Abstract
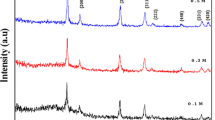
CeO2 particles of different morphologies had been successfully synthesized by a facile hydrothermal process with Ce(NO3)3⋅6H2O as cerium source, urea as precipitant and polyvinylpyrrolidone (PVP) as surfactant. The structure and optical properties of CeO2 nanocomposites were characterized by various techniques, including X-ray diffraction (XRD), scanning electron microscopy (SEM), ultraviolet-visible (UV‒Vis), photoluminescence (PL), and Raman spectroscopies. X-ray diffraction (XRD) data show that the synthesized CeO2 polymeric balls have a fluorite structure. According to the XRD, the grain size of the synthesized ceria (CeO2) becomes larger with the increase in the dosage of PVP, which is consistent with the Raman spectroscopy results. The band gaps (Eg) of cerium dioxide samples are larger than those of bulk ceria. The production of defects and the formation of oxygen vacancies can be explained as Ce4+ is reduced to Ce3+.






Similar content being viewed by others
REFERENCES
Z. U. Rehman, M. Uzair, H. T. Lim, and B. H. Koo, J. Alloys Compd. 726, 284 (2017).
M. Mazidi, R. M. Behbahani, and A. Fazeli, Appl. Catal. B: Environ. 209, 190 (2017).
H. Zhang, S. Yuan, J. L. Wang, M. C. Gong, and Y. Q. Chen, Chem Eng. J. 327, 1066 (2017).
Y. Xie, Y. Y. Yin, S. H. Zeng, M. Y. Gao, and H. Q. Su, Catal. Commun. 99, 110 (2017).
B. Dong, L. Y. Li, Z. F. Dong, R. Xu, and Y. Wu, Int. J. Hydrogen Energy 87, 1 (2017).
W. X. Zou, Y. Shao, Y. Pu, Y. D. Luo, J. F. Sun, K. L. Ma, C. J. Tang, F. Gao, and L. Dong, Appl. Catal. B: Environ. 218, 51 (2017).
Y. Ono and H. Fujii, Ceram. Int. 41, 15231 (2015).
M. E. Contreras-García, M. L. García-Benjume, V. I. Macías-Andrés, E. Barajas-Ledesma, A. Medina-Flores, and M. I. Espitia-Cabrera, Mater. Sci. Eng. B 183, 78 (2014).
J. Calvache-Muñoz, F. A. Prado, and J. E. Rodríguez-Páez, Colloids Surf., A 529, 146 (2017).
C. Zhang, F. M. Meng, and L. N. Wang, Mater. Lett. 130, 202 (2014).
H. I. Chen and H. Y. Chang, Ceram. Int. 31, 795 (2005).
F. S. Sangsefidi and M. Salavati-Niasari, Int. J. Hydrogen Energy 42, 2007 (2017).
I. A. Mudunkotuwa and V. H. Grassian, Environ. Sci. Nano 2, 429 (2015).
D. Tomova, V. Iliev, A. Eliyas, and S. Rakovsky, Sep. Purif. Technol. 156, 715 (2015).
C. C. Han, L. Yan, W. Zhao, and Z. F. Liu, Int. J. Hydrogen Energy 42, 12276 (2017).
S. Ghasemi, S. Rahman Setayesh, A. Habibi-Yangjeh, M. R. Hormozi-Nezhad, and M. R. Gholami, J. Hazard. Mater. 199–200, 170 (2012).
A. Primo, T. Marino, A. Corma, R. Molinari, and H. García, J. Am. Chem. Soc. 133, 6930 (2011).
H. Liu, M. Wang, Y. Wang, Y. Liang, W. Cao, and Y. Su, J. Photochem. Photobiol. A: Chem. 223, 157 (2011).
T. Cao, Y. Li, C. Wang, L. Wei, C. Shao, and Y. Liu, J. Sol–Gel. Sci. Technol. 55, 105 (2010).
L. Q. Jing, J. Zhou, J. R. Durrant, J. W. Tang, D. N. Liu, and H. G. Fu, Energy Environ. Sci. 5, 6552 (2012).
C. Guzmán, G. del Angel, R. Gómez, F. Galindo, R. Zanella, G. Torres, C. Angeles Chavez, and J. L. G. Fierro, J. Nano Res. 5, 13 (2009).
I. Kosacki, V. Petrovsky, H. U. Anderson, and P. J. Colomban, J. Am. Ceram. Soc. 85, 2646 (2002).
Funding
This work was supported by the Anhui Provincial Natural Science Foundation of China (1508085SME219).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fanming Meng, Wu, H. & Gao, C. Hydrothermal Synthesis of Monocrystalline CeO2 Polymeric Nano-Balls and Their Optical Properties. Russ. J. Phys. Chem. 95, 754–761 (2021). https://doi.org/10.1134/S003602442104018X
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S003602442104018X