Abstract
Melanoma is a malignant tumor transforming from normal melanocytes, with strong invasion and low survival rate. Nitidine chloride (NC) is a natural benzophenidine alkaloid extracted from the roots of traditional Chinese medicine zanthoxylum nitidum. In recent years, many studies have found that NC may have strong anti-tumor activity. However, the efficacy of NC against melanoma has rarely been reported, and the potential molecular mechanism remains unknown. Our study explored the effects of NC on the proliferation and apoptosis of human melanoma A375 and A2058 cells. CCK-8 was utilized to detect the effects of different concentrations of NC on the proliferation of A375 and A2058 cells. Within a certain concentration and time range, NC can inhibit the viability of A375 and A2058 cells significantly in a time-dose-dependent manner. In addition, NC-induced apoptosis was confirmed by DAPI staining and flow cytometry analysis of Annexin V-FITC/PI. Moreover, we observed that the ratio of Bax/Bcl-2 in the cytoplasm increases, activating Caspase-3/-9 and ultimately inducing apoptosis via the mitochondrial pathway. Our study indicated that NC has anti-tumor properties against human melanoma cells through inhibiting proliferation and inducing apoptosis. Therefore, the results provide new insights for future work on the utilization of NC in malignant melanoma treatment.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
INTRODUCTION
Melanoma, a malignant tumor arising from melanocytes, is the most mortal skin cancer. Due to its strong invasiveness, melanoma accounts for about 75% of skin cancer deaths though it only accounts for 4% of skin cancer cases (Davis et al., 2019). At present, the main therapies for melanoma are surgery, chemotherapy and radiotherapy (Agha and Tarhini, 2017). Surgical resection of skin lesions can cure early melanoma, when melanoma has metastasized, radiotherapy and chemotherapy are often applied. However, all of these therapies still have some disadvantages due to various side effects and drug resistance. The survival rate of melanoma patients has significantly increased thanks to the remarkable strides made in targeted therapy and immunotherapy, including the development of BRAF, CTLA-4, and PD-1 inhibitors. However, these drugs are expensive and prone to secondary resistance in a short period of time (Ugurel et al., 2017). Therefore, it remains urgent to develop more safe, effective and affordable potential anti-tumor drugs.
In recent years, more and more evidence suggest that many traditional Chinese medicinal herbs and their phytochemicals exhibit potential anti-tumor effect, which provides an alternative therapy strategy for malignant melanoma treatment. Therefore, it is of great clinical significance to identify effective anti-melanoma drugs from Chinese herbal medicine. Many traditional Chinese medicines contain good efficacy, mild side effects and numerous targets, and the main anti-tumor components have been identified (Li and Zhang, 2014). Nitidine, a quaternary ammonium alkaloid, is a Chinese traditional herb. It is mainly extracted from the root of Zanthoxylum nitidum, which is often distributed in Northeastern Asia (Fig. 1) (Cui et al., 2020). NC has multiple biological activities since its discovery in 1959, including anti-malarial, anti-fungal and anti-inflammatory (Bouquet et al., 2012; Del Poeta et al., 1999; Yang et al., 2019a). In recent years, literature revealed that NC also has anti-tumor properties. It has shown marked effects in inhibiting cell proliferation and promoting apoptosis in malignant tumor cells such as ovarian cancer cells, colorectal cancer cells and human glioblastoma cells (Khan et al., 2018). While it is rarely reported whether NC can also inhibit proliferation and promote apoptosis of melanoma cells.
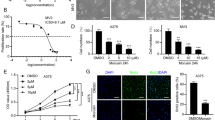
(a) Chemical structure of nitidine chloride. Cell viability of A375 (b) and A2058 (c) cells after treatment of NC in different concentration (** P < 0.01 vs. control); Morphological changes of A375 (d) and A2058 (e) cells were observed under inverted microscope after treating with different concentrations of NC for 24 h (×200).
In this article, we aim at investigating the effect of NC in human melanoma A375 and A2058 cells and discuss the molecular insight into NC-involved anti-tumor feature in vitro.
