Abstract
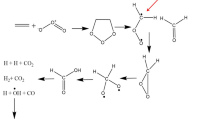
The role of hydrogen bonding in the autocatalytic reaction of acetic anhydride with tert-butyl hydroperoxide in CC14 has been elucidated. The stage-by-stage mechanisms of formation of hydrogen-bonded complexes. Structure and energy parameters of stable conformers of the compounds and energy of the major and transition states have been determined by means of B3LYP/aug-cc-pVDZ and B3LYP/aug-cc-pVTZ methods. The contribution of the O···HO hydrogen bonds in the stabilization of the complexes has been determined using the noncovalent interactions index. 1-Hydroxy-1-alkylperoxy compound is the most stable intermediate.
Similar content being viewed by others
References
Comprehensive Organic Synthesis II, Knochel, P. and Molander, G.A., Eds., Amsterdam: Elsevier Ltd., 2014.
Varfolomeeva, V.V., Russ. J. Gen. Chem., 2011, vol. 81, no. 9, p. 1812. doi 10.1134/S1070363211090131
Samuilov, A.Ya. and Samuilov, Ya.D., Butlerovsk. Soobshch., 2011, vol. 28, no. 19, p. 1.
Nosachevva, I.M., Revkov, O.A., and Perkel’, A.L., Vestn. KuzGTU, 2004. 40, no. 3, p. 78.
Ferreira, R., Garcia, H., Sousa, A.F., Guerreiro, M., Duarte, F.J.S., Freire, C.S.R., Calhorda, M.J., Silvestre, A.J.D., Kunz, W., Rebelo, L.P.N., and Pereira, C.S., RSC Adv., 2014, vol. 4, p. 2993. doi 10.1039/c3ra45910a
Desiraju, G.G.R. and Steiner, T., The Weak Hydrogen Bond in Structural Chemistry and Biology, Oxford: Oxford University Press, 1999.
Varfolomeeva, V.V., Candidate Sci. (Chem.) Dissertation, Samara: Samar. Gos. Univ., 2000.
Non-Covalent Interactions in the Synthesis and Design of New Compounds, Maharramov, A.M., Mahmudov, K.T., Kopylovich, M.N., and Pombeiro, A.J.L., Hoboken: John Wiley & Sons, 2016. doi 10.1002/9781119113874
Carey, F.A. and Sundberg, R.J., Advanced Organic Chemistry, New York: Springer, 2007.
Strazzolini, P., Giumanini, A.G., and Verardo, G., Tetrahedron, 1994, vol. 50, no. 1, p. 217. doi 10.1016/S0040-4020(01)80747-X
Varfolomeeva, V.V., Russ. J. Gen. Chem., 2007, vol. 77, no. 11, p. 1891. doi 10.1134/S1070363207110084
Antonovskii, V.L. and Khursan, S.L., Fizicheskaya khimiya organicheskikh peroksidov (Physical Chemistry of Organic Peroxides), Moscow: Akademkniga, 2003.
Antonovskii, V.L. and Terentev, V.A., Zh. Fiz. Khim., 1969, vol. 43, no. 11, p. 2727.
Uspekhi khimii organicheskikh perekisnykh soedinenii i autookisleniya (Advanced in Chemistry of Organic Peroxide Compounds and Autooxidation), Emmanujel’, N.M., Ed., Moscow: Khimiya, 1969, p. 442.
Organic Peroxides, Swern, D., Ed., New York: Wiley InterSci., 1972, vols. 1–3, p. 112.
The Chemistry of Peroxides, Patai, S., Ed., Chichester: John Wiley & Sons, 1983, p. 212.
Antonovskii, V.L. and Terentev, V.A., Zh. Fiz. Khim., Zh. Fiz. Khim., 1969, vol. 43, no. 10, p. 2549.
Terentev, V.A. and Varfolomeeva, V.V., Zh. Obshch. Khim., 1994, vol. 64, no. 1, p. 98.
