Abstract
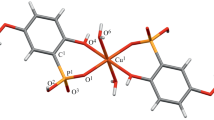
Dissociation and complexation with Cu(II) cation is studied for phosphoryl-containing podands (tetrabasic 1,5-bis[2-(dioxyphosphoryl)-4-ethylphenoxy]acidic-type-3-oxapentane (H4LP) and dibasic 1,5-bis[2-(oxyethoxyphosphoryl)-4-ethylphenoxy]-pentane (H2LP)) and their carbonyl analog (polyether dibasic acid 1,5-bis[2-(oxycarbonylphenoxy)]oxapentane (H2LC) in water in the presence of 5% dimethylformamide potentiometrically, spectrophotometrically, and conductometrically. Phosphoryl podands exhibit good complexing properties and can selectively bind metal cations. The dissociation constants are determined and the distribution diagram of ionized species of the compounds under study depending on pH is plotted. The molar ratio between the metal and podand in all complexes is found to be 1 : 1. The stability constants of complexes with Cu2+ cation are calculated. The most stable copper(II) complexes are formed with the tetrabasic podand.
Similar content being viewed by others
References
M. Wehbe, A. W. Y. Leung, M. J. Abrams, et al., Dalton Trans. 46, 10758 (2017). doi https://doi.org/10.1039/c7dt01955f
T. J. Wadas, E. H. Wong, G. R. Weisman, and C. J. Anderson, Chem. Rev. 110, 2858 (2010). doi https://doi.org/10.1021/cr900325h
C. S. Cutler, H. M. Hennkens, N. Sisay, et al., Chem. Rev. 113, 858 (2013). doi https://doi.org/10.1021/cr3003104
Adv. Drug. Delivery Rev. 60, 1347 (2008). doi https://doi.org/10.1016/j.addr.2008.04.006
Mohd Zulkefeli, Asami Suzuki, Motoo Shiro, et al. Inorg. Chem. 50, 10113 (2011). doi https://doi.org/10.1021/ic201072q
E. Kimura, Bull. Jpn. Soc. Coord. Chem. 59, 26 (2012). doi https://doi.org/10.4019/bjscc.59.26
T. Meierhofer, I. C. Rosnizeck, T. Graf, et al., J. Am. Chem. Soc. 133, 2048 (2011). doi https://doi.org/10.1021/ja108779j
V. C. Pierre, S. M. Harris, and S. L. Pailloux, Acc. Chem. Res. 51, 342 (2018). doi https://doi.org/10.1021/acs.accounts.7b00301
S. M. Harris, K. Srivastava, A. B. League, et al., Dalton Trans. 47, 2202 (2018). doi https://doi.org/10.1039/C7DT04203E
R. Puranik, S. Bao, A. M. Bonin, et al., Cell. Biosci. 6, 1 (2016). doi https://doi.org/10.1186/s13578-016-0076-8
T. J. Hubin, N.-A. Prince, K. D. Roewe, et al., Bioorg. Med. Chem. 22, 3239 (2014). doi https://doi.org/10.1016/j.bmc.2014.05.003
D. E. C. Corbridge, Phosphorus: Chemistry, Biochemistry and Technology (CRC Press, New York, 2013).
C. Hill, A Series of Advances, Vol. 19: Ion Exchange and Solvent Extraction, Ed. by B. A. Moyer (CRC Press, Boca Raton, FL, 2010).
K. L. Nash, R. E. Barrans, R. Chiarizia, et al., Solv. Extr. Ion Exch. 18, 605 (2000) https://doi.org/10.1080/07366290008934700.
E. O. Otu and R. Chiarizia, Solv. Extr. Ion Exch. 19, 885 (2001).
E. O. Otu and R. Chiarizia, Solv. Extr. Ion Exch. 19, 1017 (2001).
S. Nishihama, R. P. Witty, L. R. Martin, and K. L. Nash, Solv. Extr. Ion Exch. 31, 370 (2013) https://doi.org/10.1080/07366299.2013.800404.