MATERIALS AND METHODS
Cell Lines and Cell Culture
NC (cat. no. 413917; purity: 98%) was purchased from Beijing Bailingwei Technology Co., Ltd. The human melanoma A375 cell line applied in this experiment was donated by the Dermatology Hospital (Institute) of the Chinese Academy of Medical Sciences and A2058 cell line was purchased from Procell Life Science & Technology Co., Ltd. All cells were processed in DMEM high-glucose culture medium (Gibco, USA) supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin in a humidified incubator in 5% CO2 at 37°C. NC was dissolved in Dimethyl sulfoxide (DMSO) and diluted with culture medium. The terminal concentration of DMSO was less than 0.1% in the experiment.
CCK-8 Assay
Briefly, 1 × 105 cells/mL were seeded in 96-well plates for 24 h. Cells were then treated with NC at various concentrations for 24, 48 and 72 h. At the end of incubation, 10 µL CCK‑8 solution (Shanghai Yeasen Biotechnology Co., Ltd.) was added and incubated for an additional 1–4 h at 37°C. In the presence of an electron carrier, 2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium monosodium salt (WST-8) in the CCK8 kit can be reduced by dehydrogenase in the mitochondria to produce orange-yellow methylate products (formazan). The color’s hue is negatively correlated with cytotoxicity and inversely correlated with cell proliferation. Finally, the absorbance at 450 nm was determined in each well using the PerkinElmer EnSpire microplate reader (Promega, USA).
DAPI Staining
2 × 105 cells/mL were seeded in 6-well plates for 24 h in order to observe morphological changes. After treated with NC at various concentrations, the cells were fixed with 4% paraformaldehyde. After 15–30 min, the cells were permeabilized with 0.5% Triton X-100 for 15 min. Next, the cells were stained with DAPI at room temperature for 5–15 min. Finally, after the cells were washed with PBS three times, fluorescence images were captured using a laser scanning microscope (Nikon TE2000-U, Janpan).
Reverse Transcription-Quantitative Polymerase Chain Reaction (RT‑qPCR)
Total RNA was extracted from cells using trizol reagent (cat. no. 15596026, Thermo Fisher Scientific, Inc.) following the manufacturer’s instructions. RNA concentration was assessed using a nucleic acid concentration detector (Thermo Fisher Scientific, Inc.). Then cDNA was synthesized by reverse transcription from 2 μg RNA using All-in-One™ First-Strand cDNA Synthesis Kit (cat. no. QP006, iGeneBio, Inc.) according to the manufacturer’s instructions. Real time PCR was performed with the All-in-one qPCR Mix (cat. no. QP001, iGeneBio, Inc.) to examine the mRNA level. GAPDH was used as the standard reference. The thermocycling conditions were as follows: initial denaturing step at 95°C for 10 min, followed by 40 cycles of denaturing step at 95°C for 10 s, annealing step at 60°C for 20 s and final extension at 72°C for 5 min. The comparative 2‑ΔΔCt method was used to calculate the relative expression level of mRNA (Livak and Schmittgen, 2001). The experiments were performed in triplicate.
The primer sequences (5'–3') used were as follows: Bcl-2 forward, 5'-AGTACCTGAACCGGCACCT-3' and reverse, 5'-GCCGTACAGTTCCACAAAGG-3'; Bax forward, 5'-CAGGATGCGTCCACCAAGA-3' and reverse, 5'-CAGTTGAAGTTGCCGTCAGAA-3'; Caspase-3 forward, 5'-GATCGTTGTAGAAGTCTAACTGGAA-3' and reverse, 5'-CACCACTGTCTGTCTCAATGC-3'; Caspase-9 forward, 5'-GGGCTCACTCTGAAGACCTG-3' and reverse, 5'-CCCGAGTGAGACTTCTGGAC-3'; GAPDH forward, 5'-CGCTGAGTACGTCGTGGAGTC-3' and reverse, 5'-GCTGATGATCTTGAGGCTGTTGTC-3'.
