Abstract
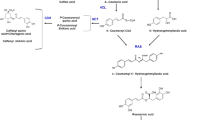
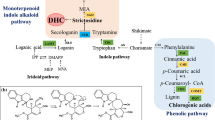
In different plant species, secondary metabolite biosynthesis is regulated by the phytohormone jasmonic acid (JA), which is derived by the action of lipoxygenase. In this study, we examined mono- and sesquiterpenoid accumulation and the related signal transduction pathways and biosynthetic genes in adventitious root cultures of Panax ginseng C.A. Meyer as induced by yeast extract (YE, 3 g/L), a biotic elicitor, and salicylic acid (SA, 200 μM), a signaling elicitor. The lipoxygenase (LOX) gene was highly expressed in 24 and 12 h after treatment with SA and YE. JA content was significantly increased in 24 h after SA treatment. The H2O2 content was the highest in 24 and 72 h after the onset of SA and YE treatment, respectively. RNA blot analysis showed that farnesyl diphosphate synthase (FPS) and isopentenyl pyrophosphate isomerase (IPPI) genes encoding enzymes of the biosynthesis of mono- and sesquiterpenoids were up-regulated by both elicitors. Farensol, isochiapin B sesquiterpenoids, champhor, and cineole monoterpenoids were highly accumulated after 24 h of SA treatment, while YE treatment induced bacchotricuneatin C, guaiazulene, isochiapin B, and p-benzoquinone sesquiterpenoid production. These results suggest that mono- and sesquiterpenoid accumulation induced by SA and YE occurs due to the IPPI and FPS expression and may be mediated by reactive oxygen species signaling and jasmonic acid signal transduction.
Similar content being viewed by others
Abbreviations
- DW:
-
dry weight
- FPS:
-
farnesyl diphosphate synthase
- FW:
-
fresh weight
- IPPI:
-
isopentenyl pyrophosphate isomerase
- JA:
-
jasmonic acid
- LOX:
-
lipoxygenase
- SA:
-
salicylic acid
- YE:
-
yeast extract
References
Montesano, M., Brader, G., and Palva, E.T., Pathogen-derived elicitors: searching for receptors in plants, Mol. Plant Pathol., 2003, vol. 4, pp. 73–79.
Vasconsuelo, A. and Boland, R., Molecular aspects of the early stages of elicitation of secondary metabolites in plants, Plant Sci., 2007, vol. 172, pp. 861–875.
Zhao, J., Lawrence, C., Davis, T., and Verpoorte, R., Elicitor signal transduction leading to production of plant secondary metabolites, Biotechnol. Adv., 2005, vol. 23, pp. 283–333.
Cragg, G.M., Paclitaxel (taxol): a success story with valuable lessons for natural product drug discovery and development, Med. Res. Rev., 1998, vol. 18, pp. 315–331.
Dhingra, V., Rao, K.V., and Narasu, M.L., Current status of artemisinin and its derivatives as antimalarial drugs, Life Sci., 2000, vol. 66, pp. 279–300.
Lichtenthaler, H.K., Schwender, J., Disch, A., and Rohmer, M., Biosynthesis of isoprenoids in higher plant chloroplasts proceeds via a mevalonate-independent pathway, FEBS Lett., 1997, vol. 400, pp. 271–274.
Murashige, T. and Skoog, F., A revised medium for rapid growth and bioassay with tobacco tissue cultures, Plant Physiol., 1962, vol. 15, pp. 473–499.
Liu, X., Yang, Y.L., Lin, W.H., Tong, J.H., Huang, Z.G., and Xiao, L.T., Determination of both jasmonic acid and methyl jasmonate in plant samples by liquid chromatography tandem mass spectrometry, Chinese Sci. Bull., 2010, vol. 55, pp. 2231–2235.
Brennan, T. and Frenkel, C., Involvement of hydrogen peroxide in regulation of senescence in pear, Plant Physiol., 1977, vol. 59, pp. 411–416.
Lee, S. and Chappell, J., Biochemical and genomic characterization of terpene synthases in Magnolia grandiflora, Plant Physiol., 2008, vol. 147, pp. 1017–1033.
Gundlach, H., Muller, M.J., Kutchan, T.M., and Zenk, M.H., Jasmonic acid is a signal transducer in elicitor-induced plant cell cultures, Proc. Natl. Acad. Sci. USA, 1992, vol. 89, pp. 2389–2393.
Khalili, M., Hasanloo, T., Kazemi, Tabar, S.K., and Rahnama, H., Influence of exogenous salicylic acid on flavonolignans and lipoxygenase activity in the hairy root cultures of Silybum marianum, Cell Biol. Int., 2009, vol. 33, pp. 988–994.
Hasanloo, T., Sepehrifar, R., Rahnama, H., and Shams, M., Evaluation of the yeast-extract signaling pathway leading to silymarin biosynthesis in milk thistle hairy root culture, J. Microbiol. Biotechnol., 2009, vol. 25, pp. 1901–1909.
