Abstract
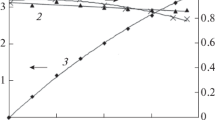
Trends in the development of electrochemical methods for solving environmental problems were considered. Modern tendencies in research on the electrochemical destruction of toxic organic substances resistant to oxidation were analyzed. The use of three-dimensional (3D) electrodes and combined methods (electro-Fenton process combined with ultrasound, UV irradiation, etc.) increases the efficiency of destruction. Special attention was paid to studies at increased pressures. Increased pressure initially created with an inert gas did not substantially affect the kinetics of the anodic process. Increased pressure initially created with an electrochemically active gas (oxygen) accelerated the anodic oxidation of organic compounds (phenol and azo dyes (direct black 2S and stable disperse yellow 4K). The organic substances under study inhibit the cathodic reduction of oxygen, and the positive role of increased pressure is proven.
Similar content being viewed by others
References
Mook, W.T., Chakrabarti, M.H., Aroua, M.K., Khan, G.M.A., Ali, B.S., Islam, M.S., and Abu Hassan, M.A., Desalination, 2012, vol. 285, p. 1.
Zhelovitskaya, A.V., Ermolaeva, E.A., and Dresvyannikov, A.F., Vestn. Kazan. Tekhnol. Univ., 2008, no. 6, p. 211.
Sires, I. and Brillas, E., Environ. Int., 2012, vol. 40, p. 212.
Kosheleva, A.M., Chaenko, N.V., Kornienko, G.V., Vlasenko, V.I., and Kornienko, V.L., Russ. J. Electrochem., 2013, vol. 49, p. 105.
Solozhenko, E.G., Soboleva, N.M., and Goncharuk, V.V., Khim. Tekhnol. Vody, 2004, vol. 26, no. 3, p. 219.
Prabir, G., Nath, S.A., and Subhabrata, R., Desalination, 2011, vol. 266, p. 213.
Garcha, O., Isarain-Chavez, E., El-Ghenymy, A., Brillas, E., and Peralta-Hernandez, J.M., J. Electroanal. Chem., 2014, vol. 728, p. 1.
Almeida, L.C., Silva, B.F., and Zanoni, M.V., J. Electroanal. Chem., 2014, vol. 734, p. 43.
Brillas, E., Sauleda, R., and Casado, J., J. Electrochem. Soc., 1999, vol. 146, p. 4539.
Kornienko, V.L., Kolyagin, G.A., Kornienko, G.V., Chaenko, N.V., Kosheleva, A.M., Kenova, T.A., and Vasil’eva, I.S., Zh. Prikl. Khim., 2014, vol. 87, p. 3.
Yan Xiao-ru, Wang Jian-zhong, Zhang Ping, Xi Jin-e, and Zhang Wei, Water Wastewater, 2010, vol. 41, no. 6, p. 41.
Brillas, E., Sauleda, R., and Casado, J., J. Electrochem. Soc., 1998, vol. 145, p. 759.
Jun-jie Lin, Xiao-song Zhao, Dan Liu, Zhi guo, Ying Zhang, and Hui Xu, J. Hazard. Mater., 2008, vol. 157, p. 541.
Barros, W., Franko, P., Steter, J., Robson, S., Rocha, R., Lanza, M., J. Electroanal. Chem., 2014, vols. 722–723, p. 46.
Zhu, X. P., Ni, J.R., Xing, X., Li, H.N., and Jiang, Y., Electrochim. Acta, 2011, vol. 56, p. 1270.
Chen, Y., Shi, W., Han, W.Q., Sun, X.Y., Li, J.S., and Wang, L.J., Electrochim. Acta, 2011, vol. 58, p. 383.
Lei Ma, Senlin, Synn., Zsyan, Ren., Xyanjao Bey, and Peicsyan Lu., Russ. J. Electrochem., 2014, vol. 50, p. 569.
Yi, F., Chen, S., and Yuan, Ch., J. Hazard. Mater., 2008, vol. 157, p. 79.
Gasanova, F.G. and Aliev, Z.M., Ekol. Prom-st, 2009, p. 2.
Serikava, R.M., Isaka, M., Su, Q., Usui, T., Nishimura, T., Sato, H., and Hamada, S., J. Appl. Electrochem., 2000, vol. 30, p. 875.
Epron, F., Chollier-Brym, M.J., Lamy-Pitara, E., and Barbier, J., Appl. Catalysis, 1999, vol. 176, p. 221.
Zhou, M., Lei, L., and Dai, O.A., Chem. Commun., 2007, p. 853.
Alverez-Gallegos, A. and Pletcher, D., Electrochim. Acta, 1998, vol. 44, p. 2483.
Orudshev, Ph.Ph., Aliev, Z.M., Gasanova, Ph.G., Isaev, A.B., and Chabanov, N.C., Russ. J. Electrochem., 2015, vol. 51, p. 1108.
Aliev, Z.M., Doctoral (Engineering) Dissertation, Novocherkassk: NPI, 2001.
Tarasevich, M.R., Shepelev, V.Ya., and Burshteyn, R., Elektrokhimiya, 1973, vol. 9, p. 1693.
Gordon, A. and Ford, R., The Chemist’s Companion, New York: Wiley, 1972.
Mairanovskii, S.G., Stradyn’, Ya.P., and Bezuglyi, V.D., Polyarografiya v organicheskoi khimii (Polarography in Organic Chemistry), Moscow: Khimiya, 1975.
Kharlamova, T.A. and Tedoradze, G.A., Russ. Chem. Rev., 1987, vol. 56, p. 29.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © T.A. Kharlamova, Z.M. Aliev, 2016, published in Elektrokhimiya, 2016, Vol. 52, No. 3, pp. 291–300.
Presented at the 18th All-Russian Conference on Organic Electrochemistry, Tambov, September 15–19, 2014.
Rights and permissions
About this article
Cite this article
Kharlamova, T.A., Aliev, Z.M. Use of electrolysis under pressure for destructive oxidation of phenol and azo dyes. Russ J Electrochem 52, 251–259 (2016). https://doi.org/10.1134/S102319351603006X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S102319351603006X