Abstract
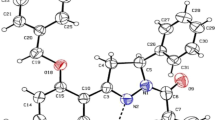
In this work, the electron density distribution of (Z)-3-N-(ethyl)-2-N '-(3-methoxyphenyl imino) thiazolidine-4-one is determined by single-crystal X-ray refinements using a spherical structure factors and multipolar model of Hansen and Coppens. The following crystal properties are determined: laplacian of electron density and critical points. These parameters are basic for understanding the nature of intra- and intermolecular charge transfer and to highlight the chemical reactivity of the molecule. From the deformation density maps, density accumulations are visible in bonding as well as in the lone-pair regions of oxygen atoms (O1 and O2). The results of reduced density gradient, of natural bond orbital and of Hirshfeld surface analysis allow obtaining respectively; information’s of the intra non-bonded interactions, charge transfer and to understand what kind of interatomic contacts give the largest contributions in crystal structure. The relative high value of local reactivity descriptors indicate that S atom is the most preferred site for nucleophilic attacks whereas C4 and C8 sites are the most nucleophilic centers. In addition, the compound under investigation presents a biological activity when it is docked into the protein (PDB ID: 2AZ5) with the binding energy system of –6.3 kcal/mol. This compound showed excellent interaction with amino acids constitutes this protein and showed significant inhibition of tumor necrosis factor.












Similar content being viewed by others
REFERENCES
D. Havrylyuk, B. Zimenkovsky, O. Vasylenko, A. Gzella, and R. Lesyk, J. Med. Chem. 55, 8630 (2012).
M. Fesatidou, P. Zagaliotis, C. Camoutsis, A. Petrou, P. Eleftheriou, and C. Tratrat, Bioorg. Med. Chem. 26, 4664 (2018).
I. N. Colmers, S. L. Bowker, S. R. Majumdar, and J. A. Johnson, Can. Med. Assoc. J. 184, E675 (2012).
S. Nirwan, V. Chahal, and R. Kakkar, J. Heterocycl. Chem. 56, 1239 (2019).
J. L. Pinto, J. A. Henao, and V. Kouznetsov, Powder Diffract. 33, 225 (2018).
A. Djafri, A. Chouaih, J.-C. Daran, A. Djafri, and F. Hamzaoui, Acta Crystallogr., E 73, 511 (2017). https://doi.org/10.1107/S205698017003218
R. Rahmani, A. Djafri, J.-C. Daran, A. Djafri, A. Chouaih, and F. Hamzaoui, Acta Crystallogr., E 72, 155 (2016).
M. Boulakoud, K. Toubal, S. Yahiaoui, G. Chita, A. Chouaih, and A. Djafri, J. Struct. Chem. 56, 1373 (2015).
N. Benhalima, K. Toubal, A. Chouaih, G. Chita, S. Maggi, A. Djafri, and F. Hamzaoui, J. Chem. Crystallogr. 41, 1729 (2011).
Y. Megrouss, F. Triki-Baara, N. Boukabcha, A. Chouaih, A. Hatzidimitriou, A. Djafri, and F. Hamzaoui, Acta Chim. Slov. 66, 490 (2019).
T. Gruene and E. Mugnaioli, Chem. Rev. 121, 11823 (2021). https://doi.org/10.1021/acs.chemrev.1c00207
B. T. W. Lo, L. Ye, and Sh. Ch. E. Tsang, Chemistry 4, 1778 (2018). https://doi.org/10.1016/j.chempr.2018.04.018
E. C. Escudero-Adán, A. Bauzá, A. Frontera, and P. Ballester, Chem. Phys. Chem. 16, 2530 (2015). https://doi.org/10.1002/cphc.201500437
P. W. Betteridge, J. R. Carruthers, R. I. Cooper, K. Prout, and D. J. Watkin, J. Appl. Crystallogr. 36, 1487 (2003). https://doi.org/10.1016/j.chempr.2018.04.018
L. Palatinus and G. Chapuis, J. Appl. Crystallogr. 40, 786 (2007). https://doi.org/10.1107/S0021889807029238
N. K. Hansen and P. Coppens, Acta Crystallogr., A 34, 909 (1978). https://doi.org/10.1107/S0567739478001886
C. Jelsch, B. Guillot, A. Lagoutte, and C. Lecomte, J. Appl. Crystallogr. 38, 38 (2005). https://doi.org/10.1107/S0021889804025518
P. Coppens, X-Ray Charge Densities and Chemical Bonding, Vol. 4 of IUCr Texts on Crystallography (Oxford, New York, 1997).
M. A. Spackman, Ann. Rep. Prog. Chem. C 94, 177 (1998). https://doi.org/10.1039/PC09417777
E. R. Johnson, S. Keinan, P. Mori-Sánchez, J. Contreras-García, A. J. Cohen, and W. Yang, J. Am. Chem. Soc. 132, 6498 (2010). https://doi.org/10.1021/ja100936w
E. D. Glendening, J. K. Badenhoop, A. E. Reed, J. E. Carpenter, and F. Weinhold, NBO Version 3.1 (Theor. Chem. Inst., Univ. Wisconsin, Madison, WI, 1995).
