Abstract
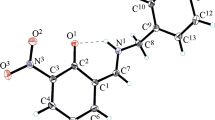
Azomethine, derived from 3,5-dichlorosalicylaldehyde and allylamine, was synthesized and studied by X-ray diffraction. The compound exists in the enol-imine tautomeric form (the hydrogen atom is located on the oxygen atom). The structure is stabilized by the intramolecular О(1)–Н(1А)···N(1) hydrogen bond (О–Н, 0.82 Å; H···N, 1.91 Å; O···N, 2.580 Å; О–H–N, 138°).

Similar content being viewed by others
REFERENCES
V. A. Bren’ and V. I. Minkin, Izv. Vyssh. Uchebn. Zaved., Khim., Khim. Tekhnol. 25 (6), 665 (1982).
V. I. Minkin, L. P. Olekhnovich, and Yu. A. Zhdanov, Molecular Design of Tautomeric Compounds (D. Reidel, Doldrecht, 1988).
A. D. Garnovskii, D. A. Garnovskii, I. S. Vasil’chenko, et al., Usp. Khim. 66 (5), 434 (1997).
K. Ogawa, T. Fujiwara, and J. Harada, Mol. Cryst. Liq. Cryst. Sci. Technol. A 344 (1), 169 (1999).
Y. Elerman, M. Kabak, E. Kavlakoglu, et al., J. Mol. Struct. 510 (1–3), 207 (1999).
G. M. Sheldrick, Acta Crystallogr. A 64 (1), 112 (2008).
H. A. Rudbari, M. Khorshidifard, B. Askari, et al., Polyhedron 100, 180 (2015).
V. S. Sergienko, V. L. Abramenko, Yu. E. Gorbunova, and A. V. Churakov, Russ. J. Inorg. Chem. 62 (2), 175 (2017).
V. S. Sergienko, V. Abramenko, and Yu. E. Gorbunova, Russ. J. Inorg. Chem. 62 (8), 1038 (2017).
V. S. Sergienko, V. L. Abramenko, and Yu. E. Gorbu-nova, Crystallogr. Rep. 64 (1), 67 (2019).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by T. Safonova
Rights and permissions
About this article
Cite this article
Sergienko, V.S., Abramenko, V.L. & Gorbunova, Y.E. Prototropic Tautomerism of Salicylideneimines. Crystal Structure of 3,5-Dichlorosalicylideneallylimine. Crystallogr. Rep. 65, 53–55 (2020). https://doi.org/10.1134/S106377452001023X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S106377452001023X