Abstract
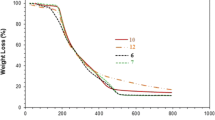
Specific features of sulfur polycondensation with naphthalene in the presence of AlCl3 and the influence exerted by synthesis conditions on the structure and properties of aromatic polysulfides being formed were studied. It was shown that the soluble polynaphthylene sulfides markedly surpass linear poly(1,4-phenylene sulfide) in thermal-oxidative stability. It was found that the predominance of the dehydropolycondensation of naphthalene to give a polynaphthylene structure cross-linked by sulfur over the polysulfurization of naphthalene and incorporation of sulfur into a linear chain. A high stability was demonstrated of polynaphthylene structures formed via dehydropolycondensation of naphthalene under the action of AlCl3, followed by sulfurization of polynaphthylene with elementary sulfur into lateral chains.
Similar content being viewed by others
References
Nedel’kin, V.I., Poluchenie i svoistva organicheskikh soedinenii sery (Synthesis and Properties of Organic Compounds of Sulfur), Belen’kii, L.I., Ed., Moscow.: Khimiya, 1998. pp. 505–546.
Nedel’kin, V.I., Zachernyuk, B.A., and Andrianova, O.B., Zh. Ross. Khim. O–va im. D.I. Mendeleeva, 2005, vol. 49, no. 6, pp. 3–10.
Novyi spravochnik khimika i tekhnologa. Osnovnye svoistva neorganicheskikh, organicheskikh i elemento-organicheskikh soedinenii (New Reference Book of Chemists and Technologists. Basic Properties of Inorganic, Organic, and Organoelement Compounds), St. Petersburg: Mir Sem’ya, 2002.
Stacy, C.J., J. Appl. Polym. Sci., 1986, vol. 32, no. 3, pp. 3959–3969.
Koch, W. and Heitz, W., Makromol. Chem., 1983, vol. 184, no. 4, pp. 779–792.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © L.A. Korneeva, V.I. Nedel’kin, B.A. Zachernyuk, N.V. Kuleshov, 2015, published in Zhurnal Prikladnoi Khimii, 2015, Vol. 88, No. 12, pp. 1711–1715.
Rights and permissions
About this article
Cite this article
Korneeva, L.A., Nedel’kin, V.I., Zachernyuk, B.A. et al. Synthesis of aromatic polysulfides on the basis of sulfur and naphthalene. Russ J Appl Chem 88, 1958–1962 (2015). https://doi.org/10.1134/S10704272150120095
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S10704272150120095