Abstract—
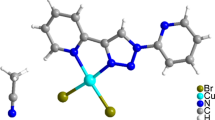
One novel distorted cubane zinc cluster, [Zn4(L)4]·(DMF)2 (1, H2L = 1-{2-Hydroxy-3-[(2- hydroxy-3-methoxy-benzylidene)-amino]-phenyl}-ethanone, DMF = N,N'-dimethyl-formamide), was obtained by room temperature synthesis. The cluster was characterized using elemental analysis, powder X‑ray diffraction, IR spectroscopy, and single-crystal X-ray diffraction. 1 is a cubane zinc cluster. The L ligand of 1 displays two different coordination modes: One is the μ3-L-κ6O1,N1,O2:O2:O2,O3 and the other is μ2-L-κ5O1,O2:O2,N1,O3 coordination modes. Hirshfeld surface analysis revealed that H···H interaction was the most abundant intermolecular interaction. The cluster emits a red fluorescence of 622 at 517 nm excite.




Similar content being viewed by others
REFERENCES
Y. J. Liu, Q. H. Li, D. J. Li, et al., Angew. Chem. Int. Ed. 60, 4849 (2021). https://doi.org/10.1002/anie.202012919
J. Liu, G. P. Yang, J. Jin, et al., Chem. Commun. 56, 2395 (2020). https://doi.org/10.1039/c9cc09664g
W. W. Zhao, J. L. Peng, W. K. Wang, et al., Coord. Chem. Rev. 377, 44 (2018). https://doi.org/10.1016/j.ccr.2018.08.023
Y. P. Wu, J. W. Tian, S. Liu, Angew. Chem. Int. Ed. 58, 12185 (2019). https://doi.org/10.1002/anie.201907136
A. Palii, S. Aldoshin, B. Tsukerblat, Coord. Chem. Rev. 426, 213555 (2021). https://doi.org/10.1016/j.ccr.2020.213555
Q. Wang, L. P.Yu, Z. Q. Zhang, et al., J. Mol. Struct. 1223, 129220 (2021). https://doi.org/10.1016/j.molstruc.2020.129220
J. M. Wang, Q. J. Deng, S.H. Zhang, Chin. J. Struct. Chem. 39, 118 (2020). https://doi.org/10.14102/j.cnki.0254-5861.2011-2395
Y. J. Zhang, Q. Chen, H. Y. Zhang, et al., J. Clust. Sci. 33, 1339 (2021). https://doi.org/10.1007/s10876-021-02064-x
Y. Zhao, X. G. Yang, X. M. Lu, et al., Inorg. Chem. 58, 6215 (2019). https://doi.org/10.1021/acs.inorgchem.9b00450
M. Y. She, Z. H. Wang, J. Chen, et al., Coord. Chem. Rev. 432, 213712 (2021). https://doi.org/10.1016/j.ccr.2020.213712
Y. M. Zeng, H. Y. Zhang, Y. J. Zhang, et al., Appl. Organomet. Chem. 34, e5712 (2020). https://doi.org/10.1002/aoc.5712
Y. H. Qiao, Y. T. Chen, S. H. Zhang, et al., Arab. J. Chem. 14, 103237 (2021). https://doi.org/10.1016/j.arabjc.2021.103237
J. Masternak, M. Zienkiewicz-Machnik, M. Kowalik, et al., Coord. Chem. Rev. 327, 242 (2016). https://doi.org/10.1016/j.ccr.2016.01.007
Y. Unver, S. Deniz, F. Çelik, et al., J. Enzyme Inhib. Med. Chem. 31, 89 (2016). https://doi.org/10.1080/14756366.2016.1206088
K. Karrouchi, L. Chemlal, J. Taoufik, et al., Ann. Pharm. Fr. 74, 431 (2016). https://doi.org/10.1016/j.pharma.2016.03.005
X. Liu, J. R. Hamon, Coord. Chem. Rev. 389, 94 (2019). https://doi.org/10.1016/j.ccr.2019.03.010
W. Zafar, S.H. Sumrra, Z.H. Chohan, Eur. J. Med. Chem. 222, 113602 (2021). https://doi.org/10.1016/j.ejmech.2021.113602
