Abstract
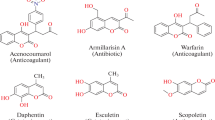
Two different series of novel analogs of 6,7-aryl/hetaryl coumarins 4a–4h and 8i–8l have been synthesized by using Suzuki–Miyara cross coupling reaction from 4-methyl-2-oxo-2H-chromen-7-yl trifluoro-methanesulfonate and 4-methyl-2-oxo-2H-chromen-6-yl trifluoromethanesulfonate in high yields (70–90%). All synthesized compounds were elucidated by means of IR, 1H NMR, 13C NMR, and MS spectra. The synthesized compounds were tested for antiproliferative activity against different human cancer cell lines (SiHa, MDAMB-231, and PANC-1) and some of products demonstrated the distinctive effect.
Similar content being viewed by others
References
Gibbs, J.B., Science, 2000, vol. 287, p. 1969.
Varmus, H., Science, 2006, vol. 312, p. 1162.
Ma, T., Liu, L., Xue, H., Li, L., Han, C., Wang, L., Chen, Z., and Liu, G., J. Med. Chem., 2008, vol. 51, p. 1432.
Kidane, A.G., Salacinski, H. A., Tiwari, K.R., and Bruckdorfer, A.M., Biomacromol., 2004, vol. 5, p. 798.
Appendino, G., Mercalli, E., Fuzzati, N., Arnoldi, L., Stavri, M., Gibbons, S., Ballero, M., and Maxia, A., J. Nat. Prod., 2004, vol. 67, p. 2108.
Kontogiorgis, C.A. and Hadjipavlou, L.D., Bioorg. Med. Chem. Lett., 2004, vol.14, p. 611.
Jung, K., Park, Y.J., and Ryu, J.S., Synth. Commun., 2008, vol. 38, p. 4395.
Kaczka, E.A., Wolf, F.J., Ratha, F.P., and Folkers, K.J., J. Am. Chem. Soc., 1955, vol. 77, p. 6404.
Mulwad, V.V. and Shirodkhar, J.M., Indian J. Heterocycl. Chem., 2002, vol.11, p. 192.
Rajeshwar Rao, V., Srimanth, K., and Vijaya Kumar, P., Indian J. Heterocycl. Chem., 2004, vol. 14, p. 141.
Nofal, Z.M., El-Zahar, M.I., and Abd El-Karim, S.S., Egypt. J. Chem., 2005, vol. 48 p. 587.
Sashidhara, K.V., Rosaiah, J.N., Kumar, A., Bhatia, G., and Khanna, A.K., Bioorg. Med. Chem. Lett., 2010, vol. 20, p. 3065.
Patil, A.D., Freyer, A.J., Eggleston, D.S., Haltiwanger, R.C., Bean, M.F., Taylor, P.B., Caranfa, M.J., Breen, A.L., Bartus, H.R., Johnson, R.K., Hertzberg, R.P., and Westley, J.W., J. Med. Chem., 1993, vol. 36, p. 4131.
Pierson, J.-T., Dume, A., Hutter, S., Delmas, F., Laget, M., Finet, J.-P., Azas, N., and Combes, S., Eur. J. Med. Chem., 2010, vol. 45 p. 864.
Pisani, L., Muncipinto, G., Miscioscia, T. F., Nicolotti, O., Leonetti, F., Catto, M., Caccia, C., Salvati, P., Soto-Otero, R., Mendez- Alvarez, E., Passeleu, C., and Carotti, A., J. Med. Chem., 2009, vol. 52, 6685.
Boland, G.M., Donnelly, D.M.X., Finet, J.-P., and Rea, M.D., J. Chem. Soc. Perkin Trans., 1996, vol. 1, p. 2591.
Donnelly, D.M.X., Finet, J.-P., Guiry, P.J., and Rea, M.D., Synth. Commun., 1999, vol. 29, p. 2719.
Ganina, O.G., Daras, E., Bourgarel-Rey, V., Peyrot, V., Andresyuk, A.N., Finet, J.-P., Fedorov, A.Yu., Beletskaya, I.P., Combes, Bioorg. Med. Chem., 2008, vol. 16, p. 8806.
Reddy M.A., Jain, N., Yada, D., Kishore, C., Vangala, J.R., Surendra, P.R., Addlagatta, A., Kalivendi, S.V., and Sreedhar, B., J. Med. Chem., 2011, vol. 54, p. 6751.
Author information
Authors and Affiliations
Corresponding author
Additional information
The text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Jayaprakash Rao, Y., Yadaiah goud, E., Hemasri, Y. et al. Synthesis and antiproliferative activity of 6,7-aryl/hetaryl coumarins. Russ J Gen Chem 86, 184–189 (2016). https://doi.org/10.1134/S1070363216010291
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070363216010291