Western Blotting Analysis
Cells were seeded into T25 culture flask and incubated for 24 h. Following the administration of various concentrations of NC for 24 hours, the cells were collected. The isolation of proteins was conducted on ice for 30 min by using RIPA lysis buffer (Solarbio, Bejing, China). Ultrasonic cracking was then performed 10 times, 5 s each, 3 s apart. Next, the lysates were placed on ice for 15 min and then centrifuged at 16 000 g at 4°C for 15 min. Protein concentration was determined by a BCA Protein Assay kit (Solarbio, Bejing, China). Cell protein lysates were equalized between samples using 4× loading buffer and then cooked in 100°C water bath for 10 min to denature the protein. The protein loading mass of each well was 30 μg. Samples were electrophoresed through 10% SDS-PAGE and transferred to PVDF membranes. The membranes were blocked with 5% fat-free milk for 2 h at room temperature. Then, the membranes were incubated with the diluted primary antibodies at 4°C overnight. The primary antibodies included: Bcl-2 (cat. no. 80313-1-RR; 1 : 5000; Proteintech), Bax (cat. no. 50599-2-Ig; 1 : 10 000; Proteintech), Cleaved Caspase-3 (cat. no. 19677-1-AP; 1 : 500; Proteintech), Cleaved Caspase-9 (cat. no. 10380-1-AP; 1 : 500; Proteintech), GAPDH (cat. no. AC001; 1 : 5000; ABclonal). TBST washed membranes three times for 10 min each on a shaking table, and then the membranes were incubated with the diluted secondary antibody, Dylight 800 Goat Anti-Rabbit IgG (cat. no. A23920, 1 : 2000, Abbkine), at 37°C for 1 h. TBST washed membranes three times for 10 min each. Finally, the membranes were scanned on Odyssey CLx imaging analyzer (LI-COR, USA). Image J software was used for densitometry.
Flow Cytometry
Human melanoma A375 and A2058 cells were treated with different concentrations of NC for 24 h, and then cells were digested into single cells using 0.25% EDTA-free trypsin. Lastly, the cells were stained with the Annexin V-FITC and PI solution (BD, USA) at 4°C. Cell apoptosis was calculated after 10–15 min by flow cytometry.
Statistical Analysis
The results are presented as mean ± standard deviation and each experiment was repeated at least three times. The statistical significance was determined by one-way ANOVA to compare three or more groups or by Student’s t-test for the comparison between two groups. P < 0.05 was considered as statistically significant.
RESULTS
NC Treatment Inhibited Human Melanoma A375 and A2058 Cells Proliferation
Our study elucidated the cytotoxic effect of NC on human melanoma A2058 and A375 cells. The cell viability in the presence of various concentrations of NC was analyzed by CCK8 assay. The results manifested that NC treatment could inhibit the cells proliferation in comparison with 0 μmol NC group (P < 0.01). Then A375 and A2058 cells treated with indicated concentrations of NC were observed under inverted microscope. It was found that the growth of cells was inhibited with the increase of NC concentration (Fig. 1).
NC Treatment Induced the Apoptosis of Human Melanoma A375 and A2058 Cells
DAPI staining was observed under inverted fluorescence microscope (Fig. 2). The outcomes showed that the nuclei of negative control group were regular in shape, consistent in size and uniform in distribution, while the nuclei of the NC group were irregular, accompanied by the formation of apoptotic bodies with signature dense staining, indicating that NC can promote the apoptosis of A375 and A2058 cells. To estimate the effect of NC on apoptosis, flow cytometry analysis using double staining with Annexin V‑FITC/PI was performed in A375 and A2058 cells. After being treated with NC for 24 h, compared with the control group, the apoptotic rates in the treated cells significantly increased in a dose-dependent manner (Fig. 3). These results manifested that NC induced apoptosis in A375 and A2058 cells.
Apoptotic morphology after NC treatment for 24 h in melanoma cells was detected by DAPI staining. In morphological analysis, NC induced apoptosis recognized as nuclear chromatin condensation and fragmentation in A375 (a) and A2058 (b) cells (×200). Quantification of DNA fragmentation and nuclear condensation determined by DAPI in A375 (c) and A2058 (d) cells treated with NC for 24 h. The arrows indicate chromatin condensation in A375 and A2058 cells (* P < 0.05 vs. control, ** P < 0.01 vs. control).
The apoptosis rate of A375 and A2058 cells treated with NC for 24 h was detected by flow cytometry. Apoptosis rate of A375 (a) and A2058 cells (b) treated with different concentrations of NC for 24 h was detected by flow cytometry; Statistical analysis of the apoptosis rate of A375 (c) and A2058 cells (d) after NC treatment (* P < 0.05 vs. control, ** P < 0.01 vs. control).
NC Treatment Induced the Apoptosis of Human Melanoma Cells through Mitochondrial Pathway
To further explore the underlying mechanism of the inhibition of NC on human melanoma cells, the expression of mitochondrial apoptosis‑related proteins was detected via western blot analysis and RT-qPCR (Fig. 4). The results revealed that after different concentration of NC treatment, the expression levels of the proapoptotic proteins Bax was obviously increased in A375 and A2058 cells in a dose-dependent manner compared with control group, while antiapoptotic protein Bcl‑2 was significantly decreased. In addition, the expression of Caspase 3 and Caspase 9 distinctly increased at mRNA and protein level, which manifested that the promotion of human melanoma cells apoptosis by NC may be associated with the mitochondrial apoptosis pathway.
