Abstract
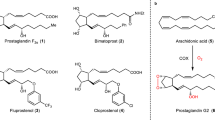
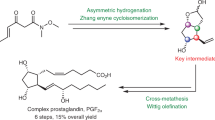
The review covers the literature on the total synthesis of cross-conjugated cyclopentenone prostaglandins (PGs), published from 2000 until present. In view of the specific features differentiating cyclopentenone PGs from prostaglandins of other types, special attention in the review is focused on the biochemistry and physiological functions of the most important representatives of cyclopentenone PGs. Data on analogs of cyclopentenone PGs are presented, and certain aspects of structure–activity correlations and potential practical applications of these compounds are discussed.
Similar content being viewed by others
References
Funk, C.D., Science, 2001, vol. 294, p. 1871. doi https://doi.org/10.1126/science.294.5548.1871
Jahn, U., Galano, J.-M., and Durand, T., Angew. Chem., Int. Ed., 2008, vol. 47, p. 5894. doi https://doi.org/10.1002/anie.200705122
Das, S., Chandrasekhar, S., Yadav, J.S., and Grée, R., Chem. Rev., 2007, vol. 107, p. 3286. doi https://doi.org/10.1021/cr068365a
Rouzer, C.A. and Marnett, L.J., Chem. Rev., 2003, vol. 103, p. 2239. doi https://doi.org/10.1021/cr000068x
Fitzpatrick, F.A. and Wynalda, M.A., J. Biol. Chem., 1983, vol. 258, p. 11713.
Lefils-Lacourtablaise, J., Socorro, M., Géloën, A., Daira, P., Debard, C., Loizon, E., Guichardant, M., Dominguez, Z., Vidal, H., Lagarde, M., and Bernoud-Hubac, N., PLOS One, 2013, vol. 8, p. e63997. doi https://doi.org/10.1371/journal.pone.0063997
Straus, D.S. and Glass, C.K., Med. Res. Rev., 2001, vol. 21, p. 185. doi https://doi.org/10.1002/med.1006
Brunoldi, E.M., Zanoni, G., Vidari, G., Sasi, S., Freeman, M.L., Milne, G.L., and Morrow, J.D., Chem. Res. Toxicol., 2007, vol. 20, p. 1528. doi https://doi.org/10.1021/tx700231a
Uchida, K. and Shibata, T., Chem. Res. Toxicol., 2008, vol. 21, p. 138. doi https://doi.org/10.1021/tx700177j
Pande, V. and Ramos, M.J., Bioorg. Med. Chem. Lett., 2005, vol. 15, p. 4057. doi https://doi.org/10.1016/j.bmcl.2005.06.025
Kudva, A.K., Kaushal, N., Mohinta, S., Kennet, M.J., August, A., Paulson, R.F., and Prabhu, K.S., PLOS One, 2013, vol. 8, p. e80622. doi https://doi.org/10.1371/journal.pone.0080622
Verbitski, S.M., Mullally, J.E., Fitzpatrick, F.A, and Ireland, C.M., J. Med. Chem., 2004, vol. 47, p. 2062. doi https://doi.org/10.1021/jm030448l
Miftakhov, M.S., Adler, M.E., Akbutina, F.A., and Tolstikov, G.A., Russ. Chem. Rev., 1994, vol. 63, p. 543. doi https://doi.org/10.1070/RC1994v063n06ABEH000102
Hubich, A.I. and Sholukh, M.V., Biochem. (Moscow), 2006, vol. 71, p. 229. doi https://doi.org/10.1134/S0006297906030011
Tsukimoto, A., Sugiyama, R., Abe, M., Nishitsuji, H., Shimizu, Y., Shimotohno, K., Kawai, G., and Takaku, H., Antiviral Res., 2015, vol. 117, p. 1. doi https://doi.org/10.1016/j.antiviral.2015.01.013
Diez-Dacal, B. and Pérez-Sala, D., Cancer Lett., 2012, vol. 320, p. 150. doi https://doi.org/10.1016/j.canlet.2012.03.003
Kobayashi, Y., Murugesh, M.G., and Nakano, M., Tetrahedron Lett., 2001, vol. 42, p. 1703. doi https://doi.org/10.1016/S0040-4039(00)02329-7
Kobayashi, Y., Murugesh, M.G., Nakano, M., Takahisa, E., Usmani, S.B., and Ainai, T., J. Org. Chem., 2002, vol. 67, p. 7110. doi https://doi.org/10.1021/jo020375y
Nakata, K. and Kobayashi, Y., Org. Lett., 2005, vol. 7, p. 1319.
