Abstract
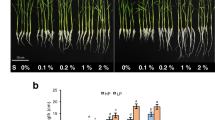
It is well-recognized that plant-derived smoke and karrikinolide (KAR1) are capable of promoting seed germination and seedling growth in many plants. In addition, recent findings have suggested that smoke and KAR1 can alleviate the deleterious effects of unfavorable environmental conditions on plant growth. In this study, we aimed to determine the effects of smoke-water and KAR1 on growth and physiological parameters in wheat (Triticum aestivum L.) seedlings subjected to boron (B) stress. To accomplish this goal, 7-day-old seedlings were grown in nutrient solutions containing 12 mM boric acid with or without the presence of smoke-water (0.4 and 1%) and KAR1 (0.1 µM) for 5 days. Hydrogen peroxide, malondialdehyde, proline, total phenolic contents, antioxidant enzyme activities, and B concentration were determined for each treatment group. The results of the present study show that smoke-water and KAR1 improve root growth and decrease B accumulation in wheat seedlings under B stress. Moreover, KAR1 and smoke-water (0.4%), albeit not significant, led to a slight reduction in B-triggered oxidative injury. Our findings also suggest that the increased activities of glutathione reductase, peroxidase, and superoxide dismutase in B-treated seedlings return almost to control levels in the presence of KAR1 and smoke-water (0.4%). In conclusion, this study provides evidence that smoke and KAR1 have the potential to be used in agriculture in order to reduce the negative effects of excess B on plant growth.



Similar content being viewed by others
REFERENCES
Reid, R., Boron toxicity and tolerance in crop plants, in Crop Improvement Under Adverse Conditions, Tuteja, N. and Gill, S., Eds., New York: Springer-Verlag, 2013, p. 333.
Nable, R.O., Bañuelos, G.S., and Paull, J.G., Boron toxicity, Plant Soil, 1997, vol. 193, p. 181.
Reid, R., Update on boron toxicity and tolerance in plants, in Advances in Plant and Animal Boron Nutrition, Xu, F., Goldbach, H., Brown, P., Bell, R., Fujiwara, T., Hunt, C., Goldberg, S. and Shi, L., Eds., Dordrecht: Springer-Verlag, 2007, p. 83.
Çatav, Ş.S., Genç, T.O., Oktay, M.K., and Küçükakyüz, K., Effect of boron toxicity on oxidative stress and genotoxicity in wheat (Triticum aestivum L.), Bull. Environ. Contam. Toxicol., 2018, vol. 100, p. 502.
Landi, M., Margaritopoulou, T., Papadakis, I.E., and Araniti, F., Boron toxicity in higher plants: an update, Planta, 2019, vol. 250, p. 1011.
Apel, K. and Hirt, H., Reactive oxygen species: metabolism, oxidative stress, and signal transduction, Annu. Rev. Plant Biol., 2004, vol. 55, p. 373.
Gill, S.S. and Tuteja, N., Reactive oxygen species and antioxidant machinery in abiotic stress tolerance in crop plants, Plant Physiol. Biochem., 2010, vol. 48, p. 909.
Landi, M., Degl’Innocenti, E., Pardossi, A., and Guidi, L., Antioxidant and photosynthetic responses in plants under boron toxicity: a review, Am. J. Agric. Biol. Sci., 2012, vol. 7, p. 255.
Sharma, P., Jha, A.B., Dubey, R.S., and Pessarakli, M., Reactive oxygen species, oxidative damage, and antioxidative defense mechanism in plants under stressful conditions, J. Bot., 2012, vol. 2012, p. 1.
De Lange, J.H. and Boucher, C., Autecological studies on Audouinia capitata (Bruniaceae). I. Plant-derived smoke as a seed germination cue, S. Afr. J. Bot., 1990, vol. 56, p. 700.
Çatav, Ş.S., Küçükakyüz, K., Tavşanoğlu, Ç., and Pausas, J.G., Effect of fire-derived chemicals on germination and seedling growth in Mediterranean plant species, Basic Appl. Ecol., 2018, vol. 30, p. 65.
Ghebrehiwot, H.M., Kulkarni, M.G., Kirkman, K.P., and Van Staden, J., Smoke-water and a smoke-isolated butenolide improve germination and seedling vigor of Eragrostis tef (Zucc.) Trotter under high temperature and low osmotic potential, J. Agron. Crop Sci., 2008, vol. 194, p. 270.
Jamil, M., Kanwal, M., Aslam, M.M., Khan, S.U., Malook, I., Tu, J., and Rehman, S.U., Effect of plant-derived smoke priming on physiological and biochemical characteristics of rice under salt stress condition, Aust. J. Crop Sci., 2014, vol. 8, p. 159.