Protein and mRNA expression levels of Bax, Bcl-2, Caspase-3 and Caspase-9 in A375 and A2058 cells treated with NC for 24 h. (a) Treated with different concentrations of NC for 24 h were determined by western blotting analysis. Expression level of GAPDH was used as a loading control; Statistical analysis of Bax, Bcl-2, Caspase-3 and Caspase-9 protein expression levels in A375 (b) and A2058 (c) cells after NC treatment; Expression of Bax, Bcl-2, Caspase-3 and Caspase-9 mRNA in A375 (d) and A2058 (e) cells treated with different concentrations of NC for 24 h (* P < 0.05 vs. control, ** P < 0.01 vs. control).
DISCUSSION
NC has been shown to exert antitumor effects in some types of tumors. A study reported that the activation of the Fas pathway resulted in the apoptotic mechanism of NC on ovarian cancer SKOV3 cells. In addition, NC could increase the expression of Fas, FADD, Caspase-8 and Caspase-3, which manifested that the NC-induced apoptosis of cancer cells is closely related to exogenous apoptotic pathways (Chen et al., 2018). Another study revealed that NC can exert mTOR-dependent cell arrest activity in U87 human glioma cells (Liu et al., 2016). Moreover, NC induced apoptosis, cell cycle arrest, and synergistic cytotoxicity with doxorubicin in human breast cancer cell lines MCF-7 and MDA-MB-231 (Sun et al., 2014).
In this study, to detect the inhibitory effect of NC on the proliferation of A375 and A2058 cells, CCK-8 assay was used to assess the effects of different concentrations of NC on the viability of A375 and A2058 cells. As presented in Fig. 1, NC inhibited the growth of melanoma cells in a dose- and time-dependent manner. The anti-proliferative effect of NC on melanoma cells also showed changes in cell morphology. It was observed by inverted microscope that with the increase of NC concentration, NC could significantly inhibit the proliferation of melanoma cells. And cell morphology was shrunk and adherent cells were decreased.
Types of cell death can be divided into two broad categories based on functional differences, regulated cell death (RCD) and accidental cell death (ACD). Based on morphological, biochemical and functional perspectives, the Nomenclature Committee on Cell Death (NCCD) categorized RCD into more than ten forms in 2018. Many human disorders can be treated by targeting RCD, so great effort is being made to create RCD modulators for use as therapeutics in clinical settings (Galluzzi et al., 2018). Among them, targeting apoptosis is the most successful method to treat cancer except for surgery (Zhang et al., 2019). Apoptosis is an active process in which apoptotic cells undergo significant morphological changes including chromatin condensation, fragmented nuclei and cellular shrinkage (Chen et al., 2019). Our study further explored whether NC can cause apoptosis in A375 and A2058 cells. As shown in DAPI staining, we clearly observed the obvious morphological features of apoptosis, and with the increase of NC drug concentration, the bright blue fluorescence observed under inverted fluorescence microscope increased significantly. AnnexinV-FITC/PI staining flow cytometry further confirmed that NC could induce the increase of apoptosis rate of A375 and A2058 cells in a dose-dependent manner.
