Abstract
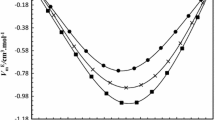
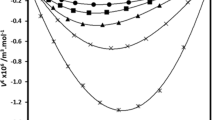
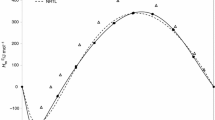
In view of industrial importance of binary {ethyl alcohol + (propan-1-ol/propan-2-ol)} mixtures, the densities (ρ) and refractive indices (n D ) of these alkanols mixtures were measured for different compositions at 303.15 K. Molar volumes (V m) and excess molar volumes (V E) of these binary mixtures were calculated from experimental density data of pure solvents and solvents mixtures. The measured refractive index and density data was used to calculate specific refractions (R D ), molar refractions (R M) and apparent molar refractions (R φ, i ) of binary mixtures. From mole fraction dependence of apparent molar refractions, the limiting apparent molar refractions (R ○φ, i ) of propan-1-ol and propan-2-ol have been determined. The graphical values of R ○φ, i for propan-1-ol and propan-2-ol were found to be 9.5664 and 7.405 cm3 mol−1 respectively. Structural changes, geometrical fittings and molecular interactions in binary mixtures of these alkanols have been discussed.
Similar content being viewed by others
References
H. Watanabe, Y. Tanji, H. Unno, and K. Hori, J. Biosci. Bioeng. 106, 226 (2008).
M. P. Pena, V. Martinez-Soria, and J. B. Monton, Fluid Phase Equilib. 166, 53 (1999).
Y. Xu, X. Chen, and Y. Zhu, Sensors 8, 1872 (2008).
H. Iloukhani, H. A. Zarei, and M. Behroozi, J. Mol. Liq. 135, 141 (2007).
H. Iloukhani, S. M. Rezaei, and H. A. Zarei, Thermochim. Acta 438, 9 (2005).
Z. Atik and K. Lourddani, J. Solution Chem. 35, 1453 (2006).
A. Pal and G. Dass, Can. J. Chem. 78, 444 (2000).
J. E. Baragi, M. I. Aralaguppi, T. M. Aminabhavi, M. Y. Kariduraganavar, and A. S. Kittur, J. Chem. Eng. Data 50, 910 (2005).
