Abstract—
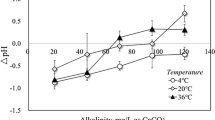
Rates of mass transfer controlled Fe/Cu galvanic corrosion at the wall lining of a cylindrical agitated vessel in different fertilizer electrolytic media were studied. Variables investigated are impeller rotation speed, impeller geometry, electrolyte type, electrolyte concentration, area ratio (anode/cathode), electrolyte temperature, and effect of polyethylene oxide (drag reducing polymer) as a corrosion inhibitor. The corrosion rate was determined by following the change of the concentration of anode metal ions (Fe++) in the electrolyte with time. Scanning electron microscopy (SEM) was carried out for steel sheets subjected to different electrolytes. The results showed that the galvanic corrosion rate increased with increasing the impeller rotation speed, the electrolyte concentration, and temperature. The presence of polyethylene oxide decreased the rate of corrosion by 11–35.21% depending on polymer concentration and operating conditions. Axial flow impeller produces corrosion rates higher than that with the radial flow type under the same operating parameters. Application of the results in the design of agitated vessels handling fertilizer media was discussed.











Similar content being viewed by others
REFERENCES
Sheir, L.L. and Jarman, R.A., Corrosion (Metal/Environment Reaction), London: Butterworth-Heinemann, 1998.
Roberge, P.R., Handbook of Corrosion Engineering, New York: McGraw-Hill, 2000.
Fontana, M.G., Corrosion Engineering, New York: McGraw-Hill, 1987.
Al-Ammeri, R.S., Influence of drag reducing additives on power consumption in agitated vessels, Chem. Eng. Commun., 1987, vol. 59, nos. 1–6, p. 1.
Quraishi, A.Q., Mashelkar, R.A., and Ulbrecht, J., Torque suppression in mechanically stirred liquids and multiphase liquid systems, J. Non-Newtonian Fluid Mech., 1976, vol. 1, no. 3, p. 223.
Mashelkar, R.A., Kale, D.D., and Ulbrecht, J.J., Rotational flow of non-Newtonian fluids—Torque suppression with agitators, Trans. Inst. Chem. Eng., 1975, vol. 53, p. 150.
Findlay, A. and Kitchener, J.K., Practical Physical Chemistry, London: Longman, 1965.
Little, R., Smidt, S., Huang, P., Romans, J., Dedrick, J., and Matuszko, J.S., Improved drag reduction by control of polymer particle size, Ind. Eng. Chem. Res., 1991, vol. 30, no. 2, p. 403.
Mahato, B.K., Voora, S.K., and Shemilt, L.W., Steel pipe corrosion under flow condition—An isothermal correlation for a mass transfer model, Corros. Sci. 1968, vol. 8, no. 3 p. 173.
Foroulis, Z.A., The influence of velocity and dissolved oxygen on the initial corrosion behavior of iron in high purity water, Corrosion, 1979, vol. 3, no. 8, p. 340.
Nesic, S. and Postlethwaite, J., Hydrodynamics of disturbed flow and erosion–corrosion. Part I—Single-phase flow study, Can. J. Chem. Eng., 1991, vol. 69, no. 3, p. 698.
Shreir, L.L., Jarman, R.A., and Burstein, G.T., Corrosion: Metal/Environment Reactions, London: Butterworth-Heinemann, 2000, vol. 1, 3rd ed.
Dkhireche, N., Dahami, A., Rochdi, A., Hmimou, J., Touir, R., Ebn Touhami, M., El Bakri, M., El Hallaoui, A., Anouar, A., and Takenouti, H., Corrosion and scale inhibition of low carbon steel in cooling water system by 2-propargyl-5-o-hydroxyphenyltetrazole, J. Ind. Eng. Chem., 2013, vol. 19, no. 6, p. 1996.
Quevedo, M.C., and Genesca, J., Influence of turbulent flow on the corrosion of Al–Zn–Mg galvanic anode in artificial seawater media, J. Mater. Corros., 2009, vol. 60, no. 6, p. 424.
