Abstract
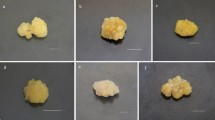
In vitro callus cultures of common chicory (Cichorium intybus L.) were obtained and their growth and biochemical characteristics depending on the hormonal composition of the MS medium and the spectral composition of light were studied. The study of the effect of light culture on callus tissue formation and inulin accumulation in it was carried out in opaque grow tents with radiation aligned with the flux density of photosynthetic photons and different ratios of its levels in the region of 660 nm (R, red) and 730 nm (FR, far red). The control variant was placed under white linear fluorescent lamps. The resulting cultures were characterized by high proliferative activity and the capability for morphogenesis. It has been established that the interaction of two factors—the presence of auxins in the nutrient medium (IAA or NAA at a concentration of 7.5 mg/L in combination with BAP 0.5 mg/L) and cultivation under light culture conditions (FR > R, FR = R, FR < R)—had a significant impact on the biosynthetic potential of cell cultures. In the obtained cultures, a study of the quantitative content of inulin was carried out. It has been shown that the high content of inulin (7.55–7.95%) in callus cultures was on the MS medium in combination with IAA at FR > R illumination. This is probably due to the fact that well proliferating and highly morphogenic callus tissue was formed under these conditions. The obtained results confirm the hypothesis about the specificity of cultured cells to in vitro synthesize and accumulate secondary metabolites in dedifferentiated cells and the dependence of this process on factors of chemical and physical nature.





Similar content being viewed by others
REFERENCES
McClelland, J.W., Allen, J.C., and Zakir, S., Bio-medicinal effect of sweet potato in people with diabetes, J. Am. Diet. Assoc., 2007, vol. 8, p. A104. https://doi.org/10.1016/j.jada.2007.05.396
Ikanone, C.E.O. and Oyekan, P.O., Effect of boiling and frying on the total carbohydrate, vitamin C and mineral contents of Irish (Solanun tuberosum) and Sweet (Ipomea batatas) potato tubers, Niger. Food J., 2014, vol. 32, p. 33. https://doi.org/10.1016/S0189-7241(15)30115-6
Mohammad, K.A., A comprehensive review of sweet potato (Ipomoea batatas Lam): revisiting the associated health benefits, Trends Food Sci. Technol., 2021, vol. 115, p. 512. https://doi.org/10.1016/j.tifs.2021.07.001
World Health Organization, WHO traditional medicine strategy: 2014-2023, World Health Organization, 2013.
Tutelyan, V.A., Sukhanov, B.P., Kochetkova, A.A., Sheveleva, S.A., and Smirnova, E.A., Russian regulations on nutraceuticals, functional foods, and foods for special dietary uses, in Nutraceutical and Functional Food Regulations in the United States and around the World, Bagchi., D., Ed., Academic Press, 2019, p. 399. https://doi.org/10.1016/B978-0-12-816467-9.00026-5
Roberfroid, M., Inulin-type fructans: functional food ingredients, J. Nutr., 2007, vol. 137, p. 2493S-2502S. https://doi.org/10.1201/9780203504932
Barclay, T., Ginic-Markovic, M., Cooper, P., and Petrovsky, N., Inulin—a versatile polysaccharide with multiple pharmaceutical and food chemical uses, J. Excip. Food Chem., 2016, vol. 1, p. 1132.
