Abstract
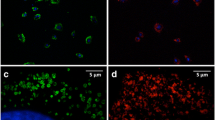
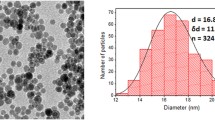
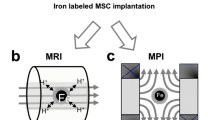
This study evaluated the feasibility of using uncoated iron (II, III) oxide nanoparticles (IONP) obtained by electric explosion of wire in air for labelling living mesenchymal stromal cells and their subsequent visualization by magnetic resonance imaging (MRI) using the 1T and 1.5T clinical MRI scanners. The uptake of uncoated IONP by MSC was demonstrated for the wide range of IONP concentration in the cell culture medium. The cells did not change their proliferative activity, viability, and the set of surface markers. Iron oxide nanoparticles obtained by an electric explosion of wire in an atmosphere of air had a shape close to spherical. According to dynamic lateral light scattering, laser diffraction, and transmission electron microscopy, the particle size varied from 14 to 136 nm. Particles up to 136 nm accounted for 75%, while particles less than 36 nm accounted for 10%. A wide range of particle sizes made it possible to select MRI parameters suitable for labelled cells detection in animal tissues in both the T2 and the T1 relaxation mode.






Similar content being viewed by others
REFERENCES
C. Villa, S. Erratico, P. Razini, F. Fiori, F. Rustichelli, Y. Torrente, and M. Belicchi, Int. J. Mol. Sci. 11 (3), 1070 (2010). https://doi.org/10.3390/ijms11031070
D. J. Korchinski, M. Taha, R. Yang, N. Nathoo, and J. F. Dunn, Magn. Reson. Insights, No. 8, 15 (2015). https://doi.org/10.4137/MRI.S23557
Ch. Kittel, Introduction to Solid State Physics (Wiley, New York, 1953).
A. I. Zhernovoy, Nauch. Priborostroen. 28 (2), 45 (2018).
A. G. Kolhatkar, A. C. Jamison, D. Litvinov, R. C. Willson, and T. R. Lee, Int. J. Mol. Sci. 14 (8), 15977 (2013). https://doi.org/10.3390/ijms140815977
N. M. Yudintceva, Y. A. Nashchekina, M. I. Blinova, L. V. Smagina, M. A. Shevtsov, and I. V. Voronkina, FEBS J. 284 (S1), 382 (2017). https://doi.org/10.1111/febs.14174
Wahajuddin and S. Arora, Int. J. Nanomed. 7, 3445 (2012). https://doi.org/10.2147/IJN.S30320
H. Elkhenany, M. Abd Elkodous, N. I. Ghoneim, T. A. Ahmed, S. M. Ahmed, I. K. Mohamed, and N. El-Badri, Int. J. Biol. Macromol. 143, 763 (2019). https://doi.org/10.1016/j.ijbiomac.2019.10.031
L. S. Arias, J. P. Pessan, A. P. M. Vieira, T. M. Toito de Lima, A. C. B. Delbem, and D. R. Monteiro, Antibiotics (Basel, Switz.) 7 (2), 46 (2018). https://doi.org/10.3390/antibiotics7020046
A. Jordan, P. Wust, R. Scholz, B. Tesche, H. Fähling, T. Mitrovics, T. Vogl, J. Cervós-navarro, and R. Felix, Int. J. Hyperthermia 12 (6), 705 (1996). https://doi.org/10.3109/02656739609027678
Q. Feng, Y. Liu, J. Huang, K. Chen, J. Huang, and K. Xiao, Sci. Rep. 8 (1), 2082 (2018). https://doi.org/10.1038/s41598-018-19628-z
M. I. Lerner, Doctoral Dissertation in Engineering (Tomsk Polytech. Univ., Tomsk, 2007).
M. I. Lerner, N. V. Svarovskaya, S. G. Psakhie, and O. V. Bakina, Nanotechnol. Russ. 4 (11–12), 741 (2009). https://doi.org/10.1134/S1995078009110019
DelsaMax PRO Light Scattering Analyzer. Instructions for Use (Beckman Coulter, 2013), p. 188.
I. Mindukshev, S. Gambaryan, L. Kehrer, C. Schuetz, A. Kobsar, N. Rukoyatkina, V. O. Nikolaev, A. Krivchenko, S. P. Watson, U. Walter, and J. Geiger, Clin. Chem. Lab. Med. 50 (7), 1253 (2012).
A. A. Aisenstadt, N. I. Enukashvili, T. L. Zolina, L. V. Alexandrova, and A. B. Smoljaninov, Mechnikov Vestn. Severo-Zapad. Gos. Med. Inst. 7 (2), 14 (2015).
V. V. Bagaeva, A. A. Aizenshtadt, L. V. Aleksandrova, et. al. RF Patent No. 2620981 C2, Byull. Izobret., No. 16, 1 (2017).
M. Dominici, K. Le Blanc, I. Mueller, I. Slaper-Cortenbach, F. Marini, D. Krause, R. Deans, A. Keating, D. J. Prockop, and E. Horwitz, Cytotherapy 8 (4), 315 (2006). https://doi.org/10.1080/14653240600855905
L. Li, K. Y. Mak, J. Shi, H. K. Koon, C. H. Leung, C. M. Wong, C. W. Leung, C. S. Mak, N. M. Chan, W. Zhong, K. W. Lin, E. X. Wu, and P. W. Pong, J. Nanosci. Nanotechnol. 12 (12), 9010 (2012). https://doi.org/10.1166/jnn.2012.6755
R. Das, B. K. Das, R. Shukla, and A. Shyam, J. Phys.: Conf. Ser. 390 (1), 2051 (2012). https://doi.org/10.1088/1742-6596/390/1/012051
Q. Li, C. W. Kartikowati, S. Horie, T. Ogi, T. Iwaki, and K. Okuyama, Sci. Rep. 7 (1), 9894 (2017). https://doi.org/10.1038/s41598-017-09897-5
N. V. Abramov and P. P. Gorbik, Poverkhnost, No. 4(19), 246 (2012).
U. Jeong, X. Teng, Y. Wang, H. Yang, and Y. Xia, Adv. Mater. 19 (1), 33 (2007). https://doi.org/10.1002/adma.200600674
ACKNOWLEDGMENTS
The authors are grateful to the staff of the Confocal Microscopy and Image Analysis Group of the Center for Cell Technologies of the Institute of Cytology of the Russian Academy of Sciences G.I. Stein and M.L. Vorob’ev for assistance in the work with an Olympus FV3000 confocal microscope (Nikon, Japan).
Funding
This work was carried out within the framework of research work (no. AAAA-A18-118052990081-0) of a state order to the Ministry of Health of the Russian Federation no. 056-00105-18-00.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by A. Barkhash
Rights and permissions
About this article
Cite this article
Enukashvily, N.I., Kotkas, I.E., Bogolyubov, D.S. et al. Detection of Cells Containing Internalized Multidomain Magnetic Iron (II, III) Oxide Nanoparticles Using the Magnetic Resonance Imaging Method. Tech. Phys. 65, 1360–1369 (2020). https://doi.org/10.1134/S1063784220090145
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1063784220090145