Montañés, M.T., Tovar, R.S., García-Antón, J., and Herranz, V.P., The influence of Reynolds number on the galvanic corrosion of the copper/AISI 304 pair in aqueous LiBr solutions, Corros. Sci., 2009, vol. 51, no. 11, p. 2733.
Dawnson, A. and Trass, O., Mass transfer at rough surfaces, Int. J. Heat Mass Transfer, 1972, vol. 15, pp. 1317–1336.
Berger, K.F. and Hau, F.B., Local mass transfer distribution on surfaces roughened with small square ribs, Int. J. Heat Mass Transfer, 1979, vol. 22, no. 12, p. 1645.
Hasan, B.O., Galvanic corrosion of carbon steel–brass couple in chloride containing water and the effect of different parameters, J. Pet. Sci. Eng., 2014, vol. 124, p. 137.
Slaiman, Q.J.M. and Hasan, B.O., Study on corrosion rate of carbon steel pipe under turbulent flow conditions, Can. J. Chem. Eng., 2012, vol. 88, no. 6, p. 1114.
Kalinnikov, V.T., Makarov, D.V., Makarov, V.N., Oxidation sequence of sulfide minerals in operating and out-of-service mine waste storage, Theor. Found. Chem. Eng., 2001, vol. 35, no. 1, p. 63.
Heitz, E., Mechanistically based prevention strategies of flow-induced corrosion, Electrochim. Acta, 1996, vol. 41, no. 4, p. 503.
Montañés, M.T., Sánchez-Tovar, R., García-Antó, J., and Pérez-Herranz, V., Effects of flow variations on the galvanic corrosion of the copper/AISI 304 stainless steel pair in lithium bromide using a zero-resistance ammeter, Int. J. Electrochem. Sci., 2012, vol. 7, no. 1, p. 747.
Shadley, J.R., Shirazi, S.A., Dayalan, E., Ismail, M., and Rybicki, E.F., Erosion-corrosion of a carbon steel elbow in a carbon dioxide environment, Corrosion, 1996, vol. 52, no. 9, p. 714.
Al Hossani, H.I., Saber, T.M.H., Mohammed, R.A., and Shams El Din, A.M., Galvanic corrosion of copper-base alloys in contact with molybdenum-containing stainless steels in Arabian Gulf water, Desalination, 1997, vol. 109, no. 1, p. 25.
Sánchez-Tovar, R., Montañés, M.T., and Garcia, J., The effect of temperature on the galvanic corrosion of the copper/AISI 304 pair in LiBr solutions under hydrodynamic conditions, Corros. Sci., 2009, vol. 52, no. 3, p. 722.
Montañes, M.T., Sánchez-Tovar, R., Garcia-Antón, J., Pérez-Herranz, V., The influence of Reynolds number on the galvanic corrosion of the copper/AISI 304 pair in aqueous LiBr solutions, Corros. Sci., 2009, vol. 51, no. 11, p. 2733.
Zahran, R.R., and Sedahmed, G.H., Galvanic corrosion of zinc in turbulently moving saline water containing drag reducing polymers, Mater. Lett., 1997, vol. 31, no. 1, p. 29.
Perry, R.H. and Green, D.W., Perry’s Chemical Engineers’ Handbook, New York: McGraw-Hill, 2008, 8th ed.
Friend, J.A.N. and Denett, J., VIII.—The rate of solution of iron in dilute sulphuric acid both when stationary and under rotation, J. Chem. Soc. Trans., 1922, vol. 121 p. 41.
Welty, J.R., Wicks, C.E., Wilson, R.E., and Rorrer, G.L., Fundamentals of Momentum, Heat, and Mass Transfer, New York: Wiley, 2008.
Incropera, F.P. and Dewitt, D.P., Fundamentals of Heat and Mass Transfer, New York: Wiley, 1990, 3rd ed.
Edward, J.T., Molecular volumes and the Stokes–Einstein equation, J. Chem. Educ., 1970, vol. 47, no. 4, p. 261.