Zanoni, G., Porta, A., De Toma, Q., Castronovo, F., and Vidari, G., J. Org. Chem., 2003, vol. 68, p. 6437.
Chapleo, S.B., Finch, M.A.W., Lee, T.V., Roberts, S.M., and Newton, R.F., J. Chem. Soc., Perkin Trans. 1, 1980, p. 2084.
Miller, E., Morel, A., Saso, L., and Saluk, J., Oxid. Med. Cell. Long., 2014, ID 572491, 1. doi https://doi.org/10.1155/2014/572491
Galano, J.-M., Mas, E., Barden, A., Mori, T.A., Signorini, C., De Felice, C., Barrett, A., Opere, C., Pinot, E., Schwedhelm, E., Benndorf, R., Roy, J., Le-Guennec, J.-Y., Oger, C., and Durand, T., Prostagl. Lipid Mediators, 2013, vol. 107, p. 95. doi https://doi.org/10.1016/j.prostaglandins.2013.04.003
Zanoni, G., Brunoldi, E.M., Porta, A., and Vidari, G., J. Org. Chem., 2007, vol. 72, p. 9698.
Subbanagounder, G., Wong, J.W., Lee, H., Faull, K.F., Miller, E., Witztum, J.L., and Berliner, J.A., J. Biol. Chem., 2002, vol. 277, p. 7271. doi https://doi.org/10.1074/jbc.M107602200
Jung, M.E., Berliner, J.A., Angst, D., Yue, D., Koroniak, L., Watson, A.D., and Li, R., Org. Lett., 2005, vol. 7, p. 3933.
Jung, M.E., Berliner, J.A., Koroniak, L., Gugiu, B.G., and Watson, A.D., Org. Lett., 2008, vol. 10, p. 4207. doi https://doi.org/10.1021/ol8014804
Acharya, H.P. and Kobayashi, Y., Tetrahedron Lett., 2005, vol. 46, p. 8435. doi https://doi.org/10.1016/j.tetlet.2005.09.193
Kawashima, H. and Kobayashi, Y., Org. Lett., 2014, vol. 16, p. 2598.
Egger, J., Bretscher, P., Freigang, S., Kopf, M., and Carreira, E.M., Angew. Chem., Int. Ed., 2013, vol. 52, p. 5382. doi https://doi.org/10.1002/anie.201300739
Zhu, C., Shen, X., and Nelson, S.G., J. Am. Chem. Soc., 2004, vol. 126, p. 5352.
Acharya, H.P. and Kobayashi, Y., Angew. Chem., Int. Ed., 2005, vol. 44, p. 3481. doi https://doi.org/10.1002/anie.200500534
Yan, X., Lee, S., Gugiu, B.G., Koroniak, L., Jung, M.E., Berliner, J., Cheng, J., and Li, R., Biochem. Biophys. Res. Commun., 2014, vol. 444, p. 69. doi https://doi.org/10.1016/j.bbrc.2014.01.016
Egger, J., Bretscher, P., Freigang, S., Kopf, M., and Carreira, E.M., J. Am. Chem. Soc., 2014, vol. 136, p. 17382.
Abbasi, S., Kajimoto, K., and Harashima, H., Int. Ed. Nanomed., 2016, vol. 11, p. 2685.
Yagami, T., Yamamoto, Y., and Koma, H., Mol. Neurobiol., 2018, vol. 55, p. 2227. doi https://doi.org/10.1007/s12035-017-0435-4
Berger, J. and Moller, D.E., Ann. Rev. Med., 2002, vol. 53, p. 409.
Lee, E., Yin, Z., Sidoryk-Wegrzynowicz, M., Jiang, H., and Aschner, M., Free Rad. Biol. Med., 2012, vol. 52, p. 1067. doi https://doi.org/10.1016/j.freeradbiomed.2011.12.016
Rasin, M.S., Pulmonologiya, 2013, vol. 2, p. 95.
Scher, J.U. and Pillinger, M.H., Clin. Immunol., 2005, vol. 114, p. 100. doi https://doi.org/10.1016/j.clim.2004.09.008
Brummond, K.M., Sill, P.C., and Chen, H., Org. Lett., 2004, vol. 6, p. 149.
