Abstract
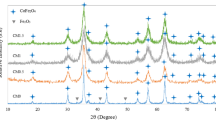
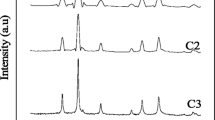
A facile and rapid method easily reproducible in ordinary laboratory settings is proposed for preparing nanosized cobalt ferrite powders, where the precipitate used is a strongly basic anion exchange resin in the OH form. The effects caused by additions of polysaccharides having various chain natures and various molar weights on the composition, yield, and particle size of the deposition product are studied. Suggested mechanisms underlying these effects are studied. This method makes it possible to appreciably reduce the precursor annealing temperature compared to that in the state-of-art methods for preparing similar oxide systems due to the formation of highly reactive precursors. Optimal conditions are found to prepare stoichiometric precursors such that, once annealed at 600°C, would form a CoFe2O4 pure phase with particle sizes of 10–20 nm. The stability of hydrosols of the thus-prepared cobalt ferrite nanoparticles is studied by dynamic and electrophoretic light scattering. The prepared material is found to be useful for the design of magnetic core–shell hybrid nanostructures.






Similar content being viewed by others
REFERENCES
U. Sitize and H. Sato, Ferrites (Mir, Moscow, 1964 (translated from Japanese) [in Russian].
I. C. Covaliu, J. Neamtua, G. Georgescua, et al., J. Nanomaterials Biostructures 6, 245 (2011).
A. A. Il’in, A. P. Il’in, N. N. Smirnov, et al., Khim. Khim. Tekhnol. 53, 86 (2010).
V. M. Talanov, Pat. 2010135592/05, Byull. Izobret., No. 23 (2013).
N. Rezlescu, E. Rezlescu, F. Tudorache, et al., Roman. Repts Phys. 61, 223 (2009).
M. Kanagaraj, P. Sathishkumar, G. K. Selvan, et al., Indian J. Pure Appl. Phys. 52, 124 (2014).
T. George, S. Joseph, A. T. Sunny, et al., J. Metastable Nanocryst. Mater., No. 23, 41 (2005).
I. V. Kasy Viswanath, Y. L. N. Murthy, et al., Int. J. Chem. Sci. 11, 64 (2013).
I. V. Lisnevskaya, I. A. Bobrova, A. V. Petrova, et al., Russ. J. Inorg. Chem. 57, 474 (2012).
I. V. Lisnevskaya, I. A. Bobrova, and T. G. Lupeiko, J. Magn. Magn. Mater. 51, 507 (2017).
N. Rezlescu, E. Rezlescu, F. Tudorache, et al., Roman. Repts in Phys. 61, 223 (2009).
S. S. Kader, D. P. Paul, S. M. Hoque, et al., Int. J. Mater., Mechan. Manuf. 2, 5 (2014).
S. V. Saikova, M. V. Panteleeva, and R. B. Nikolaeva, Russ. J. Appl. Chem. 75, 1787 (2002).
G. L. Pashkov, C. V. Saikova, M. V. Panteleeva, et al., Zh. Siber. Fed. Univ., Ser. Khim. 2, 150 (2009).
G. L. Pashkov, S. V. Saikova, M. V. Panteleeva, et al., Theor. Found. Chem. Eng. 48, 671 (2014).
G. L. Pashkov, S. V. Saikova, M. V. Panteleeva, et al., Steklo Keram. 71, 19 (2014).
A. I. Vulikh, Ion Exchange (Khimiya, Moscow, 1973) [in Russian].
S. V. Saikova, E. A. Kirshneva, M. V. Panteleeva, et al., Russ. J. Inorg. Chem. 64, 1191 (2019). https://doi.org/10.1134/S0036023619100127
S. A. Shapiro, Analytical Chemistry (Vysshaya Shkola, Moscow, 1973) [in Russian].
Powder Diffraction File (JCPDS), Nos. 43-1458; 13-398.
Powder Diffraction File (JCPDS), No. 24-0072.
Powder Diffraction File (JCPDS), No. 01-1152.
S. V. Saikova, G. L. Pashkov, and M. V. Panteleeva, A Monograph (Sib. Feder. Univ., Krasnoyarsk, 2018) [in Russian].
T. V. Trofimova, A. Yu. Pavlikov, and S. V. Saikova, Vestn. Permsk. Univ., Ser. Khim. 7, 137 (2017).
T. Itano, J. Japan Soc. Air Poll. 18, 595 (1983). https://doi.org/10.11298/taiki1978.18.595
L. M. Amreeva, G. Z. Zainelova, and B. Z. Medeubaeva, Meditsina 159 (9), 42 (2015).
G. L. Pashkov, S. V. Saikova, and M. V. Panteleeva, Theor. Found. Chem. Eng. 50 (4), 575 (2016). https://doi.org/10.1134/S0040579516040254
G. S. Nikolic and M. D. Cakic, Colloid J. 69, 464 (2007). https://doi.org/10.1134/S1061933X07040084
K. Nakamoto, Infrared and Raman Spectra of Inorganic and Coordination Compounds (Wiley, New York, 1986; Mir, Moscow, 1991).
T. Kawaguchi and M. Hasegawa, J. Mater. Sci.: Mater. Med. 11, 31 (2017). https://doi.org/10.1023/A:1008933601813
ACKNOWLEDGMENTS
Instruments of the Shared Facilities Centers of the Cola Scientific Center of the Russian Academy of Sciences, and the Siberian Federal University were used in this study.
Funding
This study was financially supported by the Russian Foundation for Basic Research, the Government of the Krasnoyarsk Territory, and the Krasnoyarsk Regional Science Fund in the frame of project no. 18-43-243014 entitled “Synthesis of Magnetic Core–Gold Shell Nanoparticles for Biomedical and Catalytic Applications.”
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by O. Fedorova
Rights and permissions
About this article
Cite this article
Saikova, S.V., Trofimova, T.V., Pavlikov, A.Y. et al. Effect of Polysaccharide Additions on the Anion-Exchange Deposition of Cobalt Ferrite Nanoparticles. Russ. J. Inorg. Chem. 65, 291–298 (2020). https://doi.org/10.1134/S0036023620030110
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023620030110