H. E. Hashem, E. A. Mohamed, A. A. Farag, et al., Appl Organomet. Chem. 35, e6322 (2021). https://doi.org/10.1002/aoc.6322
V. K. Singh, R. Kadu, H. Roy, et al., Dalton Trans. 45, 1443 (2016). https://doi.org/10.1039/c5dt03407h
Y. Sun, Y. L. Lu, M. L. Bian, et al., Eur. J. Med. Chem. 211, 113098 (2021) https://doi.org/10.1016/j.ejmech.2020.113098
H.Y. Zhang, Y. Li, W. Wang, et al., J. Coord. Chem. 69, 1938 (2016). https://doi.org/10.1080/00958972.2016.1195493
Y. Bunno, Y. Tsukimawashi, M. Kojima, et al., ACS Catal. 11, 2663 (2021). https://doi.org/10.1021/acscatal.0c05261
X. J. Li, S. Q. Xie, Y. Z. Hu, et al., Chem. Commun. 57, 2392 (2021). https://doi.org/10.1039/d0cc08403d
S. Kumari, A. Ramesh, B. Das, et al., Inorg. Chem. Front. 8, 1553 (2021). https://doi.org/10.1039/d0qi01190h
M. T. Kaczmarek, M. Zabiszak, M. Nowak, et al., Coord. Chem. Rev. 370, 42 (2018). https://doi.org/10.1016/j.ccr.2018.05.012
S. H. Sumrra, A. Suleman, Z. H. Chohan, et al., Russ. J. Gen. Chem. 87, 1281 (2017). https://doi.org/10.1134/S107036321706024X
M. Hanif, Z. H. Chohan, Spectrochim. Acta A Mol. Biomol. Spectrosc. 104, 468 (2013). https://doi.org/10.1016/j.saa.2012.11.077
M. Fondo, N. Ocampo, A. M. García-Deibe, et al., Dalton Trans. 23, 3785 (2005). https://doi.org/10.1039/b507068f
Y. T. Chen, S. N. Zhang, Y. Xiao, et al., Acta Cryst. C76, 236 (2020). https://doi.org/10.1107/S2053229620001850
S. H. Zhang, N. Li, C. M. Ge, et al., Dalton Trans. 40, 3000 (2011). https://doi.org/10.1039/c0dt01498b
L. Yang, S. H. Zhang, W. Wang, et al., Polyhedron 74, 49 (2014). https://doi.org/10.1016/j.poly.2014.02.024
A. Beheshti, W. Clegg, V. Nobakht, et al., Cryst. Growth Des. 13, 1023 (2013). https://doi.org/10.1021/cg301106g
S. Toki, J. Che, L. X. Rong, et al., Macromolecules 46, 5238 (2013). https://doi.org/10.1021/ma400504k
X. G. Yang, Z. M. Zhai, X. M. Lu, et al., Dalton Trans. 48, 10785 (2019). https://doi.org/10.1039/c9dt02178g
L. Y. Xin, G. Z. Liu, X. L. Li, et al., Cryst. Growth Des. 12, 147 (2012). https://doi.org/10.1021/cg200903k
L. F. Ma, X. Q. Li, L. Y. Wang, et al., CrystEngComm 13, 4625 (2011). https://doi.org/10.1039/c1ce05308f
S. E. Korolenko, V. V. Avdeeva, E. A. Malinina, N. T. Kuznetsov, Russ. J. Inorg. Chem. 66, 1350 (2021). https://doi.org/10.1134/S0036023621090047
M. A. Uvarova, S. E. Nefedov, Russ. J. Inorg. Chem. 66, 1837 (2021). https://doi.org/10.1134/S0036023621120202
V. V. Avdeeva, S. E. Korolenko, E. A. Malinina, et al., Russ. J. Inorg. Chem. 92, 393 (2022). https://doi.org/10.1134/S1070363222030070
Y. B. Ivanova, T. V. Shorokhova, Y. V. Khrushkova, Russ. J. Inorg. Chem. 92, 1290 (2022). https://doi.org/10.1134/S1070363222070180
M. S. Milutka, A. S. Burlov, V. G. Vlasenko, et al., Russ. J. Inorg. Chem. 92, 1297 (2022). https://doi.org/10.1134/S1070363222070192
