Abstract
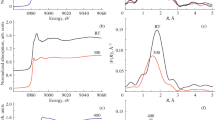
The influence of temperature conditions for synthesis on the nearest-neighbor environment of copper atoms in copper–mordenite zeolites produced by solid-phase ion exchange has been investigated. The models of the local atomic structure of active copper centers in copper–mordenite zeolite at 300 and 400°C have been established using two complementary techniques: X-ray absorption spectroscopy and density functional theory. It has been found that, at 300°C, the copper atom does not have another one in its nearest-neighbor environment (monocentric model), whereas at 400°C the center contains at least two copper atoms, which form Cu–O–Cu bridges. The structural parameters of Cu–O bonds have been determined.





Similar content being viewed by others
REFERENCES
S. E. Bozbag, E. M. C. Alayon, J. Pecháček, M. Nachtegaal, M. Ranocchiari, and J. A. van Bokhoven, Catal. Sci. Technol. 6 (13), 5011 (2016). https://doi.org/10.1039/C6CY00041J
E. M. Alayon, M. Nachtegaal, M. Ranocchiari, and J. A. van Bokhoven, Chem. Commun. 48 (3), 404 (2012). https://doi.org/10.1039/C1CC15840F
S. Grundner, W. Luo, M. Sanchez-Sanchez, and J. A. Lercher, Chem. Commun. 52 (12), 2553 (2016). https://doi.org/10.1039/C5CC08371K
E. M. C. Alayon, M. Nachtegaal, E. Kleymenov, and J. A. van Bokhoven, Microporous Mesoporous Mater. 166, 131 (2013). https://doi.org/10.1016/j.micromeso.2012.04.054
J. S. Woertink, P. J. Smeets, M. H. Groothaert, M. A. Vance, B. F. Sels, R. A. Schoonheydt, and E. I. Solomon, Proc. Natl. Acad. Sci. U.S.A. 106 (45), 18908 (2009). https://doi.org/10.1073/pnas.0910461106
C. Buono, A. Martini, I. A. Pankin, D.K. Pappas, C. Negri, K. Kvande, K. A. Lomachenko, and E. Borfecchia, Radiat. Phys. Chem. 175, 108111 (2020). https://doi.org/10.1016/j.radphyschem.2018.12.031
V. L. Sushkevich, O. V. Safonova, D. Palagin, M. A. Newton, and J. A. van Bokhoven, Chem. Sci. 11, 5299 (2020). https://doi.org/10.1039/D0SC01472A
N. V. Beznis, B. M. Weckhuysen, and J. H. Bitter, Catal Lett. 138 (1–2), 14 (2010). https://doi.org/10.1007/s10562-010-0380-6
C. Lamberti, S. Bordiga, M. Salvalaggio, G. Spoto, A. Zecchina, F. Geobaldo, G. Vlaic, and M. Bellatreccia, J. Phys. Chem. B 101 (3), 344 (1997). https://doi.org/10.1021/jp9601577
C. Lamberti, S. Bordiga, A. Zecchina, M. Salvalaggio, F. Geobaldo, and C. Otero Areán, J. Chem. Soc., Faraday Trans. 94 (10), 1519 (1998). https://doi.org/10.1039/A708778K
V. V. Srabionyan, G. B. Sukharina, S. Y. Kaptelinin, V. A. Durymanov, A. M. Ermakova, T. I. Kurzina, L. A. Avakyan, and L. A. Bugaev, Phys. Solid State 62 (7), 1222 (2020). https://doi.org/10.1134/S1063783420070252
L. Braglia, E. Borfecchia, L. Maddalena, S. Øien, K. A. Lomachenko, A. L. Bugaev, S. Bordiga, A. V. Soldatov, K. P. Lillerud, and C. Lamberti, Catal. Today 283, 89 (2017). https://doi.org/10.1016/j.cattod.2016.02.039
G. Smolentsev, G. Sukharina, A. V. Soldatov, and L. X. Chen, Nucl. Instrum. Methods Phys. Res., Sect. A 603 (1–2), 122 (2009). https://doi.org/10.1016/S0168-9002(09)00773-6
G. B. Sukharina, A. N. Kravtsova, A. V. Soldatov, Y. V. Zubavichus, N. A. Kryuchkova, and N. Mazalov, J. Phys.: Conf. Ser. 190 (1), 012148 (2009).
J. P. Perdew, K. Burke, and M. Ernzerhof, Phys. Rev. Lett. 77 (18), 3865 (1996). https://doi.org/10.1103/PhysRevLett.77.3865
P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, G. L. Chiarotti, M. Cococcioni, I. Dabo, A. Dal Corso, S. de Gironcoli, S. Fabris, G. Fratesi, R. Gebauer, et al., J. Phys.: Condens. Matter. 21 (39), 395502 (2009).
B. Ravel and M. Newville, J. Synchrotron Radiat. 12 (4), 537 (2005). https://doi.org/10.1107/S0909049505012719
Y. Joly, Phys. Rev. B 63 (12), 125120 (2001).
G. Henkelman and H. Jónsson, J. Chem. Phys. 113 (22), 9978 (2000). https://doi.org/10.1063/1.1323224
G. Henkelman, B. P. Uberuaga, and H. A. Jónsson, J. Chem. Phys. 113 (22), 9901 (2000). https://doi.org/10.1063/1.1329672
E. L. Kolsbjerg, M. N. Groves, and B. Hammer, J. Chem. Phys. 145 (9), 094107 (2016). https://doi.org/10.1063/1.4961868
J. Cejka, Zeolites and Ordered Mesoporous Materials: Progress and Prospects (Elsevier, Prague, 2005).
G. R. Hays, W. A. van Erp, N. C. M. Alma, P. A. Couperus, and R. Huis, Zeolites 4 (4), 377 (1984). https://doi.org/10.1016/0144-2449(84)90015-0
C. A. Fyfe, G. C. Gobbi, and G. L. Kennedy, J. Phys. Chem. 88 (15), 3248 (1984). https://doi.org/10.1021/j150659a023
C. Baerloche and L. B. McCusker, Database of Zeolite Structures (1996).
T. Takaishi, M. Kato, and K. Itabashi, Zeolites 15 (1), 21 (1995). https://doi.org/10.1016/0144-2449(94)00015-K
L. A. Bugaev, L. A. Avakyan, V. V. Srabionyan, and A. L. Bugaev, Phys. Rev. B 82 (6), 064204 (2010). https://doi.org/10.1103/PhysRevB.82.064204
ACKNOWLEDGMENTS
The authors thank Prof. J. van Bokhoven from the Swiss Federal Institute of Technology (ETH) for assistance. L.A. Bugaev and L.A. Avakyan are indebted to the administration of Southern Federal University for support.
Funding
This study was supported by the Russian Foundation for Basic Research, grant no. 18-32-000586_mol_a.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by V. Isaakyan
Rights and permissions
About this article
Cite this article
Pryadchenko, V.V., Sukharina, G.B., Ermakova, A.M. et al. Local Structure of Copper Centers Obtained during Solid-Phase Synthesis in Copper–Mordenite Zeolite. Tech. Phys. 66, 1018–1024 (2021). https://doi.org/10.1134/S1063784221070124
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1063784221070124