Abstract
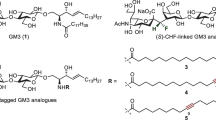
Glycolipids are components of the cell membrane capable of transport both from it and in the opposite direction, i.e. from the intercellular matrix to the membrane. The latter opens up the possibility of studying the functioning of glycolipids by their inserting into the cell membrane. In practice, synthetic analogs of glycolipids are significantly more suitable than natural glycolipids for such application, because one can vary properties, modifying their structure, as well as conjugating to other bioactive components, in addition to glycans. This study describes the synthesis of the eight glycolipids containing the same glycan part (A (type 2) tetrasaccharide) but varying in the composition of the lipid part as well as analogs of glycolipids carrying several identical carbohydrate moieties. The resulting series of synthetic analogs of glycolipids opens up the possibility of studying the immunological presentation of the glycan and its recognition by antibodies in the real microenvironment (glycocalyx) of a living cell.








Similar content being viewed by others
REFERENCES
Reitsma, S., Slaaf, D.W., Vink, H., Van Zandvoort, M.A.M.J., and Oude, EgbrinkM.G.A., Pflugers Arch. Eur. J. Physiol., 2007, vol. 454, pp. 345–359. https://doi.org/10.1007/s00424-007-0212-8
Rapoport, E.M., Khasbiullina, N.R., Komarova, V.A., Ryzhov, I.M., Gorbatch, M.M., Tuzikov, A.B., Khaidukov, S.V., Popova, I.S., Korchagina, E.Y., Henry, S.M., and Bovin, N.V., Biochim. Biophys. Acta Biomembr., 2021, vol. 1863, p. 183645.
Marcus, D.M. and Cass, L.E., Science, 1969, vol. 164, pp. 553–555. https://doi.org/10.1126/science.164.3879.553
Korchagina, E., Tuzikov, A., Formanovsky, A., Popova, I., Henry, S., and Bovin, N., Carbohydr. Res., 2012, vol. 356, pp. 238–246. https://doi.org/10.1016/j.carres.2012.03.044
Miller-Podraza, H., Chem. Rev., 2000, vol. 100, pp. 4663–4681. https://doi.org/10.1021/cr990347o
Koscielak, J., Transfus. Med., 2001, vol. 11, pp. 267–279. https://doi.org/10.1046/j.1365-3148.2001.00317.x
Karlsson, H., Johansson, L., Miller-Podraza, H., and Karlsson, K.A., Glycobiology, 1999, vol. 9, pp. 765–778. https://doi.org/10.1093/glycob/9.8.765
Morales-Serna, J.A., Boutureira, O., Diaz, Y., Matheu, M.I., and Castillon, S., Carbohydr. Res., 2007, vol. 342, pp. 1595–1612. https://doi.org/10.1016/j.carres.2007.03.028
Korchagina, E.Y. and Henry, S.M., Biochemistry, 2015, vol. 80, pp. 857–871. https://doi.org/10.1134/S0006297915070068
Frame, T., Carroll, T., Korchagina, E., Bovin, N., and Henry, S., Transfusion, 2007, vol. 47, pp. 876–882. https://doi.org/10.1111/j.1537-2995.2007.01204.x
Henry, S., ISBT Sci. Ser., 2020, vol. 15, pp. 303–309. https://doi.org/10.1111/voxs.12545
Ryzhov, I.M. and Bovin, N.V., Mendeleev Commun., 2019, vol. 29, pp. 597–612. https://doi.org/10.1016/j.mencom.2019.11.001
Ryzhov, I.M., Korchagina, E.Y., Popova, I.S., Tyrtysh, T.V., Paramonov, A.S., and Bovin, N.V., Carbohydr. Res., 2016, vol. 430, pp. 59–71. https://doi.org/10.1016/j.carres.2016.04.029
Ryzhov, I.M., Tuzikov, A.B., Perry, H., Korchagina, E.Y., and Bovin, N.V., ChemBioChem, 2019, vol. 20, pp. 131–133. https://doi.org/10.1002/cbic.201800289
Barr, K., Korchagina, E., Ryzhov, I., Bovin, N., and Henry, S., Transfusion, 2014, vol. 54, pp. 2477–2484. https://doi.org/10.1111/trf.12661
Perry, H., Bovin, N., and Henry, S., Transfusion, 2019, vol. 59, pp. 2131–2140. https://doi.org/10.1111/trf.15247
Lee, R.T. and Lee, Y.C., Bioconjug. Chem., 1997, vol. 8, pp. 762–765. https://doi.org/10.1021/bc9700796
Chiu, S.W., Jakobsson, E., Mashl, R.J., and Scott, H.L., Biophys. J., 2002, vol. 83, pp. 1842–1853. https://doi.org/10.1016/S0006-3495(02)73949-0
Smondyrev, A.M. and Berkowitz, M.L., Biophys. J., 1999, vol. 77, pp. 2075–2089. https://doi.org/10.1016/S0006-3495(99)77049-9
Henry, S., Williams, E., Barr, K., Korchagina, E., Tuzikov, A., Ilyushina, N., Abayzeed, S.A., Webb, K.F., and Bovin, N., Sci. Rep., 2018, vol. 8, p. 2845. https://doi.org/10.1038/s41598-018-21186-3
Tyrtysh, T.V., Korchagina, E.Y., Ryzhov, I.M., and Bovin, N.V., Carbohydr. Res., 2017, vol. 449, pp. 65–84. https://doi.org/10.1016/j.carres.2017.06.014
Azev, V.N., Baidakova, L.K., Chulin, A.N., Tuzikov, A.B., Kislitsin, P.G., Molchanov, M.V., and Miroshnikov, A.I., Russ. J. Bioorg. Chem., 2023, vol. 49 (in press).
Meloncelli, P.J. and Lowary, T.L., Carbohydr Res., 2010, vol. 345, pp. 2305–2322. https://doi.org/10.1016/j.carres.2010.08.012
Funding
This study was supported by the Russian Science Foundation (project no. 22-23-00756 “Synthetic glycolipid bioconjugates as instruments for the research of eukaryotic cell”).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest. This research does not contain any studies involving animals or human participants as objects.
Additional information
The article is dedicated to the memory of Academician of the Russian Academy of Sciences Vadim T. Ivanov.
Corresponding author; phone: +7 (495) 330-03-00.
Abbreviations: Ad, adipoyl; Av, aminovaleric acid; Chol, cholesterol; CMG(2), N-carboxymethylglycine based spacer; DOPE, dioleoylphosphatidylethanolamine; DSPE, distearoylphosphatidylethanolamine; DPyPE, diphytanoylphosphatidylethanolamine; GL, glycolipid; nGL, neoglycolipid.
Rights and permissions
About this article
Cite this article
Petrakova, D.O., Savchenko, M.S., Popova, I.S. et al. Synthesis of Glycolipid Analogs Containing A (Type 2) Tetrasaccharide. Russ J Bioorg Chem 49, 785–796 (2023). https://doi.org/10.1134/S1068162023040143
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162023040143