Abstract
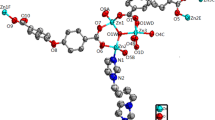
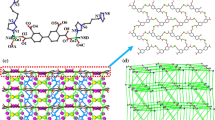
A Zn(II) coordination polymer, {[Zn(ca)(bpe)]·0.5H2O}n (H2ca = citraconic acid, bpe = 1,2-bi(4-pyridyl)ethylene) has been prepared by hydrothermal reaction, and characterized by single-crystal X-ray diffraction, elemental analysis, FT IR, PXRD, and TGA. Title compound exhibits a three-dimensional metal-organic framework with lvt topology type and 42⋅84 Schläfli symbol. The compound has good phase purity and high thermal stability. The emission spectrum of the complex is similar to that of the powdered free bpe molecule, but the obvious red-shift may be due to the coordination of bpe with Zn ions, resulting in a reduced nonradiative decay of the intraligand excited state. The center-to-center olefin bond distance of bpe ligand in the compound, 3.620 Å, is suitable to undergo [2+2] cycloaddition reaction. The 1H NMR spectrum of the cycloaddition product after UV irradiation for 24 h shows that the double bonds were partially photodimerized (82.6%) into the rctt-tpcb and rtct-tpcb derivatives.




Similar content being viewed by others
REFERENCES
S. Kitagawa and K. Uemera, Chem. Soc. Rev. 34, 109 (2005). https://doi.org/10.1039/B313997M
S. M. Oburn, D. C. Swenson, S. V. S. Mariappan, et al., J. Am. Chem. Soc. 139, 8452 (2017). https://doi.org/10.1021/jacs.7b04420
K. M. Hutchins, J. C. Sumrak, and L. R. MacGillivray, Org. Lett. 16, 1052 (2014). https://dx,doi.org/https://doi.org/10.1021/ol4035403
R. Medishetty, I. H. Park, S. S. Lee, et al., Chem. Commun. 52, 3989 (2016). https://doi.org/10.1002/chin.201616208
R. Medishetty, R. Tandiana, L. L. Koh, et al., Chem. Eur. J. 20, 1231 (2014). https://doi.org/10.1002/chem.201304246
M. M. Gan, J. G. Yu, Y. Y. Wang, et al., Cryst. Growth Des. 18, 553 (2018). https://doi.org/10.1021/acs.cgd.7b01308
W. J. Gong, Z. G. Ren, H. X. Li, et al., Cryst. Growth Des. 17, 870 (2017). https://doi.org/10.1021/acs.cgd.6b01728
F. L. Hu, Y. Mi, C. Zhu, et al., Angew. Chem. Int. Ed. 57, 12696 (2018). https://doi.org/10.1002/anie.201806076
T. D. Hamilton, G. S. Papaefstathiou, L. R. MacGillivray, J. Am. Chem. Soc. 124, 11606 (2002). https://doi.org/10.1021/ja0278281
M. Nagarathinam and J. J. Vittal, Chem. Commun. 438 (2008). https://doi.org/10.1039/b713586f
G. S. Papaefstathiou, I. G. Georgiev, T. Friščić, et al., Chem. Commun. 3974 (2005). https://doi.org/10.1039/b504477d
M. H. Mir, L. L. Koh, G. K.Tan, et al., Angew. Chem. Int. Ed. 49, 390 (2010). https://doi.org/10.1002/anie.200905898
G. S. Papaefstathiou, Z. M. Zhong, L. Geng, et al., J. Am. Chem. Soc. 126, 9158 (2004). https://doi.org/10.1021/ja047819n
G. L. Li, W. D. Yin, G. Z. Liu, et al., Inorg. Chem. Commun. 43, 165 (2014). https://doi.org/10.1016/j.inoche.2014.02.037
A. M. P. Peedikakkal, L. L. Koh, J. J. Vittal, Chem. Commun. 441 (2008). https://doi.org/10.1039/b714355a
G. L. Li, G. Z. Liu, L. F. Ma, et al., Chem. Commun. 50, 2615 (2014). https://doi.org/10.1039/c3cc49106d