Brummond, K.M., Sill, P.C., Rickards, B., and Geib, S.J., Tetrahedron Lett., 2002, vol. 43, p. 3735. doi https://doi.org/10.1016/S0040-4039(02)00633-0
Fleming, I., Henning, R., Parker, D.C., Plaut, H.E., and Sanderson, P.E.J., J. Chem. Soc., Perkin. Trans. 1, 1995, p. 317.
Acharya, H.P. and Kobayashi, Y., Tetrahedron Lett., 2004, vol. 45, p. 1199. doi https://doi.org/10.1016/j.tetlet.2003.11.143
Acharya, H.P. and Kobayashi, Y., Tetrahedron, 2006, vol. 62, p. 3329. doi https://doi.org/10.1016/j.tet.2006.01.051
Katsuki, T. and Sharpless, K.B., J. Am. Chem. Soc., 1980, vol. 102, p. 5974.
Yang, A., PhD Thesis, Pittsburgh, USA, 2007.
Bickley, J.F., Jadhav, V., Roberts, S.M., Santoro, M.G., Steiner, A., and Sutton, P.W., Synlett., 2003, p. 1170. doi https://doi.org/10.1055/s-2003-39885
Baxter, A.D., Binns, F., Javed, T., Roberts, S.M., Sadler, P., Scheinmann, F., Wakefield, B.J., Lynch, M., and Newton, R.F., J. Chem. Soc., Perkin Trans. 1, 1986, p. 889.
Meinwald, J., Labana, S.S., and Chadha, M.S., J. Am. Chem. Soc., 1963, vol. 85, p. 582. doi https://doi.org/10.1021/ja00888a022
Vostrikov, N.S., Lobko, I.F., and Miftakhov, M.S., Tetrahedron Lett., 2014, vol. 55, p. 5622. doi https://doi.org/10.1016/j.tetlet.2014.08.096
Vostrikov, N.S., Lobko, I.F., Ishimova, D.U., and Miftakhov, M.S., Russ. J. Org. Chem., 2015, vol. 51, p. 1. doi https://doi.org/10.1134/S1070428015010017
Tolstikov, G.A., Miftakhov, M.S., Valeev, F.A., Vostrikov, N.S., and Akhmetvaleev, R.R., Zh. Org. Khim., 1984, vol. 20, p. 221.
Iqbal, M., Li, Y., and Evans, P., Tetrahedron, 2004, vol. 60, p. 2531. doi https://doi.org/10.1016/j.tet.2004.01.048
Iqbal, M., Duffy, P., Evans, P., Cloughley, G., Allan, B., Lledó, A., Verdaguer, X., and Riera, A., Org. Biomol. Chem., 2008, vol. 6, p. 4649. doi https://doi.org/10.1039/B814619E
Gibson, S.E. and Stevenazzi, A., Angew. Chem., Int. Ed., 2003, vol. 42, p. 1800. doi https://doi.org/10.1002/anie.200200547
Blanco-Urgoiti, J., Añorbe, L., Pérez-Serrano, L., Domínguez, G., and Pérez-Castells, J., Chem. Soc. Rev., 2004, vol. 33, p. 32.
Van Staden, L.F., Gravestock, D., and Ager, D.J., Chem. Soc. Rev., 2002, vol. 31, p. 195. doi https://doi.org/10.1039/A908402I
Taber, D.F. and Teng, D., J. Org. Chem., 2002, vol. 67, p. 1607.
Taber, D.F. and Tian, W., J. Org. Chem., 2008, vol. 73, p. 7560.
Nani, R.R., PhD Thesis, Pasadena, USA, 2013.
Egger, J., Fischer, S., Bretscher, P., Freigang, S., Kopf, M., and Carreira, E.M., Org. Lett., 2015, vol. 17, p. 4340.
Kim, E.-H. and Surh, Y.-J., Biochem. Pharm., 2006, vol. 72, p. 1516. doi https://doi.org/10.1016/j.bcp.2006.07.030
Kim, N.-J., Moon, H., Park, T., Yun, H., Jung, J.-W., Chang, D.-J., Kim, D.-D., and Suh, Y.-G., J. Org. Chem., 2010, vol. 75, p. 7458.
Lu, Y., Nguyen, P.L., Lévaray, N., and Lebel, H., J. Org. Chem., 2013, vol. 78, p. 776.
Hegde, S., Kaushal, N., Ravindra, K.C., Chiaro, C., Hafer, K.T., Gandhi, U.H., Thompson, J.T., Van den Heuvel, J.P., Kennett, M.J., Hankey, P., Paulson, R.F., and Prabhu, K.S., Blood, 2011, vol. 118, p. 6909. doi https://doi.org/10.1182/blood-2010-11-317750
Prabhu, K.S., Paulson, R.F., Hegde, S, Kaushal, N., and Gandhi, U.N., WO Patent Appl. no. 2013/003729A1, 2013.
