Abstract
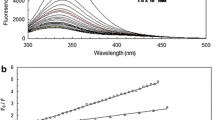
Interaction of atenolol (Atn) and metoprolol (Met) beta-blocker drugs with human serum albumin (HSA) at pH 7.4 was investigated by UV–Vis spectroscopy and molecular modeling methods. In addition, the binding interaction information of Atn and Met with HSA practicaly of binding constant (Kb) were determined by the UV–Vis absorption titration at body actual temperature (310.15 K), and were found to be 9.5 × 103, 1.4 × 104, and 2.3 × 104 M–1 for Atn, Met, and Atn + Met, respectively. The results indicated that addition of Atn in many concentrations causes decrease, while addition of Met and Met + Atn leads to an increase in absorption upon complexation with HSA. The binding mechanisms of two target drugs are different from each other, as indicated by spectroscopic analysis. The results show that interactions in all systems are exothermic with negative entropy, which lead to more spontaneous reaction at lower temperatures. Molecular dynamics simulation calculations show that the number of interactive residues between Met and protein is less than the number of interactive residues between Atn and protein.









Similar content being viewed by others
REFERENCES
H. R. Costantino, L. Illum, G. Brandt, P. H. Johnson, and S. C. Quay, Int. J. Pharm. 337, 1 (2007).
G. M. Currie, J. M. Wheat, and H. Kiat, Open Cardiovasc. Med. J. 5, 130 (2011).
T.-Y. Jiang, K.-L. Zhou, Y.-Y. Lou, D.-Q. Pan, and J.‑H. Shi, J. Biomol. Struct. Dyn. 36, 1095 (2018).
B. X. Huang, H. Y. Kim, and Ch. Dass, J. Am. Soc. Mass Spectrom. 15, 1237 (2004).
P. Keen, in Concepts in Biochemical Pharmacology (Springer, Berlin, Heidelberg, 1971), p. 213.
B. B. Brodie and C. A. M. Hogben, J. Pharm. Pharmacol. 9, 345 (1957).
Sh. Zhou, J. W. Paxton, Ph. Kestell, M. D. Tingle, and Ph. Kestell, J. Pharm. Pharmacol. 53, 463 (2001).