R. Medishetty, R. Tandiana, J. Wu, et al., Chem. Eur. J. 21, 11948 (2015). https://doi.org/10.1002/chem.201501649
S. Y. Yang, X. L. Deng, R. F. Jin, et al., J. Am. Chem. Soc. 136, 558 (2014). https://doi.org/10.1021/ja409272p
O. V. Dolomanov, L. J. Bourhis, R. J. Gildea, et al., J. Appl. Crystallogr. 42, 339 (2009). https://doi.org/10.1107/S0021889808042726
G. M. Sheldrick, Acta Crystallogr. A71, 3 (2015). https://doi.org/10.1107/S2053273314026370
G. M. Sheldrick, Acta Crystallogr. C71, 3 (2015). https://doi.org/10.1107/S2053229614024218
S. E. Korolenko, V. V. Avdeeva, E. A. Malinina, et al., Russ. J. Inorg. Chem. 66, 1350 (2021). https://doi.org/10.1134/S0036023621090047
M. H. Zuo, J. Zhou, J. H. Yu, et al., Russ. J. Inorg. Chem. 66, 982 (2021). https://doi.org/10.1134/S0036023621070159
S. E. Korolenko, K. P. Zhuravlev, V. I. Tsaryuk, et al., J. Lumin. 237, 118156 (2021). https://doi.org/10.1016/j.jlumin.2021.118156
D. Ureche, A. Rija, I. Bulhac, et al., Russ. J. Inorg. Chem. 65, 1838 (2021). https://doi.org/10.1134/S0036023620120189
L. L. Li, S. S. Feng, J. Q. Zhang, et al., Russ. J. Gen. Chem. 91, 1128 (2021). https://doi.org/10.1134/S1070363221060220
J. Buasakuna, P. Srilaoonga, K. Chainokb, et al., Inorg. Chim. Acta 511, 119839 (2020). https://doi.org/10.1016/j.ica.2020.119839
A. Halder, B. Bhattacharya, F. Haque, et al., Cryst. Growth Des. 17, 6613 (2017). https://doi.org/10.1021/acs.cgd.7b01270
G. L. Li, W. D. Yin, Q. L. Liu, et al., Chin. J. Inorg. Chem. 35, 2355 (2019). https://doi.org/10.11862/CJIC.2019.271
Y. P. Li, F. Y. Ju, G. L. Li, et al., Russ. J. Coord. Chem. 44, 214 (2018). https://doi.org/10.1134/S1070328418030028
J. Tao, M. L. Tong, J. X. Shi, et al., Chem. Commun. 20, 2043 (2000). https://doi.org/10.1039/B005753N
L. Y. Xin, G. Z. Liu, L. F. Ma, et al., Aust. J. Chem. 68, 758 (2015). https://doi.org/10.1071/CH14347
Y. C. Sun, Q. Q. Wei, Q. X. Wang, et al., Russ. J. Coord. Chem. 48, 60 (2022). https://doi.org/10.1134/S1070328422010067
V. F. Shul’gin, M. A. Kiskin, A. N. Gusev, et al., Russ. J. Coord. Chem. 47, 326 (2021). https://doi.org/10.1134/S1070328421050079
G. Z. Liu, S. H. Li, X. L. Li, et al., CrystEngComm 15, 4571 (2013). https://doi.org/10.1039/C3CE40109J
T. Caronna, R. Liantonio, T. A. Logothetis, et al., J. Am. Chem. Soc. 126, 4500 (2004). https://doi.org/10.1021/ja039884n
L. R. MacGillivray, J. F. Reid, J. A. Ripmeester, et al., Ind. Eng. Chem. Res. 41, 4494 (2002). https://doi.org/10.1021/ie010775w
Funding
This work was supported by the National Natural Science Foundation of China (project no. 21571093).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Supplementary Information
Rights and permissions
About this article
Cite this article
Gui-Lian Li, Yin, WD., Zhang, JY. et al. A 3D Zn(II) Metal-Organic Framework Based on Citraconic Acid and 1,2-Bi(4-pyridyl)ethylene Mixed Ligands: Crystal Structure, Luminescence, and [2+2] Cycloaddition Reaction. Russ. J. Inorg. Chem. 67, 1745–1750 (2022). https://doi.org/10.1134/S0036023622600800
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023622600800