Williams, S.A., Anderson, W.C., Santaguida, M.T., and Dylla, S.J., Lab. Invest., 2013, vol. 93, p. 970. doi https://doi.org/10.1038/labinvest.2013.92
Nicolaou, K.C., Heretsch, P., El Marrouni, A., Hale, C.R.H., Pulukuri, K.K., Kudva, A.K., Narayan, V., and Prabhu, K.S., Angew. Chem., Int. Ed., 2014, vol. 53, p. 10443. doi https://doi.org/10.1002/anie.201404917
Nicolaou, K.C., Pulukuri, K.K., Yu, R., Rigol, S., Heretsch, P., Grove, C.I., Hale, C.R.H., and El Marrouni, A., Chem. Eur. J., 2016, vol. 22, p. 8559. doi https://doi.org/10.1002/chem.201601449
Trost, B.M. and Bunt, R.C., Angew. Chem., Int. Ed., 1996, vol. 35, p. 99. doi https://doi.org/10.1002/anie.199600991
Carreira, E.M., Singer, R.A., and Lee, W., J. Am. Chem. Soc., 1994, vol. 116, p. 8837.
Glaus, F. and Altmann, K.-H., Chimia, 2013, vol. 67, p. 227. doi https://doi.org/10.2533/chimia.2013.227
Frick, J.A., Klassen, J.B., Bathe, A., Abramson, J.M., and Rapoport, H., Synthesis, 1992, vol. 7, p. 621. doi https://doi.org/10.1055/s-1992-26176
Bal, B.S., Childers, W.E., and Pinnick, H.W., Tetrahedron, 1981, vol. 37, p. 2091. doi https://doi.org/10.1016/S0040-4020(01)97963-3
Mercadante, M.A., Kelly, C.B., Bobbitt, J.M., Tilley, L.J., and Leadbeater, N.E., Nature Prot., 2013, vol. 8, p. 666. doi https://doi.org/10.1038/nprot.2013.028
Nicolaou, K.C., Pulukuri, K.K., Rigol, S., Heretsch, P., Yu, R., Grove, C.I., Hale, C.R.H., El Marrouni, A., Fetz, V., Brönstrup, M., Aujay, M., Sandoval, J., and Gavrilyuk, J., J. Am. Chem. Soc., 2016, vol. 138, p. 6550.
Nicolaou, K.C., Heretsch, P., Hale, C.R.H., El Marrouni, A., Pulukuri, K.K., Yu, R., and Grove, C.I., WO Patent Appl. no. 2015/048268, 2015.
Kitade, M., Tanaka, H., Oe, S., Iwashima, M., Iguchi, K., and Takahashi, T., Chem. Eur. J., 2006, vol. 12, p. 1368. doi https://doi.org/10.1002/chem.200500793
Fukushima, S., Kishimoto, S., Takeuchi, Y., and Fukushima, M., Adv. Drug Delivery Rev., 2000, vol. 45, p. 65. doi https://doi.org/10.1016/S0169-409X(00)00101-0
Fukushima, S., Takeuchi, Y., Kishimoto, S., Yamashita, S., Uetsuki, K., Shirakawa, S., Suzuki, M., Furuta, K., Noyori, R., Sasaki H., Kikuchi, Y., Kita, T., Yamori, T., Sawada, J., Kojima, M., Hazato, A., Kurozumi, S., and Fukushima, M., Anti-Cancer Drugs, 2001, vol. 12, p. 221.
