Abstract
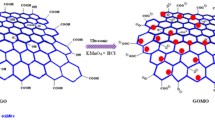
Graphene oxide (GO) has been prepared by the modified Hummers method using graphite as starting material. The product was studied by the X-ray diffraction (XRD), Raman spectroscopic, transmission electron microscopic (TEM), and scanning electron microscopic (SEM) analyses. Adsorption capacity of GO for heavy metal ions was studied for the example of the Ni2+ ions and the adsorption kinetics and adsorption isotherm were determined. It was shown that the adsorption equilibrium curves are adequately described by the Langmuir equation.
Similar content being viewed by others
References
Dubey, S.P., Gopal, K., and Bersillon, J.L., J. Environ. Biol., 2009, vol. 30, p. 327.
Wu, J. and Pisula, W., Chem. Rev., 2007, vol. 107, p. 718.
Zhu, Y., Murali, S., Cai, W., Li, X., Suk, J.W., Potts, J.R., and Ruoff, R.S., Adv. Mater., 2010, vol. 22, p. 3906.
Dreyer, D.R., Park, S., Bielawski, C.W., and Ruoff, R.S., Chem. Soc. Rev., 2010, vol. 39, p. 228.
Machida, M., Mochimaru, T., and Tatsumoto, H., Carbon, 2006, vol. 44, p. 2681.
Dural, M. U., Cavas, I., and Papageorgiou, S.K, Chem. Eng. J., 2011, vol. 168(1), p. 77.
Zhang, L., Song, X., and Liu, X., Chem. Eng. J., 2011, vol. 178, p. 26.
Hall, K.R, Eagleton, L.C., Acrivos, A., and Vermeulen, T., Ind. Eng. Chem. Fundamen., 1966, vol. 5(2), p. 212.
Zhao, L.X., Song, Sh.E, Du, N., and Hou, W.G., Acta Phys.-Chim. Sin., 2012, vol. 28(12), p. 2905.
Hu, X., Liu, Yu., Hui Wang, H., Chen, A., et al., Sep. Purif. Technol., 2013, vol. 108, p. 189.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Zheng, X., Hou, W., Lian, Q. et al. Preparation of graphene oxide and its application in Ni2+ removal. Russ J Gen Chem 86, 915–918 (2016). https://doi.org/10.1134/S1070363216040265
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363216040265