Abstract
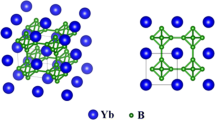
Nanocrystalline ytterbium zirconate Yb4Zr3O12 (δ-phase) was synthesized by the solid-state method with preliminary mechanical activation of a stoichiometric mixture of the corresponding oxides. The processes occurring in the course of the synthesis were studied by X-ray powder diffraction, IR spectroscopy, and thermal analysis. The average size of Yb4Zr3O12 crystallites obtained by annealing a mechanically activated oxide mixture at 900, 1000, 1100, and 1200°C was calculated by the Scherrer formula and was 12, 17, 27, and 41 nm, respectively.




Similar content being viewed by others
REFERENCES
Zhao, Z.-T., Guo, R.-F., Mao, H.-R., and Shen, P., J. Eur. Ceram. Soc., 2021, vol. 41, p. 5768. https://doi.org/10.1016/j.jeurceramsoc.2021.04.053
Lyskov, N.V., Shchegolikhin, A.N., Stolbov, D.N., Kolbanev, I.V., Gomes, E., Abrantes, J.C.C., and Shlyakhtina, A.V., Electrochim. Acta, 2022, vol. 403, article no. 139632. https://doi.org/10.1016/j.electacta.2021.139632
Wang L., Li J., Xie H., Chen Q., Xie Y., Chen Q., Xie Y., Prog. Nucl. Energy, 2021, vol. 137, article ID 103774. https://doi.org/10.1016/j.pnucene.2021.103774
Zhong, F., Shi, L., Zhao, J., Cai, G., Zheng, Y., Xiao, Y., and Long, J., Ceram. Int., 2017, vol. 43, no. 15, p. 11799. https://doi.org/10.1016/j.ceramint.2017.06.019
Zinatloo-Ajabshir, S., Salavati-Niasari, M., Sobhari, A., and Zinatloo-Ajabshir, Z., J. Alloys Compd., 2018, vol. 767, p. 1164. https://doi.org/10.1016/j.jallcom.2018.07.198
Kumar, A., Singh, D.K., and Manam, J., J. Mater. Sci.: Mater. Electron., 2019, vol. 30, p. 2360. https://doi.org/10.1007/s10854-018-0509-8
Gonzalez, M., Moure, C., Jurado, J.R., and Duran, P., J. Mater. Sci., 1993, vol. 28, p. 3451. https://doi.org/10.1007/BF01159821
Kornienko, O.A., Andrievskaya, E.R., Bykov, A.I., and Bogatyreva, Zh.D., Vіsn. Odes. Nats. Unіv.: Khіm., 2018, vol. 23, no. 1 (65), p. 83. https://doi.org/10.18524/2304-0947.2018.1(65).124549
Fabrichnaya, O., Lakiza, S.M., Kriegel, M.J., Seidel, J., Savinykh, G., and Schreiber, G., J. Eur. Ceram. Soc., 2015, vol. 35, no. 10, p. 2855. https://doi.org/10.1016/j.jeurceramsoc.2015.03.037
Fabrichnaya, O. and Seifert, H.J., CALPHAD: Comput. Coupling Phase Diagrams Thermochem., 2010, vol. 34, no. 2, p. 206. https://doi.org/10.1016/j.calphad.2010.03.001
Stolyarova, V.L., Lopatin, S.I., Fabrichnaya, O.B., and Shugurov, S.M., Rapid Commun. Mass Spectrom., 2014, vol. 28, no. 1, p. 109. https://doi.org/10.1002/rcm.6764
Simoncic, P. and Navrotsky, A., J. Am. Ceram. Soc., 2007, vol. 90, no. 7, p. 2143. https://doi.org/10.1111/j.1551-2916.2007.01678.x
Thornber, M.R. and Bevan, D.J.M., J. Solid State Chem., 1970, vol. 1, nos. 1–4, p. 536. https://doi.org/10.1016/0022-4596(70)90139-8
Stecura, S., Thin Solid Films, 1987, vol. 150, no. 1, p. 15. https://doi.org/10.1016/0040-6090(87)90305-1
Chou, Y.H., Hondow, N., Thomas, C.I., Mitchell, R., Brydson, R., and Douthwaite, R.E., Dalton Trans., 2012, vol. 41, no. 8, p. 2472. https://doi.org/10.1039/C2DT12269C
