Abstract
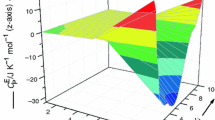
The pressure of saturated and unsaturated Br2 vapors over (cation)2{[TeBr6](Br2)} (cation = Me4N+, Et4N+) solid polybromotellurates is measured via static tensimetry using membrane zero manometers in a wide range of temperatures. Experimental data are used to determine the thermal stability of these compounds (Tdec). A physicochemical model of vaporization is proven, thermodynamic characteristics of the evaporation of bromine that bonds the moieties of polybromotellurate anions (\({{\Delta }_{{{\text{pr}}}}}H_{T}^{^\circ }\), \({{\Delta }_{{{\text{pr}}}}}S_{T}^{^\circ }\), \(\ln p\) = f(T)) are calculated, and the energies of bonding between [TeBr6] octahedra and Br2 (\({{\Delta }_{{\text{b}}}}G_{T}^{^\circ }\)) are determined. Results are compared to others for bismuth polybromide complexes studied earlier.





Similar content being viewed by others
REFERENCES
J. Pelletier and J. Caventou, Ann. Chim. Phys. 10, 142 (1819).
P. H. Svensson and L. Kloo, Chem. Rev. 103, 1649 (2003). https://doi.org/10.1021/cr0204101
K. Sonnenberg, L. Mann, F. A. Redeker, et al., Angew. Chem. Int. Ed. 59, 5464 (2020). https://doi.org/10.1002/anie.201903197
G. R. Desiraju, P. Shing Ho, L. Kloo, et al., Pure Appl. Chem. 85, 1711 (2013). https://doi.org/10.1351/PAC-REC-12-05-10
G. Cavallo, P. Metrangolo, R. Milani, et al., Chem. Rev. 116, 2478 (2016). https://doi.org/10.1021/acs.chemrev.5b00484
N. A. Korobeynikov, A. N. Usoltsev, P. A. Abramov, et al., Inorganics 11, 25 (2023). https://doi.org/10.3390/inorganics11010025
H. Keil, K. Sonnenberg, C. Müller, et al., Angew. Chem. Int. Ed. 60, 2569 (2021). https://doi.org/10.1002/anie.202013727
R. Brückner, H. Haller, S. Steinhauer, et al., Angew. Chem. Int. Ed. 54, 15579 (2015). https://doi.org/10.1002/anie.201507948
K. Sonnenberg, P. Pröhm, N. Schwarze, et al., Angew. Chem. Int. Ed. 57, 9136 (2018). https://doi.org/10.1002/anie.201803486
P. Voßnacker, A. Wüst, C. Müller, et al., Angew. Chem. Int. Ed. 61, e202209684 (2022). https://doi.org/10.1002/anie.202209684
N. A. Korobeynikov, A. N. Usoltsev, B. A. Kolesov, et al., CrystEngComm. 24, 3150 (2022). https://doi.org/10.1039/D2CE00210H
T. A. Shestimerova, N. A. Yelavik, A. V. Mironov, et al., Inorg. Chem. 57, 4077 (2018). https://doi.org/10.1021/acs.inorgchem.8b00265
T. A. Shestimerova, A. V. Mironov, M. A. Bykov, et al., Molecules 25, 2765 (2020). https://doi.org/10.3390/molecules25122765
A. V. Bykov, T. A. Shestimerova, M. A. Bykov, et al., Int. J. Mol. Sci. 24, 2201 (2023). https://doi.org/10.3390/ijms24032201
T. A. Shestimerova, M. A. Bykov, A. V. Grigorieva, et al., Mendeleev Commun. 32, 194 (2022). https://doi.org/10.1016/j.mencom.2022.03.014
T. A. Shestimerova, N. A. Golubev, M. A. Bykov, et al., Molecules 26, 5712 (2021). https://doi.org/10.3390/molecules26185712
M. Küttinger, P. A. Loichet Torres, E. Meyer, et al., Chem. - A Eur. J. 28, e202103491 (2022). https://doi.org/10.1002/chem.202103491
M. Küttinger, R. Riasse, J. Wlodarczyk, et al., J. Power Sources 520, 230804 (2022). https://doi.org/10.1016/j.jpowsour.2021.230804
W. Wu, J. Luo, F. Wang, et al., ACS Energy Lett. 6, 2891 (2021). https://doi.org/10.1021/acsenergylett.1c01146
A. N. Usoltsev, S. A. Adonin, A. S. Novikov, et al., CrystEngComm. 19, 5934 (2017). https://doi.org/10.1039/C7CE01487B
A. N. Usoltsev, S. A. Adonin, P. A. Abramov, et al., Eur. J. Inorg. Chem. 2018, 3264 (2018). https://doi.org/10.1002/ejic.150+273201800383
L. N. Zelenina, T. P. Chusova, A. V. Isakov, et al., J. Chem. Thermodyn. 141, 105958 (2020). https://doi.org/10.1016/j.jct.2019.105958
A. V. Suvorov, Thermodynamic Chemistry of the Vapor State (Khimiya, Leningrad, 1970), p. 46 [in Russian].
L. N. Zelenina, T. P. Chusova, and I. G. Vasilyeva, J. Chem. Thermodyn. 57, 101 (2013). https://doi.org/10.1016/j.jct.2012.08.005
L. N. Zelenina, T. P. Chusova, S. A. Sapchenko, and N. V. Gelfond, Russ. J. Inorg. Chem. 68, 140 (2023). https://doi.org/10.31857/S0044457X22601274
V. A. Titov and G. A. Kokovin, in Mathematical Methods in Chemical Thermodynamics (Nauka, Novosibirsk, 1980), p. 98 [in Russian].
L. V. Gurvich, Vest. Akad. Nauk SSSR, No. 3, 54 (1983).
Funding
This work was supported by the RF Ministry of Science and Higher Education, project nos. 121031700314-5 and 121031700313-8.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by M. Timoshinina
ADDITIONAL INFORMATION
This article is part of the Materials of the XV Symposium with International Participation “Thermodynamics and Materials Science,” Novosibirsk, July 3–7, 2023.
Publisher’s Note.
Pleiades Publishing remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zelenina, L.N., Chusova, T.P., Korobeinikov, N.A. et al. Thermodynamic Study of Tellurium Polybromide Complexes. Russ. J. Phys. Chem. (2024). https://doi.org/10.1134/S003602442401031X
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1134/S003602442401031X