Hirata, Y., Furuta, K., Suzuki, M., Oh-hashi, K., Ueno, Y., and Kiuchi, K., Brain Res., 2012, vol. 1482, p. 91. doi https://doi.org/10.1016/j.brainres.2012.09.008
Suzuki, M., Kiho, T., Tomokiyo, K., Furuta, K., Fukushima, S., Takeuchi, Y., Nakanishi, M., and Noyori, R., J. Med. Chem., 1998, vol. 41, p. 3084. doi https://doi.org/10.1021/jm9801657
Furuta, K., Tomokiyo, K., Satoh, T., Watanabe, Y., and Suzuki, M., ChemBioChem, 2000, vol. 1, p. 283. doi https://doi.org/10.1002/1439-7633(20001117)1:4<283::AIDCBIC283>3.0.CO;2-O
Furuta, K., Maeda, M., Hirata, Y., Shibata, S., Kiuchi, K., and Suzuki, M., Bioorg. Med. Chem. Lett., 2007, vol. 17, p. 5487. doi https://doi.org/10.1016/j.bmcl.2006.12.004
Weaving, R., Roulland, E., Monneret, C., and Florent, J.-C., Tetrahedron Lett., 2003, vol. 44, p. 2579. doi https://doi.org/10.1016/S0040-4039(03)00237-5
Łukasik, B., Mikołajczyk, M., Bujacz, G., and Zurawiński, R., Org. Biomol. Chem., 2015, vol. 13, p. 807. doi https://doi.org/10.1039/C4OB01535E
Żuraviwiński, R., Mikina, M., and Mikołajczyk, M., Tetrahedron: Asymmetry, 2010, vol. 21, p. 2794. doi https://doi.org/10.1016/j.tetasy.2010.11.007
Chen, M. and Hartwig, J.F., Angew. Chem., Int. Ed., 2014, vol. 53, p. 8691. doi https://doi.org/10.1002/anie.201403844
Sugiura, M., Kinoshita, R., and Nakajima, M., Org. Lett., 2014, vol. 16, p. 5172.
Dübon, P., Schelwies, M., and Helmchen, G., Chem Eur. J., 2008, vol. 14, p. 6722. doi https://doi.org/10.1002/chem.200800495
Schelwies, M., Dübon, P., and Helmchen, G., Angew. Chem., Int. Ed., 2006, vol. 45, p. 2466. doi https://doi.org/10.1002/anie.200503945
Iqbal, M. and Evans, P., Tetrahedron Lett., 2003, vol. 44, p. 5741. doi https://doi.org/10.1016/S0040-4039(03)01297-8
Arisetti, N. and Reiser, O., Org. Lett., 2015, vol. 17, p. 94.
Żurawiński, R., Mikołajczyk, M., Ciećlak, M., Królewska, K., and Kaźmierczak-Barańska, J., Org. Biomol. Chem., 2015, vol. 13, p. 7000. doi https://doi.org/10.1039/C5OB00550G
Tanaka, H., Hasegawa, T., Kita, N., Nakahara, H., Shibata, T., Oe, S., Ojika, M., Uchida, K., and Takahashi, T., Chem. Asian J., 2006, vol. 1, p. 669. doi https://doi.org/10.1002/asia.200600172
Vostrikov, N.S., lobko, I.F., Spirikhin, L.V., and Miftakhov, M.S., Russ. J. Org. Chem., 2016, vol. 52, p. 1765. doi https://doi.org/10.1134/S1070428016120095
Vostrikov, N.S., Lobko, I.F., Spirikhin, L.V., Vakhitova, Y.V., Pivnitsky, K.K., and Miftakhov, M.S., Mendeleev Commun., 2017, vol. 27, p. 125.
Ratnayake, A.S., Bugni, T.S., Veltri, C.A., Skalicky, J.J., and Ireland, C.M., Org. Lett., 2006, vol. 8, p. 2171.
Mullally, J.E. and Fitzpatrick, F.A., Mol. Pharm., 2002, vol. 62, p. 351.
Woodward, D.F., Wang, J.W., and Poloso, N.J., Pharm. Rev., 2013, vol. 65, p. 1135. doi https://doi.org/10.1124/pr.112.007088
Kozak, K.R., Morrow, B.C., Wang, L.H., Ma, L.H., Weinander, R., Jacobsson, P.J., and Marnett, L., J. Biol. Chem., 2002, vol. 277, p. 44877. doi https://doi.org/10.1074/jbc.M206788200
Ladin, D.A., Soliman, E., Escobedo, R., Fitzgerald, T.L., Yang, L.V., Burns, C., and Van Dross, R., Mol. Cancer Therapeutics, 2017, vol. 16, p. 838. doi https://doi.org/10.1158/1535-7163.MCT-16-0484
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.V. Loza, A.M. Gimazetdinov, M.S. Miftakhov, 2018, published in Zhurnal Organicheskoi Khimii, 2018, Vol. 54, No. 11, pp. 1575–1620.
Rights and permissions
About this article
Cite this article
Loza, V.V., Gimazetdinov, A.M. & Miftakhov, M.S. Cross-Conjugated Cyclopentenone Prostaglandins. Recent Advances. Russ J Org Chem 54, 1585–1629 (2018). https://doi.org/10.1134/S1070428018110015
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428018110015