Karaulov, A.G. and Zoz, E.I., Refract. Ind. Ceram., 1999, vol. 40, p. 479. https://doi.org/10.1007/BF02762623
Gonzalez, M., Moure, C., Jurado, J.R., and Duran, P., J. Mater. Sci., 1993, vol. 28, p. 3451. https://doi.org/10.1007/BF0115
Rossell, H.J., J. Solid State Chem., 1976, vol. 19, no. 2, p. 103. https://doi.org/10.1016/0022-4596(76)90156-0
Rejith, R., Krishnan, R.R., John, A., Thomas, J.K., and Solomon, S., Ionics, 2019, vol. 25, p. 5091. https://doi.org/10.1007/s11581-019-03097-z
Kong, S.L., Karatchevtseva, I., Gregg, D.J., Blackford, M.G., Holmes, R., and Triani, G., J. Eur. Ceram. Soc., 2013, vol. 33, p. 3273. https://doi.org/10.1016/j.jeurceramsoc.2013.05.011
Kaliyaperumal, C., Sankarakumar, A., and Paramasivam, T., J. Alloys Compd., 2020, vol. 813, article ID 152221. https://doi.org/10.1016/j.jallcom.2019.152221
Heinicke, G., Tribochemistry, Berlin: Akademie, 1984.
Fundamental’nye osnovy mekhanicheskoi aktivatsii, mekhanosinteza i mekhanokhimicheskikh tekhnologii (Fundamentals of Mechanical Activation, Mechanosynthesis, and Mechanochemical Technologies), Avvakumov, E.G., Ed., Novosibirsk: Sib. Otd. Ross. Akad. Nauk, 2009.
Lapshin, O.V., Boldyreva, E.V., and Boldyrev, V.V., Russ. J. Inorg. Chem., 2021, vol. 66, p. 433. https://doi.org/10.1134/S0036023621030116
Salazar-Zertuche, M., Diaz-Guillen, J.A., AcostaGarcía, J.O., Diaz-Guillen, J.C., Montemayor, S.M., Burciaga-Diaz, O., Bazaldua-Medellin, M.E., and Fuentes, A.F., Int. J. Hydrogen Energy, 2019, vol. 44, no. 9, p. 12500. https://doi.org/10.1016/j.ijhydene.2018.11.141
Kaminskii, Yu.D., Mekhanokhimicheskie reaktory planetarnogo tipa. Teoriya i praktika (Planetary Mechanochemical Reactors. Theory and Practice), Novosibirsk: Nauka, 2015.
Kalinkin, A.M., Usoltsev, A.V., Kalinkina, E.V., Nevedomskii, V.N., and Zalkind, O.A., Russ. J. Gen. Chem., 2017, vol. 87, no. 10, p. 2258. https://doi.org/10.1134/S1070363217100024
Kalinkin, A.M., Vinogradov, V.Yu., and Kalinkina, E.V., Inorg. Mater., 2021, vol. 57, no. 2, p. 178. https://doi.org/10.1134/S0020168521020072
Dorofeev, G.A., Streletskii, A.N., Povstugar, I.V., Protasov, A.V., and Elsukov, E.P., Colloid J., 2012, vol. 74, no. 6, p. 678. https://doi.org/10.1134/S1061933X12060051
Avvakumov, E.G., Mekhanicheskie metody aktivatsii khimicheskikh protsessov (Mechanical Methods of Activation of Chemical Processes), Novosibirsk: Nauka, 1986.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
A.M. Kalinkin is a member of the Editorial Board of the Zhurnal Obshchei Khimii. The other authors declare the absence of conflict of interest.
Supplementary information
Rights and permissions
About this article
Cite this article
Kalinkin, A.M., Kuz’menkov, O.A., Kalinkina, E.V. et al. Mechanically Activated Solid-State Synthesis of Nanocrystalline Yb4Zr3O12. Russ J Gen Chem 92, 1056–1061 (2022). https://doi.org/10.1134/S1070363222060172
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363222060172