Kaise, T., Ohshima, E., and Kobayashi, S., Jpn. J. Pharmacol., 2002, vol. 88, p. 379. doi 10.1254/jjp.88.379
Ashiro, M., Mochizuki, H., Sakurada, Y., Ishii, K., Oda, K., Kimura, Y., Sasaki, T., Ishiwata, K., and Yanai, K., Clin. Pharmacol. Ther., 2006, vol. 61, p. 16. doi 10.1111/j.l365-2125.2005.02514.x0
Hamilton, S.A., Duddle, J., Herdman, M.J., Trigg, C.J., and Davies, R.J., Clin. Exp. Allergy, 1994, vol. 24, p. 955. doi 10.1111/j.1365-2222.1994.tb02728.x
Shimamura, T., Shiroishi, M., Weyand, S., Tsuji-moto, H., Winter, G., and Katritch, V., Nature, 2011, vol. 475, p. 65. doi 10.1038/naturel0236
Ueno, E., Oshima, H., Saito, I., Matsumoto, H., Yoshi-mura, Y., and Nakazawa, H., J. AOAC Int., 2004, vol. 87, p. 1003
Barreda, M., Lopez, F.J., Villarroya, M., Beltran, J., Garcia-Baudin, J.M., and Hernandez, F., J. AOAC Int., 2006, vol. 89, p. 1080
Oshima, E., Kumazawa, T., Otaki, S., Obase, H., Ohmori, K., Ishii, H., Manabe, H., Tamura, T., and Shuto, K., US Patent no. 5116863, 1992.
Lever, W.O., Jr. and Leighton, H.J., EP Patent no. 0214779, 1990.
Bosch Cartes, J., Bachs Roca, J., Gomez Gomez, A.M., Alonso Marin, Y., and Bessa Sanchez, M., Int. Patent Appl. Pub. no. WO 2006010459, 2006.
Tarur, V., Bhise, N., Sathe, D., Naidu, A., Aher, U., Paul, S., Verma, S., Sawant, K., Naik, T., and Amre, R., Int. Patent Appl. Pub. no. WO2007105234 A2, 2007.
Castellin, A., Ferrari, C., and Galvagni, M., US Patent Appl. Pub. no. 20110065936 AI, 2011.
Bosch, J., Bachs, J., Gomez, A.M., Griera, R., Marta, E., and Amat, M., J. Org. Chem., 2012, vol. 77, p. 6340. doi 10.1021/jo300925c
Sano, T., Sugaya, T., and Kasai, M., Org. Process Res. Dev., 1998, vol. 2, p.169. doi 10.1021/op970055u
Nishimura, K. and Kinugawa, M., Org. Process Res. Dev., 2012, vol. 16, p. 225. doi 10.1021/op200312m
Acknowledgments
The authors thank the management of AMRI Hyderabad Research Center for giving an opportunity to carry out this research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare the absence of conflict of interests.
Rights and permissions
About this article
Cite this article
Endoori, S., Gulipalli, K.C., Bodige, S. et al. Novel and Efficient Synthesis of Deuterium-Labeled Olopatadine-d6. Russ J Org Chem 55, 1387–1393 (2019). https://doi.org/10.1134/S1070428019090197
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428019090197