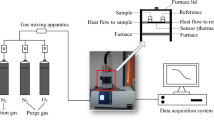
Abstract
Kinetic regularities of the mass loss and heat and-gas release were studied in the thermal decomposition of a solid propellant composed of aluminum, ammonium perchlorate, and a polymer binder. It was shown that, under heating from 40 to 340°C under permanent vacuum conditions, propellant samples decompose without ignition, with the limiting mass loss in the decomposition being 48%. When experiments were performed in air, the propellant formulation decomposes with sharp ignition, with the inflammation temperature (270–287°C) and amount of volatiles released by this instant of time (10–16 wt %) dependent on the heating rate. The kinetic regularities of the mass loss in the decomposition of a solid propellant were described in terms of the polychromatic kinetics model that assumes that the reaction system has ensembles of particles differing in reactivity. The distribution functions of the mass fractions of the propellant by activation energies of decomposition were calculated. The heat release kinetics in the decomposition of a propellant formulation in the temperature range 153–270°C in a closed evacuated system is described by a sum of equations for two parallel reactions: 1st-order reaction with a heat effect Q1 = 200 ± 5 kJ kg–1 and 1st-order autocatalysis with heat effect Q2 = 1900 ± 50 kJ kg–1. The rate constants and the activation parameters of the process were determined.
Similar content being viewed by others
References
Obnosov, B.V., Sorokin, V.A., Yanovskii, L.S., Yagodnikov, D.A., Frantskevich, V.P., Zhivotov, N.P., Surikov, E.V., Kobko, G.G., Tikhomirov, M.A., and Sharov, M.S., Konstruktsiya i proektirovanie kombinirovannykh raketnykh dvigatelei na tverdom toplive (Design of Combined Solid-Fuel Rocket Engines), Sorokin, V.A., Ed., Moscow: Mosk. Gos. Tekhn. Univ. im. N.E. Bau mana, 2014, p. 223.
Hedman, T.D., Propulsion Power Res., 2016, vol. 5, no. 2, pp. 87–96. https://doi.org/10.1016/j.jppr.2016.04.002
Cerri, S., Bohn, M.A., Menke, K., and Galfetti, L., Central Eur. J. Energetic Mater., 2009, vol. 6, no. 2, pp. 149–165.
Bihari, B.K., Rao, N.P., Gupta, M., and Murthy, K.P.S., Proc. 11th Int. High Energy Materials Conf. & Exhibits, HEMCE-2017, Pune, India, 23–25 November 2017, pp. 386–389.
Prasuła, P. and Czerwińska, M., Proc. 20th Seminar on New Trends in Research of Energetic Materials, Pardubice, 18–20 April, 2017, pp. 916–926.
Musanic, S. and Suceska, M., Central Eur. J. Energetic Mater., 2013, vol. 10, no. 2, pp. 225–244.
Biggs, G.L., 45th AIAA/ASME/SAE/ASEE Joint Propulsion Conf., Denver, Colorado, 2–5 August 2009, pp. 2009–5423.
Goikhman, B.D. and Smekhunova, T.P., Russ. Chem. Rev. 1980, vol. 49, no. 8, pp. 748–758.
Gonçalves, R.F.B., Silva, R.P., Rocco, J.A., and Iha, K., 44th AIAA/ASME/SAE/ASEE Joint Propulsion Conf., Hartford, CT, 21–23 July 2008, pp. 4959–4966.
Sell, T., Vyazovkin, S., and Wight, C.A., Combust. Flame, 1999, no. 119, pp. 174–181. https://doi.org/10.1016/S0010-2180(99)00036-X
Almeida, L.E.N., Martins, A.F., Gomes, S.R., and Cunha, F.A., 49th AIAA/ASME/SAE/ASEE Joint Propulsion Conf., San Jose, CA, 14–17 July, 2013, pp. 4088–5006. https://doi.org/10.2514/6.2013-4088
Mezroua, A., Phd. Ecole Militaire Polytechnique, Algiers, Algeria, 2015.
Smirnov, L.P., Russ. Chem. Rev., 2004, vol. 73, no. 11, pp. 1121–1141.
Sandakov, G.I., Smirnov, L.P., Sosikov, A.I., Summanen, K.T., and Volkova, N.N., J. Polym. Sci., Part B: Polym. Phys., 1994, vol. 32, pp. 1585–1592. https://doi.org/10.1002/polb.1994.090320901
Volkova, N.N., Tarasov, V.P., and Erofeev, L.N., Polym. Sci., Ser. A, 2008, vol. 50, no. 6, pp. 698–703.
