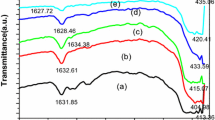
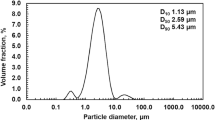
Abstract
Pure crystalline compounds MIIU3O10 · n H2O (MII = Ca, Sr) and BaU2O7 · 3 H2O were obtained by reactions of synthetic schoepite (UO3 · 2.25 H2O) with aqueous solutions of calcium, strontium, and barium nitrates under hydrothermal conditions at 200°C and pH 7‒8. Their structure, dehydration, and thermal decomposition were studied by chemical analysis, X-ray fluorescence spectrometry, X-ray powder diffraction, IR spectroscopy, and thermogravimetric analysis.
Similar content being viewed by others
References
Chernorukov, N.G., Nipruk, O.V., Knyazev, A.V., Arova, M.I., and Chaplieva, K.A., Russ. J. Inorg. Chem., 2013, vol. 58, no. 1, p. 9. doi https://doi.org/10.1134/S0036023613010051
Chernorukov, N.G., Nipruk, O.V., Chernorukov, G.N., and Sedelkina, O.S., Radiochemistry, 2015, vol. 57, no. 4, p. 378. doi https://doi.org/10.1134/S1066362215040062
Finch, R.J., Burns, P.C., Hawthorne, F.C., and Ewing, R.C., Can. Mineral., 2006, vol. 44, p. 1197.
Cejka, J., Sejkora, J., Skala, R., Cejka, J., Novotna, M., and Ederova, J., Neues Jahrb. Mineral., Abh., 1998, vol. 174, p. 159.
Pagoaga, M.K., Appleman, D.E., and Stewart, J.M., Mineral. Mag., 1986, vol. 50, p. 125.
Pagoaga, M.K., Appleman, D.E., and Stewart, J.M., Am. Mineral., 1987, vol. 72, p. 1230.
Sobry, R., J. Inorg. Nucl. Chem., 1973, vol. 35, no. 5, p. 1516. doi https://doi.org/10.1016/0022-1902(73)80240-4
Sobry, R., J. Inorg. Nucl. Chem., 1973, vol. 35, no. 8, p. 2754. doi https://doi.org/10.1016/0022-1902(73)80506-8
Dunn, H., X-Ray Diffraction Data for Some Uranium Compounds, ORNL-2092, Oak Ridge National Laboratory, Oak Ridge, Tennessee, 1956.
Peters, J., Mem. Soc. Roy. Sci. Liege, 1967, p. 5.
Volod’ko, M.V., Komyak, A.I., and Umreiko, D.S., Uranilovye soedineniya (Uranyl Compounds). Minsk: Belarus. Gos. Univ., 1981, vol. 1.
Yukhnevich, G.V., Infrakrasnaya spektroskopiya vody (Infrared Spectroscopy of Water), Moscow: Nauka, 1973.
Cordfunke, E.H.P. and Loopstra, B.O., J. Inorg. Nucl. Chem., 1967, vol. 29, p. 51. doi https://doi.org/10.1016/0022-1902(67)80143-X
Sali, S.K., Sampath, S., and Venugopal, V., J. Nucl. Mater., 2000, vol. 277, p. 106. doi https://doi.org/10.1016/S0022-3115(99)00145-2
Allen, G.C. and Griffiths, A.J., J. Chem. Soc., Dalton Trans., 1979, p. 315. doi https://doi.org/10.1039/dt9790000315
Loopstra, B.O. and Rietveld, H.M., Acta Crystallogr., Sect. B, 1969, vol. 25, p. 787. doi https://doi.org/10.1107/S0567740869002974
Nipruk, O.V., Knyazev, A.V., Chernorukov, G.N., and Pykhova, Yu.P., Radiochemistry, 2011, vol. 53, no. 2, p. 146. doi https://doi.org/10.1134/S1066362211020044
Author information
Authors and Affiliations
Corresponding author
Additional information
Russian Text © N.G. Chernorukov, O.V. Nipruk, G.N. Chernorukov, R.V. Abrazheev, K.A. Chaplieva, 2019, published in Zhurnal Obshchei Khimii, 2019, Vol. 89, No. 1, pp. 83–88.
Rights and permissions
About this article
Cite this article
Chernorukov, N.G., Nipruk, O.V., Chernorukov, G.N. et al. Synthesis and Properties of Calcium, Strontium, and Barium Uranates. Russ J Gen Chem 89, 71–75 (2019). https://doi.org/10.1134/S1070363219010134
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363219010134