Khan, P., Rehman, S.U., Jamil, M., Irfan, S., Waheed, M.A., Aslam, M.M., Kanwal, M., and Shakir SK., Alleviation of boron stress through plant derived smoke extracts in Sorghum bicolor, J. Stress Physiol. Biochem., 2014, vol. 10, p. 153.
Akhtar, N., Khan, S., Malook, I., Rehman, S.U., and Jamil, M., Pb-induced changes in roots of two cultivated rice cultivars grown in lead-contaminated soil mediated by smoke, Environ. Sci. Pollut. Res., 2017, vol. 24, p. 21298.
Velikova, V., Yordanov, I., and Edreva, A., Oxidative stress and some antioxidant systems in acid rain-treated bean plants: protective role of exogenous polyamines, Plant Sci., 2000, vol. 151, p. 59.
Shabnam, N., Tripathi, I., Sharmila, P., and Pardha-Saradhi, P., A rapid, ideal, and eco-friendlier protocol for quantifying proline, Protoplasma, 2016, vol. 253, p. 1577.
Maksimović, J.J.D. and Živanović, B.D., Quantification of the antioxidant activity in salt-stressed tissues, in Plant Salt Tolerance, Shambhala, S. and Cuin, T.A., Ed., New York: Springer-Verlag, 2012, p. 237.
Nakano, Y. and Asada, K., Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts, Plant Cell Physiol., 1981, vol. 22, p. 867.
Aebi, H., Catalase in vitro, Methods Enzymol., 1984, vol. 105, p. 121.
Foyer, C.H. and Halliwell, B., The presence of glutathione and glutathione reductase in chloroplasts: a proposed role in ascorbic acid metabolism, Planta, 1976, vol. 133, p. 21.
Chance, B. and Maehly, A.C., Assay of catalases and peroxidases, Methods Enzymol., 1955, vol. 2, p. 764.
Beauchamp, C. and Fridovich, I., Superoxide dismutase: improved assays and an assay applicable to acrylamide gels, Anal. Biochem., 1971, vol. 44, p. 276.
Surgun, Y., Çöl, B., and Bürün, B., 24-Epibrassinolide ameliorates the effects of boron toxicity on Arabidopsis thaliana (L.) Heynh by activating an antioxidant system and decreasing boron accumulation, Acta Physiol. Plant., 2016, vol. 38, p. 71.
Baldwin, I.T., Staszak-Kozinski, L., and Davidson, R., Up in smoke: I. Smoke-derived germination cues for postfire annual, Nicotiana attenuata Torr. ex Watson, J. Chem. Ecol., 1994, vol. 20, p. 2345.
Burger, B.V., Pošta, M., Light, M.E., Kulkarni, M.G., Viviers, M.Z., and van Staden, J., More butenolides from plant-derived smoke with germination inhibitory activity against karrikinolide, S. Afr. J. Bot., 2018, vol. 115, p. 256.
Hu, H. and Brown, P.H., Absorption of boron by plant roots, Plant Soil, 1997, vol. 193, p. 49.
Miwa, K. and Fujiwara, T., Boron transport in plants: co-ordinated regulation of transporters, Ann. Bot., 2010, vol. 105, p. 1103.
Aftab, T., Khan, M.M.A., Idrees, M., Naeem, M., Moinuddin, and Hashmi, N., Methyl jasmonate counteracts boron toxicity by preventing oxidative stress and regulating antioxidant enzyme activities and artemisinin biosynthesis in Artemisia annua L, Protoplasma, 2011, vol. 248, p. 601.
Saruhan, N., Saglam, A., and Kadioglu A., Salicylic acid pretreatment induces drought tolerance and delays leaf rolling by inducing antioxidant systems in maize genotypes, Acta Physiol. Plant., 2012, vol. 34, p. 97.
Funding
This work is a part of the Ph.D. thesis of the second author, funded by the Scientific Research Projects Coordination Unit of Muğla Sıtkı Koçman University (project nos. 15/153 and 16/106).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interests. The authors declare that they have no conflicts of interest.
Statement on the welfare of humans or animals. This article does not contain any studies involving animals performed by any of the authors.
Additional information
Abbreviations: APX—ascorbate peroxidase; CAT—catalase; GR—glutathione reductase; KAR1—karrikinolide; MDA— malondialdehyde; NBT—nitro-blue tetrazolium; POD—peroxidase; ROS—reactive oxygen species; SOD—superoxide dismutase.
Rights and permissions
About this article
Cite this article
Küçükakyüz, K., Çatav, Ş.S. Physiological Effects of Smoke-water and Karrikinolide on Wheat Seedlings Grown under Boron Stress. Russ J Plant Physiol 68, 552–558 (2021). https://doi.org/10.1134/S1021443721030092
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1021443721030092