Abstract
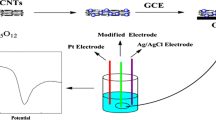
A novel modified electrode, Au nanoparticles/multi-walled carbon nanotubes modified glassy carbon electrode (AuNPs/MWCNT/GCE) was fabricated in this work. In phosphate buffer solution (PBS-KCl), the electrochemical response of Bisphenol A (BPA) at this modified electrode was investigated by differential pulse voltammetry (DPV). A sensitive oxidation peak of BPA was observed at the potential of 0.396 V. Comparing with the bare glassy carbon electrode, the oxidation peak potential shifted to less positive potentials, and the peak current enhanced 10 fold. The factors of the electrochemical response of BPA, such as supporting medium, solution pH, and surfactant were carefully optimized. Under the optimum conditions, a linear calibration curve of the peak current of BPA and concentration was obtained in the range 8.0 × 10−8–1.0 × 10−5 M, and the detection limit was 3 × 10−9 M. The oxidation peak current increases proportionally with scanning speed, which suggests that the electrochemical oxidation of BPA is adsorption-controlled. The recoveries of this method are from 96 to 103.4%, indicating that the proposed method is accurate and reliable.
Similar content being viewed by others
References
Krishnan, A., Stathis, P., Permuth, S., Tokes, L., and Feldman, D., Endocrinology, 1993, vol. 132, p. 2279.
Goodson, A., Robin, H., Summerfield, W., and Cooper, I., Food Addit. Contam., 2004, vol. 21, p. 1015.
Lopez-Cervantes, J. and Paseiro-Losada, P., Food Addit. Contam. 2003, vol. 20, p. 96.
Vom Saal, F.S., Nagel, S.C., Timms, B.G., and Welshons, W.V., Toxicology, 2005, vol. 212, p. 244.
Zhao, R.S., Wang, X., Yuan, J.P., and Zhang, L.L., Microchim. Acta, 2009, vol. 165, p. 443.
Wille, K., De Brabander, H.F., Vanhaecke, L., De Wulf, E., Van Caeter, P., and Janssen, C.R., Trends Anal. Chem., 2012, vol. 35, p. 87.
Zhenkun, Lin, Wenjing, Cheng, Yanyan, Li, Zhiren, Liu, Xiangping, Chen, and Changjiang, Huang, Anal. Chim. Acta, 2012, vol. 720, p. 71.
Wang, F., Yang, J., and Wu, K., Anal. Chim. Acta, 2009, vol. 638, p. 23.
Huanshun, Yin, Yunlei, Zhou, Shiyun, Ai, Ruixia, Han, Tiantian, Tang, and Lusheng, Zhu, Microchim. Acta, 2010, vol. 170, p. 99.
Joseph, Wang and Randhir, P. Deo, Electroanal., 2003, vol. 15, p. 23.
Wu, K.B., Wang, H., Chen, F., and Hu, S.S., Bioelectrochem., 2006. vol. 68, p. 144.
Ozoemena, K.I., Nyokong, T., Zhao, Z., and Nyokong, T., Electrochem. Commun., 2005, vol. 7, p. 679.
Caro, C.A., Bedoui, F., Paez, M.A., Cárdenas-Jirón, G.I., and Zagal, J.H., J. Electrochem. Soc., 2004, vol. 151, p. E32.
Zhang, J., Wang, Y., Lv, R., and Xu, L., Electrochim. Acta, 2010, vol. 55, p. 4039
Maduraiveeran, G. and Ramaraj, R., Electrochem. Commun., 2007, vol. 8, p. 2051.
Khalida, S., Merza, Hadi D., Al-Attabi, Zaid M., Abbas, and Hashim, A. Yusr, Green and Sustainable Chemistry, 2012, vol. 2, p. 26.
Hu, C.G., Chen, Z.L., Shen, A.G., Shen, X.C., Li, J., and Hu, S.S., Carbon, 2006, vol. 44, p. 428.
Yingliang, Wei, Luping, Zhang, Chen, Shao and Chao, Li, Chem. Anal. (Warsaw), 2009, vol. 54, p. 607.
Bard, A.J. and Faulkner, L.R., Electrochemical Methods: Fundamentals and Applications, New York: John Wiley&Sons, 1980, p. 222.
Xueping, Dang, Chengguo, Hu, Yingliang, Wei, Wanhua, Chen, and Shengshui, Hu, Electroanal., 2004, vol. 16, p. 1949.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Wang, A., Wei, Y. & Wang, C. Study on the electrocatalytic oxidation of Bisphenol A on Au nanoparticles/carbon nanotubes composite modified electrode. J Anal Chem 70, 67–71 (2015). https://doi.org/10.1134/S1061934815010049
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1061934815010049