Garfias-Mesias, L.F. and Sykes, J.M., Metastable pitting in 25 Cr duplex stainless steel, Corros. Sci., 1999, vol. 41, no. 5, p. 959.
Guo, R. and Ives, M.B., Pitting susceptibility of stainless steels in bromide solutions at elevated temperatures, Corrosion, 1990, vol. 46, no. 2, p. 125.
Yaro, A.S., Hameed, K.W., and Khadom, A.A., Study for prevention of steel corrosion by sacrificial anode cathodic protection, Theor. Found. Chem. Eng., 2013, vol. 47, no. 3, p. 266.
Askew, W.S. and Beckmann, R.B., Heat and mass transfer in an agitated vessel, Ind. Eng. Chem. Process Des. Dev., 1965, vol. 4, no. 3, p. 311.
Sedahmed, G.H., Farag, H.A., Kayar, A.M., and El-Nashar, I.M., Mass transfer at the impellers of agitated vessels in relation to their flow-induced corrosion, Chem. Eng. J., 1998, vol. 71, no. 1, p. 57.
Sedahmed, G.H., El-Taweel, Y.A., Abdel-Aziz, M.H., and El-Naqeara, H.M., Mass and heat transfer enhancement at the wall of cylindrical agitated vessel by active turbulence promoters, Chem. Eng. Process., 2014, vol. 80, p. 43.
Mansfeld, F., Liu, G., Xiao, H., Tsai, C.H., and Little, B.J., The corrosion behavior of copper alloys, stainless steels and titanium in seawater, Corros. Sci., 1994, vol. 36, no. 12, p. 2063.
Alfantazi, A.M., Ahmed, T.M., and Tromans, D., Corrosion behavior of copper alloys in chloride media, Mater. Des., 2009, vol. 30, no. 7, p. 2425.
Mansfeld, F. and Kenkel, J.V., Galvanic corrosion of Al alloys—III. The effect of area ratio, Corros. Sci., 1975, vol. 15, no. 4, p. 239.
Tsujino, B. and Miyase, S., The galvanic corrosion of steel in sodium chloride solution, Corrosion, 1982, vol. 38, no. 4, p. 226.
Sellin, R.H., Hoyt, J.W., and Serivener, O., The effect of drag reducing additives on fluid flow and their industrial applications, J. Hydraul. Res., 1982, vol. 20, no. 3, p. 29.
Sedahmed, G.H. and Griskey, R.G., Mass transfer in drag reducing fluid systems, AIChE J., 1972, vol. 18, no. 1, p. 138.
Popov, V.I., Rheodynamic factor friction drag reduction in the channel and surface, Theor. Found. Chem. Eng., 2015, vol. 49, no. 3, p. 330.
Abdel-Aziz, M.H., Nirdosh, I., and Sedahmed, G.H., Liquid–solid mass and heat transfer behavior of a concentric tube airlift reactor, Int. J. Heat Mass Transfer, 2013, vol. 58, no. 1, p. 735.
Amin, N.K., Abdel-Aziz, M.H., and El-Ashtoukhy, E-S.Z., Effect of pulp fiber suspensions on the rate of mass transfer controlled corrosion in pipelines under turbulent flow conditions, Chem. Eng. Res. Des., 2014, vol. 92, no. 11, p. 2333.
Abdel-Aziz, M.H., Solid-liquid mass transfer in relation to diffusion controlled corrosion at the outer surface of helical coils immersed in agitated vessels, Chem. Eng. Res. Des., 2013, vol. 91, no. 1, p. 43.
Sedahmed, G.H., The use of drag reducing polymers to combat diffusion controlled corrosion and erosion–corrosion in equipments operating under turbulent flow, Trends Chem. Eng., 2005, vol. 9, p. 65.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Amer, B.A., Abdel-Aziz, M.H., El-Ashtoukhy, ES.Z. et al. Galvanic Corrosion of Steel in Agitated Vessels Used in Fertilizer Industry. Theor Found Chem Eng 53, 280–291 (2019). https://doi.org/10.1134/S0040579519020015
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0040579519020015