A. A. Rostami, Chaichi, J. Mohammad, and M. Sharifi, Monatsh. Chem. 138, 967 (2007).
B. Hawrylak, K. Gracie, and R. Palepu, Can. J. Chem. 76, 464 (1998).
S. L. Oswal, K. D. Prajapati, P. Oswal, N. Y. Ghael, and S. P. Ijardar, J. Mol. Liq. 116, 73 (2005).
G. I. Egorov and D. M. Makarov, J. Solution Chem. 41, 536 (2012).
J. N. Nayak, M. I. Aralaguppi, and T. M. Aminabhavi, J. Chem. Eng. Data 46, 891 (2001).
A. E. Andreatta, A. Arce, E. Rodil, and A. Soto, J. Solution Chem. 39, 371 (2010).
L. Sarkar and M. N. Roy, J. Chem. Eng. Data 54, 3307 (2009).
I. Bahadur and N. Deenadayalu, J. Solution Chem. 40, 1528 (2011).
A. Dyshin, O. V. Eliseeva, and M. G. Kiselev, Russ. J. Phys. Chem. A 86, 563 (2012).
K. L. Zhuo, Q. Liu, Y. P. Wang, Q. H. Ren, and J. J. Wang, J. Chem. Eng. Data 51, 919 (2006).
B. Sinha, V. K. Dakua, and M. N. Roy, J. Chem. Eng. Data 52, 1768 (2007).
E. Tyunina, V. Afanas’ev, and M. Chekunov, J. Solution Chem. 41, 307 (2012).
F. E. Adrian and S. Puagsa, J. Chem. Eng. Data 45, 902 (2000).
M. J. Iqbal and M. A. Chaudhry, J. Chem. Thermodyn. 41, 221 (2009).
R. P. Schutte, T. C. Liu, and L. H. Heple, Can. J. Chem. 67, 446 (1989).
J. D. Pandey, G. P. Dubey, and N. Tripathi, Proc. Ind. Acad. Sci. (Chem. Sci.) 111, 361 (1999).
S. C. Bhatia, R. Rani, J. Sangwan, and R. Bhatia, Int. J. Thermophys. 32, 1163 (2011).
R. Belda, J. V. Herraez, and O. Diez, Phys. Chem. Liquids 43, 91 (2005).
A. Arce, E. Rodii, and A. Soto, J. Solution Chem. 27, 911 (1998).
J. Szewczyk and K. Sangwal, J. Chem. Eng. Data 33, 418 (1998).
A. Arce, A. Blanco, A. Soto, and I. Vidal, J. Chem. Eng. Data 38, 336 (1993).
L. P. Safonova, A. M. Kolker, and Yu. A. Fadeeva, Russ. J. Phys. Chem. A 80, 345 (2006).
A. Arce, E. Rodil, and A. Soto, J. Chem. Eng. Data 42, 721 (1997).
T. J. Aminabhavi and B. Gopalakrishna, J. Chem. Eng. Data 40, 856 (1995)
J. N. Nayak, M. I. Aralaguppi, and T. M. Aminabhavi, J. Chem. Eng. Data 48, 1489 (2003).
D. Rudan-Tasic and C. Klovutar, Monatsh. Chem. 134, 1185 (2003).
A. Piñeiro, P. Brocos, A. Amigo, M. Pintos, and R. Bravo,, J. Solution Chem. 31, 369 (2002).
J. Canosa, A. Rodríguez, and J. Tojo, J. Chem. Thermodyn. 35, 2021 (2003).
R. J. Jiménez Riobóo, M. Philipp, M. A. Ramos, and J. K. Krüger, Eur. Phys. J. E. 30, 19 (2009).
S. D. Deosarkar, Russ. J. Phys. Chem. A 86, 1507 (2012).
S. D. Deosarkar, A. L. Puyad, P. S. Kattekar, and T. M. Kalyankar, Russ. J. Phys. Chem. A 87, 524 (2013).
S. D. Deosarkar, A. L. Puyad, and T. M. Kalyankar, Russ. J. Phys. Chem. A 86, 775 (2012).
S. Ranjbar and Momenian Seyyed Hamid, J. Chem. Eng. Data 56, 3949 (2011).
M. V. Rathnam, S. Mankumare, K. Jain, and M. S. S. Kumar, J. Solution Chem. 41, 475 (2012).
J. N. Nayak, M. I. Aralaguppi, and T. M. Aminabhavi, J. Chem. Eng. Data 48, 628 (2003).
M. N. Ray, B. K. Sarkar, and R. Chanda, J. Chem. Eng. Data 52, 1630 (2007).
M. Hasan, A. P. Hiray, U. B. Kadam, D. F. Shirude, K. J. Kurhe, and A. B. Sawant, J Solution Chem. 40, 415 (2011).
A. F. Fucaloro, J. Solution Chem. 3, 601 (2002).
A. F. Fucaloro, Y. Pu, K. Cha, A. Williams, and K. Conrad, J. Solution Chem. 36, 61 (2007).
A. Fucaloro, A. Zanella, S. Widjaja, and J. Widjaja, J. Solution Chem. 34, 1357 (2005).
S. Glasstone, Textbook of Physical Chemistry (Van Nostrand, London, 1946).
M. Tjahjono and M. Garland, J. Solution Chem. 36, 221 (2007).
Li Hui, Y. Xu Xiang, J. Chi Chun, Liu Min, Y. Di You, and Z. Sun De, J. Chem. Eng. Data 55, 2909 (2010).
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Deosarkar, S.D., Puyad, A.L. Structure and molecular interactions in {ethanol + (propan-1-ol/propan-2-ol)} mixtures at 303.15 K. Russ. J. Phys. Chem. 88, 946–950 (2014). https://doi.org/10.1134/S0036024414060090
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024414060090