As we all know, cell apoptosis is triggered by two major types: the mitochondrial pathway (intrinsic pathway) and the death receptors pathway (extrinsic pathway) (Du et al., 2017a). Bcl-2 and Bax, as two pivotal factors, have been proved to be involved in the control of apoptosis through the mitochondrial pathway (Du et al., 2017b). In addition, caspase family are regarded as key regulators to coordinate the execution of apoptosis (Yun et al., 2013). In order to identify the type of NC-induced apoptosis effect, we examined the expression of Bcl-2, Bax and Caspase-3/-9 both at mRNA and protein levels. After treating human melanoma cells with different concentrations of NC for 24 h, NC could increase the expression of pro-apoptotic protein Bax and decrease the expression of anti-apoptotic protein Bcl-2, changing the ratio of pro-apoptotic protein and anti-apoptotic protein. In addition, the mRNA and protein levels of Caspase-3 and Caspase-9 increased, suggesting that NC further activated the downstream apoptosis-inducing Caspase-3/-9. These results manifested that NC can promote the apoptosis of human melanoma cells through mitochondrial pathway. Huang et al. (Huang et al., 2020) confirmed that NC could induce cytoprotective autophagy through AMPK-mTOR pathway. Inhibition of autophagy by 3-MA or by AMPK inhibitor potentiated NC-induced apoptosis. They also verified that NC had little impact on non-malignant human umbilical vein endothelial cells (HUVECs) by MTT assay, even after stimulation for 48 hours at 50 μmol/L. Similarly, the toxic effects of NC on normal human foreskin fibroblast cells Hs-68 were almost negligible (10% inhibitory concentration (IC10) > 20 μg/mL) (Yang et al., 2019b). These results suggested that NC, as a potential anti-tumor agent, can be used in combination with autophagy inhibitors for melanoma therapy.
In conclusion, NC has anti-tumor properties against human melanoma cells through the induction of cell apoptosis. NC can inhibit proliferation and induce apoptosis in human melanoma A375 and A2058 cells through mitochondrial pathway. Therefore, our study adds to the growing body of evidence supporting NC as a potential anti-tumor agent in melanoma. In the future, further studies are necessary to confirm the inhibitory effect of NC on melanoma as well as its effects on normal cells and tissues.
DATA AVAILABILITY
All data generated or analyzed during this study are included in this published article.
REFERENCES
Agha, A. and Tarhini, A.A., Adjuvant therapy for melanoma, Curr. Oncol. Rep., 2017, vol. 19, p. 36. https://doi.org/10.1007/s11912-017-0594-5
Bouquet, J., Rivaud, M., Chevalley, S., Deharo, E., Jullian, V., and Valentin, A., Biological activities of nitidine, a potential anti-malarial lead compound, Malar. J., 2012, vol. 11, p. 67. https://doi.org/10.1186/1475-2875-11-67
Chen, S., Yang, L., and Feng, J., Nitidine chloride inhibits proliferation and induces apoptosis in ovarian cancer cells by activating the Fas signalling pathway, J. Pharm. Pharmacol., 2018, vol. 70, pp. 778–786. https://doi.org/10.1111/jphp.12901
Chen, J., Huang, C., Liu, F., Xu, Z., Li, L., Huang, Z., and Zhang, H., Methylwogonin exerts anticancer effects in A375 human malignant melanoma cells through apoptosis induction, DNA damage, cell invasion inhibition and downregulation of the mTOR/PI3K/Akt signalling pathway, Arch. Med. Sci., 2019, vol. 15, pp. 1056–1064. https://doi.org/10.5114/aoms.2018.73711
Cui, Y., Wu, L., Cao, R., Xu, H., Xia, J., Wang, Z.P., and Ma, J., Antitumor functions and mechanisms of nitidine chloride in human cancers, J. Cancer, 2020, vol. 11, pp. 1250–1256. https://doi.org/10.7150/jca.37890
Davis, L.E., Shalin, S.C., and Tackett, A.J., Current state of melanoma diagnosis and treatment, Cancer Biol. Ther., 2019, vol. 20, pp. 1366–1379. https://doi.org/10.1080/15384047.2019.1640032
