Abstract—
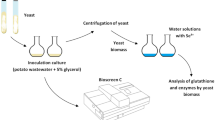
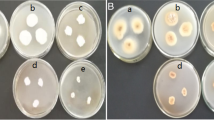
Investigation of the induction of antioxidant activity (AOA) by selenium compounds (Na2SeO3, diacetophenonyl selenide (DAPS-25), L-selenocystine) in various organisms is of interest as a way of protecting cell membranes from oxidative stress. A comparative analysis of the antioxidant activity of 23 amino acids was performed by coulometric titration with electrogenerated bromine and iodine. The activity decreases in the order: cystine > tryptophan > selenocystine > tyrosine > 3,3'-dimethyl-L-selenocystine > methionine. Only amino acids with sulfhydryl and selenol groups as more active reductants can interact with the electrogenerated iodine: cysteine > selenocysteine > threo-3-methyl-L-selenocysteine. Probably the correction of the antioxidant status at the amino acids level is based on the sulfhydryl and selenol groups in radicals. In case they are not enough, cystine, selenocystin, tryptophan, tyrosine, and methionine will act as scavenger-reductants. It was found that selenium compounds dose-dependently induce the total antioxidant activity of the Aspergillus niger mycelium and affect indicators of the antioxidant status (amino acid composition and catalase activity), which in turn stimulates the biomass accumulations. DAPS-25 and sodium selenite treatment at high doses (0.025 mg Se/L) caused the greatest effect on the total AOA induction (3.4–5.5 times). Lower concentrations (0.0025–0.00025 mg Se/L) had a lesser effect (25.8–41.7%). Activity in samples with L-selenocystin increased by 1.6–43.3%. It is noted that the iodine antioxidant activity in the mycelium was generally lower than the bromine one.








Similar content being viewed by others
REFERENCES
Jitca, G., Osz, B.E., Tero-Vescan, A., Miklos, A.P., Rusz, C.M., Batrinu, M.G., and Vari, C.E., Antioxidants, 2022, vol. 11, pp. 1–30.
Blinokhvatov, A.F., Denisova, G.V., and Il’in, D.Yu., Selen v biosfere (Selenium in Biosphere), Blinokhvatov, A.F., Ed., Penza: Penzensk. S.-kh. Akad., 2001.
Santesmasses, D., Mariotti, M., and Gladyshev, V.N., Antioxid. Redox Signal., 2020, vol. 33, pp. 525–536. https://doi.org/10.1089/ars.2020.8044
Zhang, Y., Roh, Y.J., Han, S.J., Park, I., Lee, H.M., Ok, Y.S., Lee, B.C., and Lee, S.R., Antioxidants (Basel), 2020, vol. 9, pp. 1–17. https://doi.org/10.3390/antiox9050383
Sies, H., Free Radical Biol. Med., 1993, vol. 14, pp. 313–323. https://doi.org/10.1016/0891-5849(93)90028-s
Blinokhvatov, A.F., 9-R-sim-nonahydro-10-oxa(chalcogene) anthracenes and salts of 9-R-sim-octahydro-10-oxonium (chalcogenonium) anthracene, Doctoral (Chem.) Dissertation, Saratov, 1993.
Drevko, B.I., RF Patent no. 2051681 S1, 1996.
Poluboyarinov, P.A. and Golubkina, N.A., Russ. J. Plant Physiol., 2015, vol. 62, pp. 367–374. https://doi.org/10.1134/S1021443715030164
Castillo-Godina, R.G., Foroughbakhch-Pournavab, R., and Benavides-Mendoza, A., J. Agr. Sci. Technol., 2016, vol. 18, pp. 233–244.
Bebien, M., Lagniel, G., Garin, J., Touati, D., Vermeglio, A., and Labarre, J., J. Bacteriol., 2002, vol. 184, pp. 1556–1564. https://doi.org/10.1128/jb.184.6.1556-1564.2002
Strogov, V.V. and Rodionova, T.N., Vestn. Vet., 2011, vol. 59, pp. 150–152.
Wang, H.W., Cai, D.B., Xiao, G.H., Zhao, C.L., Wang, Z.H., Xu, H.M., and Guan, Y.Q., Israeli J. Aquacult.-Bamidge, 2009, vol. 61, pp. 322–332.
Boryaev, G.I., Gavryushina, I.V., and Fedorov, Yu.N., S.-kh. Biol., 2010, vol. 45, pp. 65–70.
Dzobo, K. and Naik, Y.S., S. Afr. J. Sci., 2013, vol. 109, pp. 1–8. https://doi.org/10.1590/sajs.2013/965
Golubkina, N., Zamana, S., Seredin, T., Poluboyarinov, P., Sokolov, S., Baranova, H., Krivenkov, L., Pietrantonio, L., and Caruso, G., Plants (Basel), 2019, vol. 8, p. 102. https://doi.org/10.3390/plants8040102
Misra, S., Kwong, R.W.M., and Niyogi, S., J. Exp. Biol., 2012, vol. 215, pp. 1491–1501. https://doi.org/10.1242/jeb.062307
Poluboyarinov, P.A., Elistratov, D.G., and Shvets, V.I., Tonk. Khim. Tekhnol., 2019, vol. 14, pp. 5–24.
