Abstract
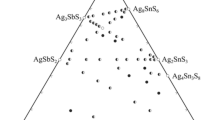
Solid–liquid phase equilibria of the quaternary system (Li+,K+//\({\text{SO}}_{4}^{{2 - }}\),\({\text{CO}}_{3}^{{2 - }}\)–H2O) and its subsystem are very important for the separation of lithium and potassium salts from the salt lake brines. In this paper, the solubilities, densities, refractive indices and pH values of the reciprocal quaternary system (Li+,K+//\({\text{SO}}_{4}^{{2 - }}\),\({\text{CO}}_{3}^{{2 - }}\)–H2O) at 308.15 K were determined experimentally using the isothermal dissolution method. The dry-salt diagram and water-phase diagram were plotted based on the experimental data. The stable phase diagram contains four invariant points, nine univariant curves and six crystallization regions corresponding to Li2CO3, Li2CO3⋅K2CO3, K2CO3⋅1.5H2O, Li2SO4⋅H2O, K2SO4, and Li2SO4⋅K2SO4. Li2CO3 has the largest crystallization field, whereas the Li2CO3⋅K2CO3 crystallization field is the smallest one.





Similar content being viewed by others
REFERENCES
C. X. Jiang, B. L. Chen, D. Y. Zhang, et al., Chin. J. Chem. Eng. 73, 481 (2022).
F. Zhang, Exploration of the technology of lithium extraction from seawater by electrolysis based on hybrid electrolyte (Nanjing Univ., 2020).
T. Ge, L. Xu, J. W. Meng, et al., Nonferr. Met. Eng. 11, 55 (2021).
P. Christmann, E. Gloaguen, J.-F. Labbé, et al., Global Lithium Resources and Sustainability Issues (Lithium Process Chemistry, 2015).
M. P. Zheng, Miner. Depos. 20, 181 (2001).
C. L. Huang, Sichuan Nonferr. Met. 1, 29 (2004).
Y. Y. Zhao, M. P. Zheng, L. Z. Bu, et al., Chin. J. Sea-lake Salt Chem. Ind. 34, 1 (2005).
M. P. Zheng and W. Qi, Conserv. Util. Miner. Resour. 5, 15 (2006).
R. Q. Yuan and F. Q. Cheng, Chin. J. Salt Lake Res. 16, 67 (2008).
S. Y. Gao, P. S. Song, M. P. Zheng, et al., Salt Lake Chemicals (Science Press, Beijing, 2007).
M. M. Yang, J. Jiang, C. Gao, et al., Russ. J. Inorg. Chem. 66, 2116 (2021).
W. Qian, N. Zhen, L. Z. Bu, et al., Chin. Inorg. Chem. Ind. 6, 28 (2018).
J. J. Li, Y. Zeng, X. D. Yu, et al., J. Chem. Eng. Data 58, 455 (2013).
J. J. Zhang, M. Y. Ren, D. C. Li, et al., Russ. J. Phys. Chem. A 94, 1565 (2020).
T. L. Deng, H. Zhou, and X. Chen, Salt-Water System Phase Diagrams and Applications (Chin. Chem. Industry Press, Beijing, 2013).
S. Q. Wang, Y. Song, X. M. Du, et al., Russ. J. Inorg. Chem. 63, 116 (2018).
F. Yuan, J. Jang, S. Q. Wang, et al., J. Mol. Liq. 337, 116334 (2021).
Analysis of Brine and Salt: Qinghai Institute of Salt Lakes (Science Press, Chin. Acad. Sciences, Beijing, 1988).
C. S. Shang, L. Y. An, and Z. W. Hu, Chem. Res. Appl. 24, 642 (2012).
J. Nyvlt, Solid - Liquid Phase Equilibria (Academia, Praha, Czech Republic, 1977).
Funding
Financial support from National Natural Science Foundation of China (22078247), and the 2023 Kunlun Talents Program: High-End Innovative and Entrepreneurial Talents of Qinghai Province are greatly acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cheng, H., Zhang, S., Han, S. et al. Solid–Liquid Stable Phase Equilibria of the Quaternary System (Li+,K+//\({\text{SO}}_{4}^{{2 - }}\),\({\text{CO}}_{3}^{{2 - }}\)–H2O) at 308.15 K. Russ. J. Phys. Chem. (2024). https://doi.org/10.1134/S0036024424020079
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1134/S0036024424020079