Abstract
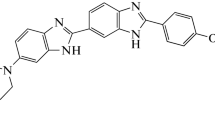
Seven fluorescent symmetric dimeric bisbenzimidazoles DB(n) capable of occupying up to one turn of the double-stranded B-form DNA have been designed and synthesized with the aim to develop DNAdependent enzyme inhibitors. The DB(n) compounds contain four 2,6-substituted benzimidazole fragments and differ by the length of the oligomethylene linker (n = 1, 2, 6, 8, 9, 10, 12) between the bisbenzimidazole blocks. The formation of DB(n)–double-stranded DNA complexes and the localization of the ligands in the DNA minor groove have been confirmed by a number of physicochemical methods.
Similar content being viewed by others
Abbreviations
- BOP:
-
(benzotriazol-1-yloxy)tris(dimethylamino)phoshonium hexafluorophosphate
- DB(n):
-
dimeric bisbenzimidazoles
- DIPEA:
-
N,N-diisopropylethylamine
- MB:
-
monomeric bisbenzimidazole
- dsDNA:
-
double-stranded DNA
- PEG:
-
polyethylene glycol
- CLCD:
-
cholesteric liquid crystalline dispersion
References
Ivanov, A.A., Salyanov, V.I., Streltsov, S.A., Cherepanova, N.A., Gromova, E S., and Zhuze, A.L., Russ. J. Bioorg. Chem., 2011, vol. 37, pp. 472–482.
Baraldi, P.G., Bovero, A., Fruttarolo, F., Preti, D., Tabrizi, M.A., Pavani, M.G., and Romagnoli, R., Med. Res. Rev., 2004, vol. 32, pp. 475–528.
Zewail-Foote, M. and Hurley, L.H., Anti-Cancer Drug Des., 1999, vol. 14, pp. 1–9.
Chaires, J.B., Curr. Opin. Struct. Biol., 1998, vol. 8, pp. 314–320.
Strekowski, L. and Wilson, B., Mutat. Res., 2007, vol. 623, pp. 3–13.
Wilson, W.D., Nguyen, B., Tanious, F.A., Mathis, A., Hall, J.E., Stephens, C.E., and Boykin, D.W., Curr. Med. Chem. Anti-Cancer Agents, 2005, vol. 5, pp. 389–408.
Neidle, S., Nat. Prod. Rep., 2001, vol. 18, pp. 291–309.
Wiederholt, K., Rajur, S.B., Giuliano, J., O’Donnell,M., and McLaughlin, L., J. Am. Chem. Soc., 1996, vol. 118, pp. 7055–7062.
Geierstanger, B.H. and Wemmer, D.E., Annu. Rev. Biophys. Biomol. Struct., 1995, vol. 24, pp. 463–493.
Chenoweth, D.M. and Dervan, P.B., J. Am. Chem. Soc., 2010, vol. 132, pp. 14521–14529.
Meng, L.-H., Liao, Z.-Y., and Pommier, Y., Curr. Top. Med. Chem., 2003, vol. 3, pp. 305–320.
Bailly, C., Curr. Med. Chem., 2000, vol. 7, pp. 39–58.
Turner, P.R. and Denny, W.A., Curr. Drug Targets, 2000, vol. 1, pp. 1–14.
Chiang, S.Y., Welch, J.J., Rauscher, F.J., 3rd, and Beerman, T.A., J. Biol. Chem., 1996, vol. 271, pp. 23999–24004.
Welch, J.J., Rauscher, F.J., 3rd, and Beerman, T.A., J. Biol. Chem., 1994, vol. 269, pp. 31051–31058.
Gabelica, V., Rosu, F., Houssier, C., and De Pauw, E., Rapid Commun. Mass Spectrom., 2000, vol. 14, pp. 464–467.
White, C.M., Satz, A.L., Gawron, L.S., Bruice, T.C., and Beerman, T.A., Biochim. Biophys. Acta, 2002, vol. 1574, pp. 100–108.