Manelis, G.B., Nazin, G.M., Rubtsov, Yu.I., and Strunin, V.A., Thermal Decomposition and Combustion of Explosives and Propellants, London: Taylor & Francis Group, 2003.
Eslami, A., Hosseini, G., and Bazrgary, M., J. Thermal Anal. Calorim., 2013, vol. 113, pp. 721–730.
Hori, K. and Iwama, A., Propellants, Explos., Pyrotech., 1990, vol. 15, pp. 99–102. https://doi.org/10.1002/prep.19900150307
D’yakov, A.P., Nikolaeva, L.I., Strunin, V.A., and Manelis, G.B., Khim. Fiz., 2003, vol. 22, no. 5, pp. 70–73.
Koptelov, A.A. and Karyazov, S.V., Dokl. Phys. Chem., 2003, vol. 389, no. 46, pp. 101–105.
Kim, E.S. and Yang, V., Combust. Flame, 2002, vol. 131, pp. 227–236. https://doi.org/10.1016/S0010-2180(02)00411-X
Garland, N.L. and Nelson, H.H., Chem. Phys. Lett., 1992, vol. 191, nos. 3-4, pp. 269–272. https://doi.org/10.1016/0009-2614(92)85299-P
Pivkina, A.N., Murav’yov, N.V., Monogarov, K.A., Meerov, D.B., Fomenkov, I.V., Skryleva, E.A., Presnyakov, M.Yu., Vasil’ev, A.L., Shishov, N.I., and Milyokhin, Yu.M., Combust., Explos. Shock Waves, 2018, vol. 54, no. 4, pp. 450–460. https://doi.org/10.15372/FGV20180409.
Galyuk, O.S., Rubtsov, Yu.I., Malinovskaya, G.F., and Manelis, G.B., Zh. Fiz. Khim., 1965, vol. 39, pp. 2319–2322.
Mikhailov, A.I., Lebedev, Ya.S., and Buben, N.Ya., Kinet. Kataliz, 1964, vol. 5, no. 6, pp. 1020–1027.
Lebedev, Ya.S., Usp. Khim., 1968, vol. 37, pp. 934–946.
Lebedev, Ya.S., Kinet. Kataliz, 1978, vol. 19, no. 6, pp. 1367–1377.
Emanuel, N.M. and Buchachenko, A.L., Chemical Physics of Polymer Degradation and Stabilization, Utrecht, The Netherlands: VNU Science Press, 1987.
Dubovitskii, V.A. and Irzhak, V.I., Polym. Sci., Ser. B, 2005, vol. 47, nos. p1-2, pp. 22–41.
Lawson, C.L. and Hanson, R.J., Solving Least Squares Problems, Revised republication, Phyladelphia: SIAM (Society for Industrial and Applied Mathematics), 1995.
Vyazovkin, S., Burnham, A.K., Criado, J.M., Pérez-Maqueda, L.A., Popescu, C., and Sbirrazzuoli, N., Thermochim. Acta., 2011, vol. 520, pp. 1–19. https://doi.org/10.1016/j.tca.2011.03.034
Manelis, G.B. and Rubtsov, Yu.I., Zh. Fiz. Khim., 1966, vol. 40, no. 4, pp. 770–774.
Funding
The study was financed by the Institute of Problems of Chemical Physics, Russian Academy of Sciences, topic “Development of high-energy materials and technologies for promising systems being developed,” State registration no. 008920140019 and financially supported by Program no. 56 of the Presidium of the Russian Academy of Sciences “Fundamentals of breakthrough technologies in the interests of national security.”
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest
The authors state that they have no conflict of interest to be disclosed in the present communication.
Russian Text © The Author(s), 2019, published in Zhurnal Prikladnoi Khimii, 2019, Vol. 92, No. 11, pp. 1463−1475.
Rights and permissions
About this article
Cite this article
Volkova, N.N., Dubovitskii, V.A., Zholudev, A.F. et al. Kinetics of Thermal Decomposition of Solid Propellant Based on Aluminum and Ammonium Perchlorate. Russ J Appl Chem 92, 1558–1569 (2019). https://doi.org/10.1134/S1070427219110132
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070427219110132