Abstract
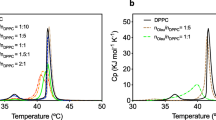
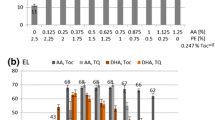
Design and research of liposome structures on the basis of soy phosphatidylcholine (PC) as nanocontainers for delivery of various functional nutraceuticals in tissues and cells of organisms, as well as models for studying biochemical processes in cell membranes, has been of undoubted interest recently. As liposomes are oxidized and destroyed under the influence of light and high temperatures, there are various ways to protect them. In the present work, the effect of temperature on the soy PC liposome oxidation initiated by a water-soluble azo-initiator AAPH was investigated; also the influence of ω-3 α-linolenic fatty acid (ALA) supplement in the liposomes on the process was studied. Inhibition of liposome oxidation by essential oil of clove buds (EOC) (Eugenia caryophyllata Thumb) or liposome encapsulation in sodium caseinate was studied. The possibility to efficiently inhibit oxidation of PC liposomes at T = 37°C by phenolic compounds (eugenol) of EOC was demonstrated; however, at an elevated temperature (60°C) the oxidation rate increased 3–4 times and the rate of inhibition of conjugated diene formation significantly decreased. Addition of ALA to liposomes led to considerable intensification of oxidation. Encapsulation of liposomes in sodium caseinate reduced liposome oxidation by 70%, and the combined effect of sodium caseinate encapsulation and EOC, by 90%. With a temperature increase, the effectiveness of protection by the caseinate envelope significantly decreased, even in the presence of clove oil.






Similar content being viewed by others
REFERENCES
Donelli, D., Rensli, D., and Rid, N., Pishcha i pishchevye dobavki. Rol’ BAD v profilaktike zabolevanii (Food and Nutritional Supplements: The Role of Dietary Supplements in the Prevention of Diseases), Moscow: Mir, 2004, p. 312.
Semenova, M.G. and Dickinson, E., Biopolymers in Food Colloids: Thermodynamics and Molecular Interactions, Leiden: Brill, 2010, p. 421.
McClements, D.J., Nanoparticle- and Microparticle-Based Delivery Systems: Encapsulation, Protection and Release of Active Compounds, New York: CRC Press Taylor and Francis Group, 2015, p. 548.
Thomas, A.H., Catala, A., and Vignoni, M., Biochim. Biophys. Acta, 2016, vol. 1858, pp. 139–145.
Mosca, M., Cerlie, A., and Ambrosone, L., Chem. Phys. Lipids, 2011, vol. 164, pp. 158–165.
Palmina, N.P., Maltseva, E.L., Chasovskaya, T.E., Kasparov, V.V., Bogdanova, N.G., Menshov, V.A., and Trofimov, A.V., Aust. J. Chem., 2014, vol. 67, pp. 858–866.
Chasovskaya, T.E., Plashchina, I.G., and Pal’mina, N.P., Dokl. Akad. Nauk, 2013, vol. 449, pp. 673–77.
Altunkaya, A., Gokmen, V., and Skibsted, L.H., Food Chem., 2016, vol. 190, pp. 25–32.
Lokhmatikov, A.V., Voskoboynikova, N., Cherepanov, D.A., Skulachev, M.V., Steihoff, H.J., Skulachev, V.P., Armen, Y., and Mulkidjanian, A.Y., Hindawi Publ. Corp. Oxid. Med. Cell. Longevity, 2016, vol. 1, article ID 8679469, pp. 1–19.
Misharina, T.A., Alinkina, E.S., Vorobjeva, A.K., Terenina, M.B., and Krikunova, N.I., Appl. Biochem. Microbiol., 2016, vol. 52, pp. 336–341.
Tesoriere, L., Allegra, M., Gentile, C., and Livrea, M.A., Free Rad. Res., 2009, vol. 43, pp. 706–717.
Tan, C., Zhang, Y., Abbas, S., Feng, B., Zhang, X., Xia, S., and Chang, D., Food Function, 2015, vol. 6, pp. 3702–3711.
Semenova, M.G., Zelikina, D.V., Antipova, A.S., Martirosova, E.I., Grigorovich, N.V., Obushaeva, R.A., Shumilina, E.A., Ozerova, N.S., Palmina, N.P., Maltseva, E.L., Kasparov, V.V., Bogdanova, N.G., and Krivandin, F.V., Food Hydrocoll., 2016, vol. 52, pp. 144–160.
Semenova, M.G., Antipova, A.S., Misharina, T.A., Alinkina, E.S., Zelikina, D.V., Martirosova, E.I., Palmina, N.P., Binyukov, V.I., Maltseva, E.L., Kasparov, V.V., Ozerova, N.S., Shumilina, E.A., Baeva, K.A., and Bogdanova, N.G., in Gums and Stabilisers for the Food Industry 18: Hydrocolloid Functionality for Affordable and Sustainable Global Food Solutions, Cambridge: Royal Soc. Chem., 2016, pp. 182–189.
