Abstract
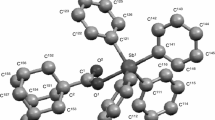
The temperature dependence of the heat capacity of triphenylantimony dibenzoate Ph3Sb(OC(O)Ph)2 is studied in the range of 6–480 K by means of precision adiabatic vacuum calorimetry and differential scanning calorimetry. The melting of the compound is observed in this temperature range, and its standard thermodynamic characteristics are identified and analyzed. Ph3Sb(OC(O)Ph)2 is obtained in a metastable amorphous state in a calorimeter. The standard thermodynamic functions of Ph3Sb(OC(O)Ph)2 in the crystalline and liquid states are calculated from the obtained experimental data: C ° p (T), H°(T)–H°(0), S°(T), and G°(T)–H°(0) for the region from T → 0 to 480 K. The standard entropy of formation of the compound in the crystalline state at T = 298.15 K is determined. Multifractal processing of the low-temperature (T < 50 K) heat capacity of the compound is performed. It is concluded that the structure of the compound has a planar chain topology.
Similar content being viewed by others
References
Guo-Cang Wang, Yong-Na Lu, Jian Xiao, et al., J. Organomet. Chem. 690, 151 (2005).
Kensuke Naka, Akiko Nakahashi, Yoshiki Chujo, Macromolecules 40, 1372 (2007).
Kensuke Naka, Akiko Nakahashi, Yoshiki Chujo, Macromolecules 39, 8257 (2006).
V. V. Sharutin, V. S. Senchurin, O. K. Sharutina, B. B. Kunkurdonova, and A. G. Burlakova, Russ. J. Inorg. Chem. 56, 197 (2011).
V. V. Sharutin, O. K. Sharutina, A. P. Pakusina, and T. P. Platonova, Russ. J. Coord. Chem. 29, 780 (2003).
Chunlin Ma, Qingfu Zhang, Jiafeng Sun, and Rufen Zhang, J. Organomet. Chem. 691, 2567 (2006).
N. N. Smirnova, I. A. Letyanina, V. N. Larina, et al., J. Chem. Thermodyn. 41, 46 (2009).
A. V. Markin, I. A. Letyanina, N. N. Smirnova, V. V. Sharutin, and O. V. Molokova, Russ. J. Phys. Chem. A 85, 1315 (2011).
I. A. Letyanina, A. V. Markin, N. N. Smirnova, A. V. Gushchin, and D. V. Shashkin, Russ. J. Phys. Chem. A 86, 1189 (2012).
A. V. Markin, I. A. Letyanina, V. A. Ruchenin, et al., J. Chem. Eng. Data 56, 3657 (2011).
I. A. Letyanina, A. V. Markin, N. N. Smirnova, et al., Vestn. NNovg. Univ., No. 3, 105 (2012).
I. A. Letyanina, A. V. Markin, N. N. Smirnova, et al., J. Chem. Eng. Data 58, 3087 (2013).
R. M. Varushchenko, A. I. Druzhinina, and E. L. Sorkin, J. Chem. Thermodyn. 29, 623 (1997).
V. M. Malyshev, G. A. Mil’ner, E. L. Sorkin, et al., Prib. Tekh. Eksp., No. 6, 195 (1985).
H. F. Flammersheim, Differential Scanning Calorimetry (Springer, Berlin, Heidelberg, 2003).
V. A. Drebushchak, J. Therm. Anal. Calorim. 79, 213 (2005).
M. E. Wieser, N. Holden, T. B. Coplen, et al., Pure Appl. Chem. 85, 1047 (2013).
S. Alford and M. Dole, J. Am. Chem. Soc. 77, 4774 (1955).
T. S. Yakubov, Dokl. Akad. Nauk SSSR 310, 145 (1990).
V. B. Lazarev, A. D. Izotov, K. S. Gavrichev, et al., Thermochim. Acta 269–270, 109 (1995).
V. V. Tarasov, Zh. Fiz. Khim. 24, 111 (1950).
V. V. Tarasov and G. A. Yunitskii, Zh. Fiz. Khim. 39, 2077 (1965).
J. P. McCullough and D. W. Scott, Calorimetry of Nonreacting Systems (Butterworth, London, 1968).
Codata Key Values for Thermodynamics, Ed. by J. D. Cox, D. D. Wagman, and V. A. Medvedev (Hemisphere, New York, 1984).
W. de Sorbo and W. W. Tyler, Acta Metall. 1, 503 (1953).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.V. Markin, N.N. Smirnova, D.V. Lyakaev, M.N. Klimova, V.V. Sharutin, O.K. Sharutina, 2016, published in Zhurnal Fizicheskoi Khimii, 2016, Vol. 90, No. 10, pp. 1439–1446.
Rights and permissions
About this article
Cite this article
Markin, A.V., Smirnova, N.N., Lyakaev, D.V. et al. Thermodynamic properties of triphenylantimony dibenzoate. Russ. J. Phys. Chem. 90, 1913–1920 (2016). https://doi.org/10.1134/S0036024416100204
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024416100204