Abstract
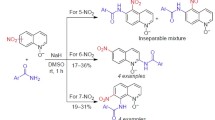
1-Substituted 1H-1,2-diazaphenalenes (1H-benzo[de]cinnolines) undergo nitration with nitric acid leading to the formation of di- and trinitro derivatives. The yield of nitration products depends on nitric acid concentration.
Similar content being viewed by others
References
Pozharskii, A.F. and Dal’nikovskaya, V.V., Russ. Chem. Rev., 1981, vol. 50, p. 816
Mezheritskii, V.V., Khimiya i biologicheskaya aktivnost’ azotistykh geterotsiklov i alkaloidov, Kartsev, V.G. and Tolstikov, G.A., Eds., Moscow: Iridium-press, 2001.
Mezheritskii, V.V., Tkachenko, V.V., Zhukovskaya, O.N., Elisevich, D.M., and Dorofeenko, G.N., Zh. Org. Khim., 1981, vol. 17, p. 627.
Elisevich, D.M., Mezheritskii, V.V., and Dorofeenko, G.N., Zh. Org. Khim., 1981, vol. 17, p. 1751.
Elisevich, D.M., Okolelova, M.S., Kazakov, A.L., Drogovoz, S.M., and Luk’yanchikov, M.S., USSR Inventor’s Certificate no. 1467961, 1987.
Pozharskii, A.F. Malysheva, E.N., Chem. Heterocycl. Compd., 1970, vol. 6, p. 99.
Elisevich, D.M., Candidate Sci. (Chem.) Dissertation, Rostov-on-Don, 1982.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.V. Mezeritskii, L.G. Minyaeva, N.I. Omelichkin, G.S. Borodkin, M.S. Korobov, 2015, published in Zhurnal Organicheskoi Khimii, 2015, Vol. 51, No. 5, pp. 689–692.
Rights and permissions
About this article
Cite this article
Mezeritskii, V.V., Minyaeva, L.G., Omelichkin, N.I. et al. Nitration of 3-methyl-1H-1,2-diazaphenalene and its n-substituted derivatives. Russ J Org Chem 51, 670–673 (2015). https://doi.org/10.1134/S1070428015050152
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070428015050152