Del Poeta, M., Chen, S.F., Von Hoff, D., Dykstra, C.C., Wani, M.C., Manikumar, G., Heitman, J., Wall, M.E., and Perfect, J.R., Comparison of in vitro activities of camptothecin and nitidine derivatives against fungal and cancer cells, Antimicrob. Agents Chemother., 1999, vol. 43, pp. 2862–2868. https://doi.org/10.1128/aac.43.12.2862
Du, C., Guo, Y., Cheng, Y., Han, M., Zhang, W., and Qian, H., Torulene and torularhodin, protects human prostate stromal cells from hydrogen peroxide-induced oxidative stress damage through the regulation of Bcl-2/Bax mediated apoptosis, Free Radical Res., 2017a, vol. 51, pp. 113–123. https://doi.org/10.1080/10715762.2017.1285024
Du, L., Fei, Z., Song, S., and Wei, N., Antitumor activity of Lobaplatin against esophageal squamous cell carcinoma through caspase-dependent apoptosis and increasing the Bax/Bcl-2 ratio, Biomed. Pharmacother., 2017b, vol. 95, pp. 447–452. https://doi.org/10.1016/j.biopha.2017.08.119
Galluzzi, L., Vitale, I., Aaronson, S.A., Abrams, J.M., A-dam, D., Agostinis, P., Alnemri, E.S., Altucci, L., Amelio, I., Andrews, D.W., Annicchiarico-Petruzzelli, M., Antonov, A.V., Arama, E., Baehrecke, E. H., Barlev, N.A., Bazan, N.G., Bernassola, F., Bertrand, M.J.M., Bianchi, K., Blagosklonny, M.V., Blomgren, K., Borner, C., Boya, P., Brenner, C., Campanella, M., Candi, E., Carmona-Gutierrez, D., Cecconi, F., Chan, F.K., Chandel, N.S., Cheng, E.H., Chipuk, J.E., Cidlowski, J.A., Ciechanover, A., Cohen, G.M., Conrad, M., Cubillos-Ruiz, J.R., Czabotar, P.E., D’Angiolella, V., Dawson, T.M., Dawson, V.L., De Laurenzi, V., De Maria, R., Debatin, K.M., DeBerardinis, R.J., Deshmukh, M., Di Daniele, N., Di Virgilio, F., Dixit, V.M., Dixon, S.J., Duckett, C.S., Dynlacht, B.D., El-Deiry, W.S., Elrod, J.W., Fimia, G.M., Fulda, S., García-Sáez, A.J., Garg, A.D., Garrido, C., Gavathiotis, E., Golstein, P., Gottlieb, E., Green, D.R., Greene, L.A., Gronemeyer, H., Gross, A., Hajnoczky, G., Hardwick, J.M., Harris, I.S., Hengartner, M.O., Hetz, C., Ichijo, H., Jäättelä, M., Joseph, B., Jost, P.J., Juin, P.P., Kaiser, W.J., Karin, M., Kaufmann, T., Kepp, O., Kimchi, A., Kitsis, R.N., Klionsky, D.J., Knight, R.A., Kumar, S., Lee, S.W., Lemasters, J.J., Levine, B., Linkermann, A., Lipton, S.A., Lockshin, R.A., López-Otín, C., Lowe, S.W., Luedde, T., Lugli, E., MacFarlane, M., Madeo, F., Malewicz, M., Malorni, W., Manic, G., Marine, J.C., Martin, S.J., Martinou, J.C., Medema, J.P., Mehlen, P., Meier, P., Melino, S., Miao, E.A., Molkentin, J.D., Moll, U.M., Muñoz-Pinedo, C., Nagata, S., Nuñez, G., Oberst, A., Oren, M., Overholtzer, M., Pagano, M., Panaretakis, T., Pasparakis, M., Penninger, J.M., Pereira, D.M., Pervaiz, S., Peter, M.E., Piacentini, M., Pinton, P., Prehn, J.H.M., Puthalakath, H., Rabinovich, G.A., Rehm, M., Rizzuto, R., Rodrigues, C.M.P., Rubinsztein, D.C., Rudel, T., Ryan, K.M., Sayan, E., Scorrano, L., Shao, F., Shi, Y., Silke, J., Simon, H.U., Sistigu, A., Stockwell, B.R., Strasser, A., Szabadkai, G., Tait, S.W.G., Tang, D., Tavernarakis, N., Thorburn, A., Tsujimoto, Y., Turk, B., Vanden Berghe, T., Vandenabeele, P., Vander Heiden, M.G., Villunger, A., Virgin, H.W., Vousden, K.H., Vucic, D., Wagner, E.F., Walczak, H., Wallach, D., Wang, Y., Wells, J. A., Wood, W., Yuan, J., Zakeri, Z., Zhivotovsky, B., Zitvogel, L., Melino, G., and Kroemer, G., Molecular mechanisms of cell death: recommendations of the Nomenclature Committee on Cell Death 2018, Cell Death Differ., 2018, vol. 25, pp. 486–541. https://doi.org/10.1038/s41418-017-0012-4
Huang, X., Hu, M., Li, K., Luo, F., and Zhu, H., Nitidine chloride efficiently induces autophagy and apoptosis in melanoma cells via AMPK-mTOR signaling pathway, Pharmazie, 2020, vol. 75, pp. 440–442. https://doi.org/10.1691/ph.2020.0478