Stroev, E.A., Biologicheskaya khimiya (Biological Chemistry), Moscow: Vysshaya Shkola, 1986.
Pinchuk, I. and Lichtenberg, D., Chem. Phys. Lipids, 2014, vol. 178, pp. 63–76.
McPherson, P.A.C., Bole, A., Cruz, K.A., Young, I.S., and McEneny, J., Chem. Phys. Lipids, 2012, vol. 165, pp. 682–688.
Lee, C., Barnett, J., and Reven, P.D., J. Lipid Res., 1998, vol. 39, pp. 1239–1247.
Hiro-jmi, M., Shoshichi, N., and Reizo, I., J. Biochem., 1984, vol. 95, pp. 51–55.
Abdrakhmanova, A.R., Kozina, E.A., Grabovskii, S.A., Kabal’nova, N.N., and Murinov, Yu.I., in Vserossiiskaya konferentsiya molodykh uchenykh “Okislenie, okislitel’nyi stress, antioksidanty,” Tezisy dokladov (All-Russia Conf. Young Scientists “Oxidation, Oxidative Stress, and Antioxidants”), Moscow: Ross. Univ. Druzhby Narodov, 2008, p. 78.
Emmanuel’, N.M., Denisov, E.T., and Maizus, Z.K., Tsepnye reaktsii okisleniya uglevodorodov v zhidkoi faze (Chain Reactions of Oxidation of Hydrocarbons in Liquid Phase), Moscow: Nauka, 1965.
Denisov, E.T. and Afanas’ev, I.B., Oxidation and Antioxidants in Organic Chemistry and Biology, Boca Raton: CRC Press, 2005.
Jirovetz, L., Buchbauer, L., Stoilova, L., Stoyanova, A., Krastanov, A., and Schmidt, A., J. Agric. Food Chem., 2006, vol. 54, pp. 6303–6307.
Samusenko, A.L., Khim. Rastit. Syr’ya, 2014, no. 2, pp. 97–102.
Brada, P.C., Alfieri, M., Culici, M., and Sasso, M.D., Fitoterapia, 2007, vol. 78, pp. 396–400.
Alinkina, E.S., Vorob’eva, A.K., Misharina, T.A., Medvedeva, I.B., Fatkullina, L.D., and Burlakova, E.B., Prikl. Analit. Khim., 2011, vol. 2, no. 2, pp. 28–36.
Semenova, M.G., Food Hydrocoll., 2017, vol. 68, pp. 114–121.
Smotrova, N.V., Chebotarev, S.A., Vorob’eva, N.S, Teisen, C., Babeeva, G., Kitl, T., and Antipova, A.C., in XVI Ezhegodnaya mezhdunarodnaya molodezhnaya konferentsiya “Biokhimicheskaya fizika” Inst. Biokhim. Fiz.–VUZy, Sbornik Trudov (XVI Annual Int. Youth Conf. “Biochemical Physics,” Inst. Biochem. Phys.–High Schools), Moscow: Ross. Univ. Druzhby Narodov, 2017, pp. 83–87.
Semenova, M.G., Antipova, A.S., Belyakova, L.E., Polikarpov, Yu.N., Anokhina, M.S., Grigorovich, N.V., and Moiseenko, D.V., Food Hydrocoll., 2014, vol. 42, pp. 149–161.
Istarova, T.A., Semenova, M.G., Sorokoumova, G.M., Selishcheva, A.A., Belyakova, L.E., Polikarpov, YuN., and Anikhina, M.S., Food Hydrocoll., 2005, vol. 19, pp. 429–440.
Author information
Authors and Affiliations
Additional information
Translated by N. Onishchenko
Abbreviations: AAPH, 2,2'-azobis(amidinopropane) dihydrochloride; PC, phosphatidylcholine; ALA, alpha-linolenic acid; DPPH, diphenylpicrylhydrazine; AO, antioxidative; AR, antiradical; Cas-Na, sodium caseinate; ROS, reactive oxygen species; DC, diene conjugates; KD, ketodienes; PUFA, polyunsaturated fatty acids; LP, lipid peroxidation; EOC, essential oil of clove buds.
Corresponding author: phone: +7 (495) 939-73-51; +7 (916) 087-98-68; e-mail: Natnik84s@yandex.ru.
Rights and permissions
About this article
Cite this article
Sazhina, N.N., Antipova, A.S., Semenova, M.G. et al. Initiated Oxidation of Phosphatidylcholine Liposomes with Some Functional Nutraceuticals. Russ J Bioorg Chem 45, 34–41 (2019). https://doi.org/10.1134/S1068162019010138
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162019010138