Casalbore, G., Mastragostino, M., and Valcher, S., J. Electroanal. Chem., 1978, vol. 87, pp. 411–418.
Abullin, I.F. and Budnikov, G.K., Zavod. Lab., 1998, vol. 64, pp. 1–12.
Sorensen, A., Gotfredsen, H., and Pittelkow, M., Chem. Commun., 2014, vol. 50, pp. 3716–3718. https://doi.org/10.1039/C4CC00523F
Sulfur in Organic and Inorganic Chemistry, Senning, A., Ed., New York: Marcel Dekker, 1972.
Pleasants, J.C., Guo, W., and Rabenstein, D.L., J. Am. Chem. Soc., 1989, vol. 111, pp. 6553–6558.
Hondal, R.J., and Ruggles, E.L., Amino Acids, 2011, vol. 41, pp. 73–89. https://doi.org/10.1007/s00726-010-0494-6
Blinokhvatov, A.F., Denisova, G.V., Ivanov, A.I., and Il’in, D.Yu., Mikol. Fitopatol., 2000, vol. 34, pp. 42–45.
Knyazeva, O.E. and Poluboyarinov, P.A., in Sovremennye problemy teoreticheskoi i eksperimental’noi khimii. Mezhvuzovskii sbornik nauchnykh trudov XIV Vserossiiskoi konferentsii molodykh uchenykh s mezhdunarodnym uchastiem (Modern Problems of Theoretical and Experimental Chemistry: Interuniversity Collection of Scientific Papers of XIV All-Russia Conference of Young Scientists with International Participation), Saratov, 2020, pp. 64–67.
Kieliszek, M., Blazejak, S., Bzducha-Wrobel, A., and Kot, A.M., Biol. Trace Elem. Res., 1989, vol. 187, pp. 316–327.
Szabados, L. and Savoure, A., Trends Plant Sci., 2010, vol. 15, pp. 89–97.
Yuan, L., Zhang, R., Ma, X., Yang, L., Zheng, Q., Chen, D., Li, M., Fan, T., Liu, Y., Pan, L., and Yin, X., Nutrients, 2018, vol. 10, p. 318. https://doi.org/10.3390/nu10030318
Chikin Yu.A., Likhachev A.N., Mikol. Fitopatol., 1997, vol. 31, no. 4, pp. 54-61.
Poluboyarinov, P.A., Moiseeva, I.Ya., Mikulyak, N.I., Golubkina, N.A., and Kaplun, A.P., Izv. Vyssh. Uchebn. Zaved., Ser. Khim. Khim. Tekhnol., 2022, vol. 65, pp. 19–29. https://doi.org/10.6060/ivkkt.20226502.6466
Poluboyarinov, P.A., Golubkina, N.A., Aniskov, A.A., Moiseeva, I.J., Glebova, N.N., and Shvets, V.I., Russ. J. Bioorg. Chem., 2019, vol. 45, pp. 241–247.
Lapin A.A., MVI-001-44538054-07. Summarnaya antioksidantnaya aktivnost’. Metodika vypolneniya izmerenii na kulonometricheskom analizatore (MVI-001-44538054-07. Total Antioxidant Activity. Methods of Measurement on a Coulometric Analyzer). Zherdevka: OOO Kontsern Otechestvennye innovatsionnye tekhnologii, 2011.
Mineev, V.G., Sychev, V.G., Amel’yanchik, O.A., Bolysheva, T.N., Gomonova, N.F., Durynina, E.P., Egorov, B.C., Egorova, E.V., Edemskaya, N.L., Karpova, E.A., and Prizhukova, V.G., Praktikum po agrokhimii (A Practical Course in Agrochemistry), Moscow: Mosk. Gos. Univ., 2001.
M-04-38-2009. Metodika opredeleniya proteinogennykh aminokislot v kormakh i syr’e (M-04-38-2009. Methodology for Determination of Proteinogenic Amino Acids in Feeds and Raw Materials), St. Petersburg: OOO Lyumeks-marketing, 2014.
Pankratov, A.N., Tsivileva, O.M., Beloborodaya, A.S., Tsymbal, O.A., and Drevko, Ya.B., Izv. Sarat. Univ. Nov. Ser., Ser.: Khim. Biol. Ekol., 2017, vol. 17, no. 3, pp. 286–298.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflict of interest. This article does not contain any studies involving animals or human participants performed by any of the authors.
Additional information
Abbreviations: GPX, glutathione peroxidase, DAPS-25, diacetophenonylselenide.
Corresponding author: phone: +7 (950) 230-48-76
Rights and permissions
About this article
Cite this article
Poluboyarinov, P.A., Kuznetsova, A.V., Moiseeva, I.Y. et al. Induction of Antioxidant Activity by Selenium Compounds in the Aspergillus niger Mycelium. Russ J Bioorg Chem 49, 823–835 (2023). https://doi.org/10.1134/S1068162023040155
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162023040155