Khan, H., Hadda, T. B., and Touzani, R., Diverse therapeutic potential of nitidine, a comprehensive review, Curr. Drug Metab., 2018, vol. 19, pp. 986–991. https://doi.org/10.2174/1389200219666180628165643
Li, F. and Zhang, W., Role of traditional Chinese medicine and its chemical components in anti-tumor metastasis, J. Cancer Res. Ther., 2014, vol. 10, pp. 20–26. https://doi.org/10.4103/0973-1482.139748
Liu, M., Wang, J., Qi, Q., Huang, B., Chen, A., Li, X., and Wang, J., Nitidine chloride inhibits the malignant behavior of human glioblastoma cells by targeting the PI3K/AKT/mTOR signaling pathway, Oncol. Rep., 2016, vol. 36, pp. 2160–2168. https://doi.org/10.3892/or.2016.4998
Livak, K.J. and Schmittgen, T.D., Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT Method, Methods, 2001, vol. 25, pp. 402–408. https://doi.org/10.1006/meth.2001.1262
Sun, M., Zhang, N., Wang, X., Cai, C., Cun, J., Li, Y., Lv, S., and Yang, Q., Nitidine chloride induces apoptosis, cell cycle arrest, and synergistic cytotoxicity with doxorubicin in breast cancer cells, Tumour Biol., 2014, vol. 35, pp. 10201–10212. https://doi.org/10.1007/s13277-014-2327-9
Ugurel, S., Röhmel, J., Ascierto, P.A., Flaherty, K.T., Grob, J.J., Hauschild, A., Larkin, J., Long, G.V., Lorigan, P., McArthur, G.A., Ribas, A., Robert, C., Schadendorf, D., and Garbe, C., Survival of patients with advanced metastatic melanoma: the impact of novel therapies-update 2017, Eur. J. Cancer, 2017, vol. 83, pp. 247–257. https://doi.org/10.1016/j.ejca.2017.06.028
Yang, N., Yue, R., Ma, J., Li, W., Zhao, Z., Li, H., Shen, Y., Hu, Z., Lv, C., Xu, X., Yang, Y., Dai, X., Liu, X., Yu, Y., and Zhang, W., Nitidine chloride exerts anti-inflammatory action by targeting Topoisomerase I and enhancing IL-10 production, Pharmacol. Res., 2019a, vol. 148, p. 104368. https://doi.org/10.1016/j.phrs.2019.104368
Yang, X.G., Jiang, B.W., Jing, Q.Q., Li, WJ., Tan, L. P., Bao, Y.L., Song, Z.B., Yu, C.L., Liu, L., Liu, Y.C., and Li, Y.X., Nitidine chloride induces S phase cell cycle arrest and mitochondria-dependent apoptosis in HaCaT cells and ameliorates skin lesions in psoriasis-like mouse models, Eur. J. Pharmacol., 2019b, vol. 863, p. 172680. https://doi.org/10.1016/j.ejphar.2019.172680
Yun, N., Kim, C., Cha, H., Park, W.J., Shibayama, H., Park, I.S., and Oh, Y.J., Caspase-3-mediated cleavage of PICOT in apoptosis, Biochem. Biophys. Res. Commun., 2013, vol. 432, pp. 533–538. https://doi.org/10.1016/j.bbrc.2013.02.017
Zhang, P., Zhang, Y., Liu, K., Liu, B., Xu, W., Gao, J., Ding, L., and Tao, L., Ivermectin induces cell cycle arrest and apoptosis of HeLa cells via mitochondrial pathway, Cell Proliferation, 2019, vol. 52, p. e12543. https://doi.org/10.1111/cpr.12543
Funding
This work was supported by the Hebei Province Introduction of Foreign Intelligence Project (S&T Program of Hebei (YZ202301)).
Author information
Authors and Affiliations
Contributions
YL and YY contributed equally to this work. GZ, JZ designed the experiments. YL obtained the experimental data of A375 cells, YY obtained the experimental data of A2058 cells and wrote the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
CONFLICT OF INTEREST
The authors of this work declare that they have no conflicts of interest.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This work does not contain any studies involving human and animal subjects.
Additional information
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access. This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Yexian Li, Yao, Y., Zeng, L. et al. Nitidine Chloride Inhibits Proliferation and Induces Mitochondrial-Mediated Apoptosis of Human Melanoma A375 and A2058 Cells In Vitro. Biol Bull Russ Acad Sci 51, 21–29 (2024). https://doi.org/10.1134/S1062359023700449
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1062359023700449