Kathy, R.N., Inulin and oligofructose: what are they?, J. Nutr., 1999, vol. 129, p. 1402S-1406S. https://doi.org/10.1093/jn/129.7.1402S
Kalyani, N.K., Kharb, S., and Thompkinson, D.K., Inulin dietary fiber with functional and health attributes—a review, Food Rev. Int., 2010, vol. 26, p. 189. https://doi.org/10.1080/87559121003590664
Boeckner, L.S., Marilynn, I.S., and Bryan, C.T., Inulin: a review of nutritional and health implications, Adv. Food Nutr. Res., 2001, vol. 43, p. 1. https://doi.org/10.1016/S1043-4526(01)43002-6
Bais, H.P. and Ravishankar, G.A., Cichorium intybus L. cultivation, processing, utility, value addition and biotechnology, with an emphasis on current status and future prospects, J. Sci. Food Agric., 2001, vol. 81, p. 467. https://doi.org/10.1002/jsfa.817
Abbas, Z.K., Saggu, S., Sakeran, M.I., Zidan, N., Rehman, H., and Ansari, A.A., Phytochemical, antioxidant and mineral composition of hydroalcoholic extract of chicory (Cichorium intybus L.) leaves, Saudi J. Biol. Sci., 2015, vol. 22, p. 322. https://doi.org/10.1016/j.sjbs.2014.11.015
Molan, A.L., Duncan, A. J., Barry, T.N., and McNabb, W.C., Effect of condensed tannins and sesquiterpene lactones extracted from chicory on the motility of larvae of deer lungworm and gastrointestinal nematodes, Parasitol. Int., 2003, vol. 52, p. 209. https://doi.org/10.1016/S1383-5769(03)00011-4
Nandagopal, S. and Ranjitha, B.D., Phytochemical and antibacterial studies of chicory (Cichorium intybus L.)—a multipurpose medicinal plant, Adv. Biol. Res., 2007, vol. 1, p. 17. https://doi.org/10.1016/S1383-5769(03)00011-4
Muthusamy, V.S., Anand, S., Sangeetha, K.N., Sujatha, S., Arun, B., and Lakshmi, B.S., Tannins present in Cichorium intybus enhance glucose uptake and inhibit adipogenesis in 3T3-L1 adipocytes through PTP1B inhibition, Chem.-Biol. Interact., 2008, vol. 174 (1), p. 69. https://doi.org/10.1016/j.cbi.2008.04.016
Atta, A.H., Elkoly, T.A., Mouneir, S M., Kamel, G., Alwabel, N.A., and Zaher, S., Hepatoprotective effect of methanolic extracts of Zingiber officinale and Cichorium intybus, Indian J. Pharm. Sci., 2010, vol. 72, p. 564. https://doi.org/10.4103/0250-474X.78521
Meehye, K. and Shin, H.K., The water-soluble extract of chicory reduces glucose uptake from the perfused jejunum in rats, J. Nutr., 1996, vol. 126, p. 2236. https://doi.org/10.1093/jn/126.9.2236
Afzal, S., Afza, N., Awan, M.R., Khan, T.S., Gilani, A., Khanum, R., and Tariq, S., Ethno-botanical studies from Northern Pakistan, J. Ayub. Med. Coll. Abbotabad., 2009, vol. 21, p. 52.
Abbasi, A.M., Khan, M.A., Ahmad, M., Zafar, M., Khan, H., Muhammad, N., and Sultana, S., Medicinal plants used for the treatment of jaundice and hepatitis based on socio-economic documentation, Afr. J. Biotechnol., 2009, vol. 8, p. 1643.
Jamshidzadeha, A., Khoshnood, M.J., Dehghani, Z., and Niknahad, H., Hepatoprotective activity of Cichorium intybus L. leaves extract against carbon tetrachloride induced toxicity, Iran. J. Pharm. Res., 2006, vol. 1, p. 41. https://doi.org/10.22037/ijpr.2010.651
Hassan, H.A., The prophylactic role of some edible wild plants against nitrosamine precursor’s experimentally-induced testicular toxicity in male albino rats, J. Egypt. Soc. Toxicol., 2008, vol. 38, p. 1.
Nayeemunnisa, A., Alloxan diabetes-induced oxidative stress and impairment of oxidative defense system in rat brain: neuroprotective effects of Cichorium intybus L., Int. J. Diabetes Metabol., 2009, vol. 17, p. 105. https://doi.org/10.1159/000497681
Mulabagal, V., Wang, H., Ngouajio, M., and Nair, M.G., Characterization and quantification of health beneficial anthocyanins in leaf chicory (Cichorium intybus) varieties, Eur. Food Res. Technol., 2009, vol. 230, p. 47. https://doi.org/10.1007/s00217-009-1144-7
Hassan, H.A. and Yousef, M.I., Ameliorating effect of chicory (Cichorium intybus L.)-supplemented diet against nitrosamine precursors-induced liver injury and oxidative stress in male rats, Food Chem. Toxicol., 2010, vol. 48, p. 2163. https://doi.org/10.1016/j.fct.2010.05.023
Taylor, R.L., Weeds of Roadsides and Waste Ground in New Zealand, Christchurch, New Zealand: Caxton Press, 1981, p. 177.
Parsons, J.L., Cameron, S.I., Harris, C.S., and Smith, M.L., Echinacea biotechnology: advances, commercialization and future considerations, Pharm. Biol., 2018, vol. 56, p. 485. https://doi.org/10.1080/13880209.2018.1501583
Toponi, M., Action combining kinetin and acid Indole acetic on the neoformation of organs by fragments of leaves of endive (Cichorium intybus L) grown in vitro, C.R. Acad. Sci. Paris, 1963, vol. 257, p. 3030.