M. J. Frisch et al., Gaussian 09, Revision A.11.4 (Gaussian Inc., Wallingford, CT, 2009).
F. Weinhold and C. R. Landis, Chem. Educ. Res. Pract. 2, 91 (2001). https://doi.org/10.1039/B1RP90011K
L. J. Farrugia, J. Appl. Crystallogr. 45, 849 (2012). https://doi.org/10.1107/S0021889812029111
R. F. W. Bader, Atoms in Molecules: A Quantum Theory (Clarendon, Oxford, UK, 1990).
C. Kalaiarasi, C. George, and R. G. Gonnade, Acta Crystallogr., B 75, 942 (2019). https://doi.org/10.1107/S2052520619011272
R. F. W. Bader, J. Phys. Chem. A 113, 10391 (2009). https://doi.org/10.1021/jp906341r
T. S. Koritsanszky and P. Coppens, Chem. Rev. 101, 1583 (2001). https://doi.org/10.1021/cr990112c
H. Benaissi, M. Drissi, S. Yahiaoui, and Y. Megrouss, J. Optoelectron. Biomed. Mater. 10, 73 (2018).
Tian Lu and Feiwu Chen, J. Comput. Chem. 33, 580 (2012). https://doi.org/10.1002/jcc.22885
W. Humphrey, A. Dalke, and K. Schulten, J. Mol. Graph. 14, 33 (1996). https://doi.org/10.1016/0263-7855(96)00018-5
M. Cerón, T. Gomez, M. Calatayud, and C. Cárdenas, J. Phys. Chem. A 124, 2826 (2020). https://doi.org/10.1021/acs.jpca.0c00950
P. W. Ayers, W. Yang, and L. J. Bartolotti, in Chemical Reactivity Theory: A Density Functional View, Ed. by P. K. Chattaraj (CRC, Taylor and Francis Group, Boca Raton, 2010).
N. Khelloul, K. Toubal, N. Benhalima, R. Rahmani, A. Chouaih, A. Djafri, and F. Hamzaoui, Acta Chim. Slov. 63, 619 (2016). https://doi.org/10.17344/acsi.2016.2362
S. K. Wolff, D. J. Grimwood, J. J. McKinnon, D. Jayatilaka, and M. A. Spackman, Crystal Explorer 3.1 (Univ. Western Austral., Perth, Australia, 2012).
R. Vafazadeh, A. Kazemi-nasab, and A. C. Willis, Acta Chim. Slov. 66, 1010 (2019). https://doi.org/10.17344/acsi.2019.5333
L. Bejaoui, J. Rohlíček, V. Eigner, A. Ismail, M. El Bour, and R. Ben Hassen, Acta Chim. Slov. 66, 603 (2019). https://doi.org/10.17344/acsi.2019.5002
PASS Online prediction. http://www.way2drug.com/passonline/.
P. P. Shinoj Kumar, G. Krishnaswamy, N. R. Desai, S. Sreenivasa, and D. B. Aruna Kumar, Chem. Data Collect. 31, 100617 (2021). https://doi.org/10.1016/j.cdc.2020.100617
R. P. D. Bank, RCSB PDB: Homepage. https://www.rcsb.org/.
H. T. Idriss and J. H. Naismith, J. Micro. Res. Tech. 50, 184 (2000). https://doi.org/10.1002/1097-0029(20000801)50:3<184::AID-JEMT2>3.0.CO;2-H
O. T. P. Kim, M. D. Le, H. X. Trinh, and H. V. Nong, Biophys. Physicobiol. 13, 173 (2016). https://doi.org/10.2142/biophysico.13.0_173
Accelrys Software Inc., Discovery Studio Modeling Environment, Releases 3.5 (Accelrys Discovery Studio, San Diego, 2012).
O. Trott and A. J. Olson, J. Comput. Chem. 31, 455 (2010). https://doi.org/10.1002/jcc.21334
R. S. Shinde, V. H. Masand, and M. K. Patil, Indian J. Pharm. Sci. 81, 851 (2019). https://doi.org/10.36468/pharmaceutical-sciences.579
T. Topal, Y. Zorlu, and N. Karapınar, J. Mol. Struct. 1239, 130514 (2021). https://doi.org/10.1016/j.molstruc.2021.130514
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Megrouss, Y., Yahiaoui, S., Boukabcha, N. et al. Charge Density Study, DFT Calculations, Hirshfield Surface Analysis and Molecular Docking of (Z)-3-N-(Ethyl)-2-N '-(3-methoxyphenyl imino) thiazolidine-4-one. Russ. J. Phys. Chem. 97, 1731–1745 (2023). https://doi.org/10.1134/S0036024423080319
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024423080319