S. H. Zhang, L. F. Ma, H. H. Zou, et al., Dalton Trans. 40, 11402 (2011). https://doi.org/10.1039/c1dt10517e
Y. Zhao, L. L. Zhai, J. Fan, et al., Polyhedron 46, 16 (2012). https://doi.org/10.1016/j.poly.2012.07.079
S. H. Zhang, R. X. Zhao, G. Li, et al., RSC Adv. 4, 54837 (2014). https://doi.org/10.1039/c4ra09687h
L. S. Long, CrystEngComm 12, 1354 (2010). https://doi.org/10.1039/b921146b
S. M. Zhang, Q. S. Deng, H. Y. Zhang, et al., J. Cluster Sci. 31, 685 (2020). https://doi.org/10.1007/s10876-019-01675-9
G. M. Sheldrick, Acta Cryst. C71, 3 (2015). https://doi.org/10.1107/S2053273314026370
O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, et al., J. Appl. Cryst. 42, 339 (2009). https://doi.org/10.1107/S0021889808042726
J. J. McKinnon, M. A. Spackman, A. S. Mitchell, Acta Cryst. B60, 627 (2004). https://doi.org/10.1107/S0108768104020300
F. H. Allen, O. Kennard, D. G. Watson, et al., J. Chem. Soc. Perkin Trans. 2. S1 (1987). https://doi.org/10.1039/P298700000S1
C. Agnew, K. B. Dillon, A. E. Goeta, et al., Inorg. Chim. Acta 362, 4260 (2009). https://doi.org/10.1016/j.ica.2009.05.063
S. K. Patel, R.N. Patel, Y. Singh, et al., Polyhedron. 161, 198 (2019). https://doi.org/10.1016/j.poly.2019.01.006
S. H. Zhang, C. Feng, J. Mol. Struct. 977, 62 (2010). https://doi.org/10.1016/j.molstruc.2010.05.010
G. H. Wei, J. Yang, J. F. Ma, et al., Dalton Trans. 23, 3080 (2008). https://doi.org/10.1039/b716657e
S. T. Wang, X. Zheng, S. H. Zhang, et al., CrystEngComm 23, 4059 (2021). https://doi.org/10.1039/d1ce00323b
S. H. Zhang, J. M. Wang, H. Y. Zhang, et al., Dalton Trans. 46, 410 (2017). https://doi.org/10.1039/c6dt04059d
S. E. Korolenko, K. P. Zhuravlev, V. I. Tsaryuk, et al., J. Lumin. 237, 118156 (2021). https://doi.org/10.1016/j.jlumin.2021.118156
P. Huo, T. Chen, J. L. Hou, et al., Inorg. Chem. 55, 6496 (2016). https://doi.org/10.1021/acs.inorgchem.6b00571
C. N. Tsai, S. Mazumder, X. Z. Zhang, et al., Inorg. Chem. 55, 7341 (2016). https://doi.org/10.1021/acs.inorgchem.6b00374
Funding
This work was supported by the National Natural Science Foundation of China (contract no. 21861014) and Talent Introduction Project of Guangdong University of Petrochemical Technology (no. 2020rc033).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflicts of interest.
Supplementary Information
Rights and permissions
About this article
Cite this article
Tao Zhou, Zhang, H., Zhang, SH. et al. Room Temperature Synthesis, Crystal Structure, Hirshfeld Surface Analysis, and Fluorescence Properties of One Novel Cubane Zinc Cluster Based on 1-{2-Hydroxy-3-[(2-Hydroxy-3-Methoxy-Benzylidene)-Amino]-Phenyl}-Ethanone. Russ. J. Inorg. Chem. 67 (Suppl 1), S35–S41 (2022). https://doi.org/10.1134/S0036023622601817
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023622601817