Reddy, B.S.P., Sondhi, S.M., and Lown, J.W., Pharmacol. Ther., 1999, vol. 84, pp. 1–111.
Nelson, S.M., Ferguson, L.R., and Denny, W.A., Mutat. Res., 2007, vol. 623, pp. 24–40.
Dervan, P.B. and Edelson, B.S., Curr. Opin. Struct. Biol., 2003, vol. 13, pp. 284–299.
Reddy, B.S., Sharma, S.K., and Lown, J.W., Curr. Med. Chem., 2001, vol. 8, pp. 475–508.
Wemmer, D.E., Annu. Rev. Biophys. Biomol. Struct., 2000, vol. 29, pp. 439–461.
Bailly, C. and Chaires, J.B., Bioconjug. Chem., 1998, vol. 9, pp. 513–538.
Cherepanova, N.A., Ivanov, A.A., Maltseva, D.V., Minero, A.S., Gromyko, A.V., Streltsov, S.A., Zhuze, A.L., and Gromova, E.S., J. Enzyme Inhib. Med. Chem., 2011, vol. 26, pp. 295–300.
Loewe, H. and Urbanietz, J., Arzneim.-Forsch. (Drug Res.), 1974, vol. 24, pp. 1927–1933.
Evdokimov, Yu.M., Salyanov, V.I., Nechipurenko, Y.D., Skuridin, S.G., Zakharov, M.A., Spener, F., and Palunbo, M., Mol. Biol., (Moscow), 2003, vol. 37, pp. 293–306.
Yevdokimov, Yu.M., Skuridin, S.G., Nechipurenko, Y.D., Zakharov, M.A., Salyanov, V.I., Kurnosov, A.A., Kuznetsov, V.D., and Nikiforov, V.N., Int. J. Biol. Macromol., 2005, vol. 36, pp. 103–115.
Gromyko, A.V., Salyanov, V.I., Strel’tsov, S.A., Oleinikov, V.A., Korolev, S.P., Gottikh, M.B., and Zhuze, A.L, Russ. J. Bioorg. Chem., 2007, vol. 33, pp. 569–578.
Pjura, P.E., Grzeskowiak, K., and Dickerson, R.E., J. Mol. Biol., 1987, vol. 197, pp. 257–271.
Teng, M.-K., Usman, N., Frederick, C.A., and Wang, A.H., Nucleic Acids Res., 1988, vol. 16 P, pp. 2671–2690.
Lisitsyna, E.S., Durandin, N.A., Ivanov, A.A., Streltsov, S.A., Susova, O.Yu., Shtil, A.A., Zhuze, A.L., and Kuzmin, V.A., Mol. Biol. (Moscow), 2012, vol. 46, pp. 823–827.
Sintezy organicheskikh preparatov. (Organic Synthesis., Moscow: IL, 1953, vol. 1, pp. 357–358.
Martin, R.F., Pardee, M., Kelly, D.P., and Mack, P.O.-L., Aust. J. Chem., 1986, vol. 39, pp. 373–381.
Wells, R.D., Larson, J.E., Grant, R.C., Shortle, B.E., and Cantor, C.R., J. Mol. Biol., 1970, vol. 54, pp. 465–497.
Author information
Authors and Affiliations
Corresponding author
Additional information
For XIV, see [1].
Original Russian Text © A.A. Ivanov, V.I. Salyanov, A.L. Zhuze, 2016, published in Bioorganicheskaya Khimiya, 2016, Vol. 42, No. 2, pp. 205–213.
Rights and permissions
About this article
Cite this article
Ivanov, A.A., Salyanov, V.I. & Zhuze, A.L. DNA sequence-specific ligands: XV. Synthesis and spectral characteristics of a new series of dimeric bisbenzimidazoles DB(1, 2, 6, 8, 9, 10, 12). Russ J Bioorg Chem 42, 183–190 (2016). https://doi.org/10.1134/S1068162016020059
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162016020059