Abstract
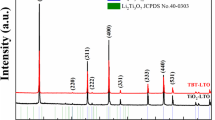
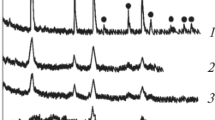
A technique was developed for the synthesis of single-phase lithium titanate of composition Li4Ti5O12 with tetraethoxytitanium as a precursor and citric acid as a complexing agent. The processes occurring during the solution and solid-phase synthesis of Li4Ti5O12 were investigated by the methods of X-ray phase analysis, differential scanning calorimetry, and scanning electron microscopy. The phase composition of the products formed at different temperatures was determined depending on the synthesis technique. It was found that the phase formation of lithium titanate is accompanied by the formation of an intermediate phase Li2TiO3 enriched in lithium.








Similar content being viewed by others
REFERENCES
Hao, Y., Lai, Q., Liu, D., Xu, Z., and Ji, X., Mater. Chem. Phys., 2005, vol. 94, pp. 382–387. https://doi.org/10.1016/j.matchemphys.2005.05.019
Babenko, A.S., Kornev, P.V., Zhukov, A.V., and Chizhevskaya, S.V., Uspekhi Khimii Khim. Tekhnol., 2015, vol. XXIX, no. 6, pp. 88–89.
Jansen, A.N., Kahaian, A.J., Kepler, K.D., Nelson, P.A., Amine, K., Dees, D.W., Vissers, D.R., and Thackeray, M.M., J. Power Sourc., 1999, vol. 81–82, pp. 902–905. https://doi.org/10.1016/S0378-7753(99)00268-2
Masatoshi, M., Satoshi, U., Eriko, Y., Keiji, K., and Shinji, I., J. Power Sourc., 2001, vol. 101, pp. 53–59. https://doi.org/10.1016/S0378-7753(01)00554-7
Zhao, B., Ran, R., Liu, M., and Shao, Z., Mater. Sci. Eng. R, 2015, vol. 98, pp. 1–71. https://doi.org/10.1016/j.mser.2015.10.001
Gu Y.-J., Guo, Z., and Liu H.-Q., Electrochim. Acta, 2014, vol. 123, pp. 576–581. https://doi.org/10.1016/j.electacta.2013.12.159
Peramunage, D.and Abraham, K.M., J. Electrochem. Soc., 1998, vol. 145, pp. 2609–2615. https://doi.org/10.1149/1.1838689
Hsiao, K.C., Liao, S.C., and Chen, J.M., Electrochim. Acta, 2008, vol. 53, pp. 7242–7247. https://doi.org/10.1016/j.electacta.2008.05.002
Shen, Y., Søndergaard, M., Christensen, M., Birgisson, S., and Iversen, B.B., Chem. Mater., 2014, vol. 26, pp. 3679–3686. https://doi.org/10.1021/cm500934z
Shu, J., Shui, M., Xu, D., Gao, S., Yi, T., Wang, D., Li, X., and Ren, Y., Ionics, 2011, vol. 17, pp. 503–509. https://doi.org/10.1007/s11581-011-0544-4
Shin J.-W., Hong C.-H., and Yoon D.-H., J. Am. Ceram. Soc., 2012, vol. 95, pp. 1894–1900. https://doi.org/10.1111/j.1551-2916.2012.05165.x
Dorrian, J.F. and Newnham, R.E., Mater. Res. Bull., 1969, vol. 4, pp. 179–183. https://doi.org/10.1016/0025-5408(69)90054-3
Deschanvers, A., Raveau, B., and Sekkal, Z., Mater. Res. Bull., 1971, vol. 6, pp. 699–704.
ACKNOWLEDGMENTS
The characterization of materials was carried out at the Shared Access Centre “Composition of Compounds” of the Institute of High Temperature Electrochemistry of the Ural Branch of the RAS.
Funding
The work was carried out within the framework of the budget plan of the Institute of High-Temperature Electrochemistry of the Ural Branch of the Russian Academy of Sciences on the topic AAAA-A19-119020190042-7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest requiring disclosure in this article.
Additional information
Translated from Zhurnal Prikladnoi Khimii, No. 1, pp. 34–41, January, 2021 https://doi.org/10.31857/S0044461821010059
Rights and permissions
About this article
Cite this article
Pershina, S.V., Antonov, B.D. & Farlenkov, A.S. Optimization of Technology for Synthesis of Li4Ti5O12 Anode Materials for Lithium-Ion Batteries. Russ J Appl Chem 94, 30–37 (2021). https://doi.org/10.1134/S1070427221010055
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427221010055