Tables of Rate and Equilibrium Constants of Heterolytic Organic Reactions, Palm, V.A., Ed., Moscow: VINITI, 1977, vol. 3, ch. 2.
Antonovskii, V.L. and Terentev, V.A., Zh. Fiz. Khim., 1966, vol. 40, no. 12, p. 3078.
Antonovskii, V.L. and Khursan, S.L., Russ. Chem. Rev., 2003, vol. 72, no. 11, p. 939. doi 10.1070/RC2003v072n11ABEH000749
Smith, M.B. and March, J., March’s Advanced Organic Chemistry: Reactions, Mechanisms and Structure, New York: Wiley, 2007.
Antonovskii, V.L. and Terentev, V.A., Zh. Org. Chem., 1967, vol. 3, no. 6, p. 1011.
NIST Chemistry WebBook, SRD 69. https://doi.org/webbook.nist.gov.
Wu, G., van Alsenoy, C., Geise, H.J., Sluyts, E., van der Veken, B.J., Shishkov, I.F., and Khristenko, L.V., J. Phys. Chem. (A), 2000, vol. 104, no. 7, p. 1576. doi 10.1021/jp993131z
Ma, C.-P., Zeng, X.-Q., and Ge, M.-F., J. Mol. Struct., 2008, vol. 875, nos. 1–3, p. 143. doi 10.1016/j.molstruc.2007.04.020
Dashevskii, V.G., Konformatsionnyi analiz organicheskikh molekul (Conformational Analysis of Organic Molecules), Moscow: Khimiya, 1982.
Hydrogen Bonding in Organic Synthesis, Pihko, P.M., Ed., Wiley-VCH, 2009.
Marechal, Y., The Hydrogen Bond and the Water Molecule: The Physics and Chemistry of Water, Aqueous and Bio Media, Amsterdam: Elsevier, 2007.
Jeffrey, G.A. and Saenger, W., Hydrogen Bonding in Biological Structures, Berlin: Springer-Verlag, 1991. doi 10.1007/978-3-642-85135-3
Kyte, J., Structure in Protein Chemistry, New York: Garland Science, 2006.
Gilli, G. and Gilli, P., The Nature of the Hydrogen Bond: Outline of a Comprehensive Hydrogen Bond Theory, Oxford: Oxford University Press, 2009.
Granovsky, A.A., Firefly Version 8. www.classic.chem.msu.su/gran/firefly/index.html.
Schmidt, M.W., Baldridge, K.K., Boatz, J.A., Elbert, S.T., Gordon, M.S., Jensen, J.H., Koseki, S., Matsunaga, N., Nguyen, K.A., Su, S., Windus, T.L., Dupuis, M., and Montgomery, J.A., J. Comput. Chem., 1993, vol. 14, no. 11, p. 1347. doi 10.1002/jcc.540141112
Computational Chemistry Comparison and Benchmark Data Base, NIST Standard Reference Database Number 101 Release 18, 2016. https://doi.org/cccbdb.nist.gov.
Johnson, E.R., Keinan, S., Mori-Sanchez, P., Contreras-Garcia, J., Cohen, A.J., and Yang, W., J. Am. Chem. Soc., 2010, vol. 132, no. 18, p. 6498. doi 10.1021/ja100936w
Contreras-Garcia, J., Johnson, E.R., Keinan, S., Chaudret, R., Piquemal, J.-P., Beratan, D.N., and Yang, W., J. Chem. Theory Comput., 2011, vol. 7, no. 3, p. 625. doi 10.1021/ct100641a
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © V.V. Varfolomeeva, 2018, published in Zhurnal Obshchei Khimii, 2018, Vol. 88, No. 5, pp. 710–716.
Rights and permissions
About this article
Cite this article
Varfolomeeva, V.V. The Role of Hydrogen Bond in the Mechanism of Autocatalytic Reaction between Acetic Anhydride and tert-Butyl Hydroperoxide. Russ J Gen Chem 88, 855–861 (2018). https://doi.org/10.1134/S107036321805002X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107036321805002X