P. R. Zalupski, D. R. McAlister, D. C. Stepinski, and A. W. Herlinger, Solv. Extr. Ion Exch. 21, 331 (2003).
M. B. Ali, A. Ya, B. H. Ahmed, et al., Solv. Extr. Ion Exch. 30, 469 (2012).
Q. Fu, L. Yang, and Q. Wang, Talanta 72, 1248 (2007). doi https://doi.org/10.1016/j.talanta.2007.01.015
G. J. Lumetta, S. I. Sinkov, J. A. Krause, and L. E. Sweet, Inorg. Chem. 55, 1633 (2016). doi https://doi.org/10.1021/acs.inorgchem.5b02524
N. K. Batchu, T. V. Hoogerstraete, D. Banerjee, and K. Binnemans, RSC Adv. 72, 45351 (2017). doi https://doi.org/10.1039/C7RA09144C
I. O. Shatrava, V. A. Ovchynnikov, K. E. Gubina, et al., Polyhedron 139, 98 (2018). doi https://doi.org/10.1016/j.poly.2017.09.038
T. I. Ignat’eva, V. E. Baulin, E. N. Tsvetkov, and O. A. Raevskii, Zh. Obshch. Khim. 60, 1503 (1990).
A. M. Safiulina, A. G. Matveeva, D. V. Ivanets, et al., Izv. Akad. Nauk, Ser. Khim., No. 1, 161 (2015). doi https://doi.org/10.1007/s11172-015-0837-2
A. M. Safiulina, A. G. Matveeva, D. V. Ivanets, et al., Izv. Akad. Nauk, Ser. Khim., No. 1, 169 (2015). doi https://doi.org/10.1007/s11172-015-0838-1
A. N. Turanov, V. K. Karandashev, V. E. Baulin, and A. Yu. Tsivadze, Radiokhimiya 56, 22 (2014). doi https://doi.org/10.1134/S1066362214010056
V. E. Baulin, O. V. Kovalenko, A. N. Turanov, et al., Radiokhimiya, 57 (2015). doi https://doi.org/10.1134/S1066362215010099
V. E. Baulin, O. V. Kovalenko, D. V. Baulin, et al., Fizikokhim. Poverkhn. Zashchita Mater. 52, 604 (2016). doi https://doi.org/10.1134/S2070205116060083
A. M. Safiullina, D. V. Ivanets, E. M. Kudryavtsev, et al., Russ. J. Inorg. Chem. 63, 1679 (2018). doi https://doi.org/10.1134/S00360236
N. M. Dyatlova, V. Ya. Temkina, and I. D. Kolpakova, Comlexons (Khimiya, Moscow, 1970) [in Russian].
V. V. Belova, N. S. Egorova, A. A. Voshkin, et al., Theor. Found. Chem. Eng. 49, 545 (2015). doi https://doi.org/10.1134/S0040579515040041
V. E. Baulin, M. A. Kiskin, and I. S. Ivanova, Russ. J. Inorg. Chem. 57, 671 (2012). doi https://doi.org/10.1134/S0036023612050038
E. G. Polle, Russ. Chem. Rev. 43, 1337 (1974).
L. I. Bulatov and I. P. Kalinkin, Manual for Photometric Analyses (Khimiya, Leningrad, 1986) [in Russian].
Acknowledgments
This study was conducted under State Assignment 2018 (topic no. 0090-2017-0024) and supported in part by the Program no. 34 of the Presidium of the Russian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Al Ansari, Y.F., Baulin, V.E. 1,5-Bis[2-(Dioxyphosphoryl)-4-ethylphenoxy]-3-oxapentane and Its Analogs: Acidity and Complexation in Aqueous Media Containing Copper(II) Cation. Russ. J. Inorg. Chem. 64, 550–555 (2019). https://doi.org/10.1134/S0036023619040028
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036023619040028