Abstract
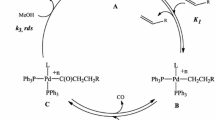
The effect of the nature of the anion in a palladium salt on a coupled process involving the oxidation of CO and hydrocarboxylation of cyclohexene in the PdX2-CuBr2-THF-H2O system was studied. The kinetics was investigated under the steady-state conditions. The kinetic isotope effects (H2O/D2O) for the rates of formation of CO2 and cyclohexanecarboxylic acid were measured. The mechanism of the coupled processes of hydrocarboxylation of cyclohexene involving the formation of palladium hydride complexes and their transformations was suggested. The kinetic model of the processes was obtained.
Similar content being viewed by others
References
L. G. Bruk, I. V. Oshanina, S. N. Gorodskii, and O. N. Temkin, Ross. Khim. Zh. 50(4), 103 (2006).
A. S. Abdullaeva, E. A. Timashova, E. Yu. Bukina, et al., Vestn. MITKhT 3(4), 63 (2008).
L. G. Bruk, O. N. Temkin, A. S. Abdullaeva, et al., Kinet. Catal. 51, 678 (2010).
O. N. Temkin, Homogeneous Catalysis with Metal Complexes. Kinetic Aspects and Mechanisms (Wiley, New York, 2012), Ch. 3, p. 239.
V. A. Likholobov and Yu. I. Ermakov, Kinet. Katal. 21, 904 (1980).
V. P. Zudin, V. A. Likholobov, and Yu. I. Ermakov, Kinet. Katal. 18, 921 (1977).
D. Bianchi, R. Bortolo, R. D’Aloisio, and M. Ricci, J. Mol. Catal. A: Chem. 150, 87 (1999).
A. S. Abdullaeva, E. A. Griva, I. V. Oshanina, et al., in Proceedings of the 6th Russian Conference on Mechanisms of Catalytic Reactions (Moscow, 2002), Vol. 2, p. 44.
L. G. Bruk, A. S. Abdullaeva, A. V. Vyrodov, et al., in Proceedings of the 14th International Symposium on Homogeneous Catalysis (Munich, Germany, 2004), p. 141.
A. S. Abdullaeva, Candidate’ Dissertation in Chemistry (Mosc. State Univ. Fine Chem. Technol., Moscow, 2005) [in Russian].
A. R. El’man, E. V. Slivinskii, and S. M. Loktev, Russ. Chem. Bull. 38, 1969 (1989).
P. Delogu and I. Donati, RF Patent No. 2125556 (1999).
Germany Patent No. DE 2916588 A1,C 0761/08 (1979).
E. A. Karakhanov, E. B. Neimerovets, V. S. Pshezhetskii, et al., USSR Inventor’s Certificate No. 1387354, Byull. Izobret. No. 18 (1989).
Chemical Encyclopedical Dictionary (Sovetsk. Entsiklopediya, Moscow, 1983) [in Russian].
E. A. Timashova, Candidate’s Dissertation in Chemistry (Mosc. State Univ. Fine Chem. Technol., Moscow, 2008).
E. A. Timashova, A. Yu. Putin, E. Yu. Bychkova, et al., Vestn. MITKhT 9, 57 (2014).
T. A. Stephenson, S. M. Morehouse, A. R. Powell, et al., J. Chem. Soc., 3632 (1965).
I. A. Efimenko, R. E. Podobedov, A. V. Churakov, et al., Russ. J. Coord. Chem. 37, 625 (2011).
A. Yu. Putin, E. Yu. Bukina, E. A. Katsman, et al., Russ. J. Phys. Chem. A 87, 465 (2013).
E. A. Katsman and A. S. Berenblyum, Software Package for Construction and Analysis of Kinetic Models and Its Application (MITKhT, Moscow, 2010) [in Russian].
I. I. Moiseev, π-Complexes in Liquid-Phase Oxidation of Olefins (Nauka, Moscow, 1970) [in Russian].
N. Yu. Kozitsyna, A. A. Bukharkina, M. V. Martens, et al., J. Organomet. Chem. 636, 69 (2001).
E. V. Gusevskaya, Quim. Nova 26, 242 (2003).
The Chemist’s Manual, Ed. by B. P. Nikol’skii (Khimiya, Moscow, Leningrad, 1965), Vol. 2 [in Russian].
A. Yu. Zakgeim, General Chemical Technology: Introduction to Modeling of Chemical Technology Processes (Universitetskaya Kniga, Logos, Moscow, 2009) [in Russian].
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.Yu. Putin, E.Yu. Bychkova, K.V. Trunilina, A.Yu. Zakgeim, E.A. Katsman, O.N. Shishilov, N.S. Akhmadullina, I.V. Oshanina, O.N. Temkin, L.G. Bruk, 2015, published in Zhurnal Fizicheskoi Khimii, 2015, Vol. 89, No. 3, pp. 373–384.
Rights and permissions
About this article
Cite this article
Putin, A.Y., Bychkova, E.Y., Trunilina, K.V. et al. Kinetics and mechanism of coupled synthesis of cyclohexanecarboxylic acid. Russ. J. Phys. Chem. 89, 359–368 (2015). https://doi.org/10.1134/S0036024415020235
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024415020235