Park, E. and Lim, H., Establishment of an efficient in vitro plant regeneration system in Chicory (Cichorium intybus L), Acta Hortic., 1999, vol. 483, p. 367. https://doi.org/10.17660/ActaHortic.1999.483.42
Velayutham, P., Kumari, B.D., and Baskaran, P., An efficient in vitro plant regeneration system for Cichorium intybus L.—an important medicinal plant, J. Agric. Technol., 2006, vol. 2, p. 287.
Rehman, R.U., Israr, M., Srivastava, P.S., Bansal, K.C., and Abdin, M.Z., In vitro regeneration of witloof chicory (Cichorium intybus L.) from leaf explants and accumulation of esculin, In Vitro Cell Dev. Biol., 2003, vol. 39, p. 142. https://doi.org/10.1079/IVP2002381
Yucesan, B., Turker, A.U., and Gurel, E., TDZ-induced high frequency plant regeneration through multiple shoot formation in witloof chicory (Cichorium intybus L.), Plant Cell Tissue Organ Cult., 2007, vol. 91, p. 243. https://doi.org/10.1007/s11240-007-9290-8
Ohadi Rafsanjani, S.M., Alvari, A., Mohammad, A.Z, Abdin, M.A., and Hejazi, M., In vitro propagation of Cichorium intybus L. and quantification of enhanced secondary metabolite (esculin), Recent Pat. Biotechnol., 2011, vol. 5, p. 227. https://doi.org/10.2174/187220811797579123
Dakshayini, K., Rao, C.V., Karun, A., Bhavyashree, U., and Ujwal, P., High-frequency plant regeneration and histological analysis of callus in Cichorium intybus: an important medicinal plant, J. Phytol., 2016, vol. 8, p. 7. https://doi.org/10.19071/jp.2016.v8.2980
Wagner, G.M. and Eneva, T., Positive effect of cefotaxime on plant regeneration from Cichorium intybus L. leaf material, Landbauforschung Voelkenrode, 1996, vol. 46, p. 166.
Cadalen, T., Morchen, M., and Blassiau, C., Development of SSR markers and construction of a consensus genetic map for chicory (Cichorium intybus L), Mol. Breed., 2010, vol. 25, p. 699. https://doi.org/10.1007/s11032-009-9369-5
Shulgina, A.A., Kalashnikova, E.A., Tarakanov, I.G., Kirakosyan, R.N., Cherednichenko, M.Y., Polivanova, O.B., Baranova, E.N., and Khaliluev, M.R., Influence of light conditions and medium composition on morphophysiological characteristics of Stevia rebaudiana Bertoni in vitro and in vivo, Horticulturae, 2021, vol. 7, p. 195. https://doi.org/10.3390/horticulturae7070195
Murashige, T. and Skoog, F., A revised medium for rapid growth and bioassays with tobacco tissue cultures, Physiol. Plant., 1962, vol. 15, p. 473. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Kas'yan, I.G. and Kas’yan, A.K., Optimization of the spectrophotometric method for the determination of inulin in Jerusalem artichoke (Helianthus tuberosus L.) tubers, Sed’maya Mezhdunarodnaya nauchno-prakticheskaya konferentsiya Lekarstvennoe rastenievodstvo: ot opyta proshlogo k sovremennym tekhnologiyam (Seventh International Scientific and Practical Conference Medicinal Plant Growing: From Past Experience to Modern Technologies), Kishinev, 2019, p. 121.
Rnjitha Kumari, B.D., Velautham, P., and Anitha, S., A comparative study on inulin and esculin content of in vitro and in vivo plants of chicory (Cichorium intybus L. Cv. Lucknow local), Adv. Biol. Res., 2007, vol. 1, p. 22.
Velayutham, P. and Ranjitha Kumari, B.D., Influence of photoperiod on in vitro flowering in Cichorium intybus L., Indian J. Plant Physiol., 2003, vol. 218, p. 90.
Funding
The work was carried out within the framework of the thematic plan-task for the implementation of research works of the Federal State Budgetary Educational Institution of Higher Education Russian State Agrarian University–Timiryazev Moscow Agricultural Academy commissioned by the Ministry of Agriculture of Russia at the expense of the federal budget in 2023.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
COMPLIANCE WITH ETHICAL STANDARDS
This work does not contain any studies involving humans and animals as research subjects.
CONFLICT OF INTEREST
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Kalashnikova, E.A., Kirakosyan, R.N., Trukhachev, V.I. et al. Features of Growth and Inulin Content in Callus Cultures Cichorium intybus L. In Vitro. Russ J Plant Physiol 70, 78 (2023). https://doi.org/10.1134/S1021443723600320
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1134/S1021443723600320