K. E. D. Coan and B. K. Shoichet, Mol. Biosyst. 3, 208 (2007).
M. George and T. E. Abraham, J. Control. Release 114, 1 (2006).
W. J. Jusko and M. Gretch, Drug Metab. Rev. 5, 43 (1976).
B. E. P. Murphy, J. Clin. Endocrinol. Metab. 27, 973 (1967).
M. Maciążek-Jurczyk, A. Sułkowska, B. Bojko, J. Równicka, and W. W. Sułkowski, J. Mol. Struct. 924, 378 (2009).
M. M. Browne, G. V. Lubarsky, M. R. Davidson, and R. H. Bradley, Surf. Sci. 553, 155 (2004).
P. B. Desai and A. K. Srivastava, Sens. Actuators, B 176, 632 (2013).
L. Kranitz and P. Lehrer, Cardiol. Rev. 12, 177 (2004).
D. E. Donald, D. A. Ferguson, and S. E. Milburn, Circul. Res. 22, 127 (1968).
A. Najafi, V. Sequeira, D. W. D. Kuster, and J. van der Velden, Eur. J. Clin. Invest. 46, 362 (2016).
C. J. Homcy, S. F. Vatner, and D. E. Vatner, Ann. Rev. Physiol. 53, 137 (1991).
D. A. Sica, T. W. B. Gehr, and S. Ghosh, Pharmacokinetics 44, 797 (2005).
M. R. Housaindokht, Z. R. Zaeri, and M. Bahrololoom, Spectrochim. Acta, Part A 85, 79 (2012).
Y. Landry and J.-P. Gies, Fundam. Clin. Pharmacol. 22, 1 (2008).
X. Li, X. Xu, J. Wang, H. Yu, X. Wang, H. Yang, H. Xu, et al., PloS One 7 (9), e43918 (2012).
P. Ascenzi, G. Fanali, M. Fasano, V. Pallottini, and V. Trezza, J. Mol. Struct. 1077, 4 (2014).
M. Bern, K. M. K. Sand, J. Nilsen, I. Sandlie, and J. T. Andersen, J. Control. Release 211, 144 (2015).
B. P. Espósito and R. Najjar, Coord. Chem. Rev. 232, 137 (2002).
F. A. de Wolf and G. M. Brett, Pharmacol. Rev. 52, 207 (2000).
Md Tabish Rehman and A. U. Khan, Curr. Pharm. Des. 21, 1785 (2015).
X. M. He and D. C. Carter, Nature (London, U.K.) 358 (6383), 209 (1992).
U. Kragh-Hansen, F. Hellec, B. de Foresta, M. le Maire, and J. V. Møller, Biophys. J. 80, 2898 (2001).
J. Costa Pessoa and I. Tomaz, Curr. Med. Chem. 17, 3701 (2010).
M. S. Benedetti, R. Whomsley, I. Poggesi, W. Cawello, F.-X. Mathy, M.-L. Delporte, P. Papeleu, and J.‑B. Watelet, Drug Metab. Rev. 41, 344 (2009).
K. Yamasaki, V. T. Giam Chuang, T. Maruyama, and M. Otagiri, Biochim. Biophys. Acta 1830, 5435 (2013).
F. Poureshghi, P. Ghandforoushan, A. Safarnejad, and S. Soltani, J. Photochem. Photobiol. B 166, 187 (2017).
X.-X. Cheng, Y. Lui, B. Zhou, X.-H. Xiao, and Y. Liu, Spectrochim. Acta, Part A 72, 922 (2009).
B. Sandhya, A. H. Hegde, Sh. S. Kalanur, U. Katrahalli, and J. Seetharamappa, J. Pharm. Biomed. Anal. 54, 1180 (2011).
H. Xu, N. Yao, H. Xu, T. Wang, G. Li, and Zh. Li, Int. J. Mol. Sci. 14, 14185 (2013).
S. Gao and R. Liu, New J. Chem. 42, 9065 (2018).
T. S. Banipal, A. Kaur, and P. K. Banipal, Spectrochim. Acta, Part A 170, 214 (2017).
X.-L. Han, F.-F. Tian, Y.-Sh. Ge, F. L. Jiang, L. Lai, D.-W. Li, Q.-L. Yu, J. Wang, Ch. Lin, and Y. Liu, J. Photochem. Photobiol. B 109, 1 (2012).
B. Knapp, S. Frantal, M. Cibena, W. Schreiner, and P. Bauer, J. Comput. Biol. 18, 997 (2011).
O. Carugo and S. Pongor, Protein Sci. 10, 1470 (2001).
H. Lei, Ch. Wu, H. Liu, and Y. Duan, Proc. Natl. Acad. Sci. U. S. A. 104, 4925 (2007).
M. R. Housaindokht, M. R. Bozorgmehr, and H. Monhemi, J. Supercrit. Fluids 63, 180 (2012).
Farideh Mirzaee, Leila Hosseinzadeh, Mohammad Reza Ashrafi-Kooshk, Sajjad Esmaeili, Sirous Ghobadi, Mohammad Hosein Farzaei, Mahmoud Reza Zad-Bari, and Reza Khodarahmi, Protein and Peptide Lettes 26 (2), 132 (2019). https://doi.org/10.2174/0929866525666181114152242
ACKNOWLEDGMENTS
The authors wish to acknowledge the support of this work by Sadri Ebrahimnia and Radvin Raoufi.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Raoufi, A., Ebrahimi, M. & Bozorgmehr, M.R. Determination of Thermodynamics Constant of Interaction among of Atenolol and Metoprolol with Human Serum Albumin: Spectroscopic and Molecular Modeling Approaches. Russ. J. Phys. Chem. 95, 1269–1276 (2021). https://doi.org/10.1134/S0036024421140181
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024421140181