Degousee, N., Triantaphylides, C., and Montillet, J.L., Involvement of oxidative processes in the signaling mechanisms leading to the activation of glyceollin synthesis in soybean (Glycine max), Plant Physiol., 1994, vol. 104, pp. 945–952.
Matsuda, F., Miyagawa, H., and Ueno, T., Involvement of reactive oxygen species in the induction of (S)-N-p-coumaroyloctopamine accumulation by β-1,3-glucooligosaccharide elicitors in potato tuber tissues, J. BioSci., 2001, vol. 56, pp. 228–234.
Zhao, J., Hu, Q., Guo, Y.Q., and Zhu, W.H., Elicitorinduced indole alkaloid biosynthesis in Catharanthus roseus cell cultures is related to Ca2+-influx and the oxidative burst, Plant Sci., 2001, vol. 161, pp. 423–431.
Kravchuk, Zh.M., Perkovs’ka, H.Yu., and Dmytriiev, O.P., Role of active forms of oxygen in the induction of phytoalexin synthesis in Allium cepa cells, Tsitol. Genet., 2003, vol. 37, pp. 30–35.
Hu, X., Neill, S.J., Cai, W., and Tang, Z., Hydrogen peroxide and jasmonic acid mediate oligogalacturonic acid-induced saponin synthesis in suspension-cultured cells of Panax ginseng, Physiol. Plant., 2003, vol. 118, pp. 414–421.
Zhao, J. and Sakai, K., Peroxidases are involved in the biosynthesis and biodegradation of β-thujaplicin in fungal elicitor-treated Cupressus lusitanica suspension cultures, New Phytol., 2003, vol. 159, pp. 719–731.
Wibberley, M.S., Lenton, J.R., and Neill, S.J., Sesquiterpenoid phytoalexins produced by hairy roots of Nicotiana tabacum, Phytochemistry, 1994, vol. 37, pp. 349–351.
Singh, G., Gavriel, J., Oakey, J.S., and Curtis, W.R., Interaction of methyl jasmonate, wounding and fungal elicitation during sequiterpene induction in Hyoscyamus muticus in root cultures, Plant Cell Rep., 1998, vol. 17, pp. 391–395.
Khorolragchaa, A., Parvin, S., Shim, J.S., Kim, Y.J., Lee, O.R., In, J.G., Kim, Y.J., Kim, S.Y., and Yang, D.C., Isolation of sesquiterpene synthase homolog from Panax ginseng C.A. Meyer, J. Ginseng Res., 2010, vol. 34, pp. 17–22.
Kittipongpatana, N., Davis, D.L., and Porter, J.R., Methyl jasmonate increases the production of valepotriates by transformed root cultures of Valerianella locusta, Plant Cell Tissue Organ Cult., 2002, vol. 71, pp. 65–75.
Wielanek, M. and Urbanek, H.K., Enhanced glucotropaeolin production in hairy root cultures of Tropaeolum majus L. by combining elicitation and precursor feeding, Plant Cell Tissue Organ Cult., 2006, vol. 86, pp. 177–186.
Mehmetoglu, U. and Curtis, W.R., Effects of abiotic inducers on sesquiterpene synthesis in hairy root and cell-suspension cultures of Hyoscyamus muticus, Appl. Biochem. Biotechnol., 1997, vol. 67, pp. 71–77.
Chen, H. and Chen, F., Effect of yeast elicitor on the secondary metabolism of Ti-transformed Salvia miltiorrhiza cell suspension cultures, Plant Cell Rep., 2000, vol. 19, pp. 710–717.
Chen, D.H., Ye, H.C., and Li, G.F., Expression of a chimeric farnesyl diphosphate synthase gene in Artemisia annua L. transgenic plants via Agrobacterium tumefaciens-mediated transformation, Plant Sci., 2000, vol. 155, pp. 179–185.
Taguchi, G., Yazawa, T., Hayashida, N., and Okazaki, M., Molecular cloning and heterologous expression of novel glucosyltransferases from tobacco cultured cells that have broad substrate specificity and are induced by salicylic acid and auxin, Eur. J. Biochem., 2001, vol. 268, pp. 4086–4094.
Kang, S.M., Jung, H.Y., Kang, Y.M., Yun, D.J., Bahk, J.D., Yang, J., and Choi, M.S., Effects of methyl jasmonate and salicylic acid on the production of tropane alkaloids and the expression of PMT and H6H in adventitious root cultures of Scopolia parviflora, Plant Sci., 2004, vol. 166, pp. 745–751.
Author information
Authors and Affiliations
Corresponding author
Additional information
This text was submitted by the authors in English.
Rights and permissions
About this article
Cite this article
Rahimi, S., Devi, B.S.R., Khorolragchaa, A. et al. Effect of salicylic acid and yeast extract on the accumulation of jasmonic acid and sesquiterpenoids in Panax ginseng adventitious roots. Russ J Plant Physiol 61, 811–817 (2014). https://doi.org/10.1134/S1021443714060156
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1021443714060156