Abstract
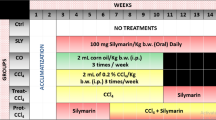
Carbon tetrachloride is a well-studied hepatotropic poison. Animal models of exposure to carbon tetrachloride resemble acute liver damage in humans. This paper presents the study of the expression of genes related to cell cycle control, apoptosis, and oxidative stress in a model of carbon tetrachloride-induced toxic hepatitis in rats. White mongrel male rats were injected with a 50% oil solution of carbon tetrachloride at a dose of 0.125–4.000 g/kg (experimental group) or olive oil (control group). The animals were decapitated 24 and 72 h after the administration of carbon tetrachloride, and the qRT-PCR expression levels of the genes encoding hemoxygenase-1 (Hmox1), cell cycle checkpoint kinase-1 (Chek1), and caspase-7 (Casp7) in the liver were analyzed. The increase in the expression levels of Hmox1 and Chek1 after exposure was detected. These genes may either play a role in promoting pathological oxidative stress in the liver or be a part of a stress response. We have concluded that the major pathway of the liver damage in carbon tetrachloride exposed animals is necrosis rather than apoptosis.



Similar content being viewed by others
REFERENCES
Valeeva E.T. 2011. Professional toxic lesions of the liver in liquid fuel production workers. Zdravookhr. Ross. Federatsii. 4, 50–51.
Kosarev V.V., Babanov S.A. 2009. Toxic (toxic-allergic) hepatitis. Gastroenterologiya. Suppl. to Consilium Medicum. 2, 29–30.
Vladimirov Yu.A. 2000. Free radicals in biological systems. Soros. Obraz. Zh. 12, 13–19.
Golikov S.N., Sanotskii I.V., Tiunov L.A. (1986) Obshchie mekhanizmy toksicheskogo deistviya (General Mechanisms of Toxic Action). Leningrad: Meditsina.
Kravchenko L.V, Trusov N.V., Uskova M.A., Aksenov I.B., Avren’eva L.I., Guseva G.V., Vasil’eva M.A., Selifanov A.V., Tutel’yan V.A. 2009. Characteristics of acute toxic action of carbon tetrachloride as a model of oxidative stress. Toksikol. Vestn. 1, 12–18.
Weber L.W., Boll M., Stampfl A. 2003. Hepatotoxity and mechanism of action of haloalcanes: Carbon tetrachloride as a toxicological model. Crit. Ref. Toxicol. 33, 105–136.
Li Volti G., Sacerdoti D., Di Giacomo C., Barcellona M.L., Scacco A., Murabito P., Biondi A., Basile F., Gazzolo D., Abella R., Frigiola A., Galvano F. 2008. Natural heme oxygenase-1 inducers in hepatobiliary function. World J. Gastroenterol. 14, 6122–6132.
Wen T., Guan L., Zhang Y.L., Zhao J.Y. 2006. Dynamic changes of heme oxygenase-1 and carbon monoxide production in acute liver injury induced by carbon tetrachloride in rats. Toxicology. 228, 51–57.
Abraham N.G., Kappas A. 2008. Pharmacological and clinical aspects of heme oxygenase. Pharmacol. Rev. 60, 79–127.
Soares M.P., Bach F.H. 2009. Heme oxygenase-1: From biology to therapeutic potential. Trends Mol. Med. 15, 50–58.
Chinnaiyan A.M., Dixit V.M. 1996. The cell-death machine. Curr. Biol. 6, 555–562.
Vladimirskaya E.B. 2002. Mechansisms of apoptotic cell death. Gematol. Transfuziol. 47, 35–40.
Ryzhov S.V., Novikov V.V. 2002. Molecular mechanisms of apoptotic processes. Ross. Bioterapevt. Zh. 1, 27–33.
Lamkanfi M., Kanneganti T.D. 2010. Caspase-7: A protease involved in apoptosis and inflammation. Int. J. Biochem. Cell Biol. 42, 21–24.
Brown E.J., Baltimore D. 2003. Essential and dispensable roles of ATR in cell cycle arrest and genome maintenance. Genes Dev. 17, 615–628.
Liu Q., Guntuku S., Cui X.S., Matsuoka S., Cortez D., Tamai K., Luo G., Carattini-Rivera S., DeMayo F., Bradley A., Donehower L.A., and Elledge S.J. 2000. Chk1 is an essential kinase that is regulated by Atr and required for the G2/M DNA damage checkpoint. Genes Dev. 14, 1448–1459.
Zhou B.B., Elledge S.J. 2000. The DNA damage response: Putting checkpoints in perspective. Nature. 408, 433–439.
Hiom K. 2005. DNA repair: How to PIKK a partner. Curr. Biol. 15. 473–475.
Belousova Yu.B. (2005) Eticheskaya ekspertiza biomedi-tsinskikh issledovanii: prakticheskie rekomendatsii (Ethical Expert Evaluation of Biomedical Research: Practical Recommendations). Moscow: Oki.
Jais A., Einwallner E., Sharif O., Gossens K., Lu T.T., Soyal S.M., Medgyesi D., Neureiter D., Paier-Pourani J., Dalgaard K., Duvigneau J.C., Lindroos-Christensen J., Zapf T.C., Amann S., Saluzzo S., et al. 2014. Heme oxygenase-1 drives metaflammation and insulin resistance in mouse and man. Cell. 158, 25–40.
Jou J., Choi S., Diehl A. 2008. Mechanisms of disease progression in nonalcoholic fatty liver disease. Semin. Liver Dis. 28, 370–379.
Yao P., Li K., Song F., Zhou S., Sun X., Zhang X., Nussler A.K., Liu L. 2007. Heme oxygenase-1 upregulated by Ginkgo biloba extract: Potential protection against ethanol-induced oxidative liver damage. Food Chem. Toxicol. 45, 1333–1342.
Randle L.E., Goldring C.E., Benson C.A., Metcalfe P.N., Kitteringham N.R., Park B.K., Williams D.P. 2008. Investigation of the effect of a panel of model hepatotoxins on the Nrf2-Keap1 defence response pathway in CD-1 mice. Toxicology. 243, 249–260.
Conde de la Rosa L., Vrenken T.E., Hannivoort R.A., Buist-Homan M., Havinga R., Slebos D.J., Kauffman H.F., Faber K.N., Jansen P.L., Moshage H. 2008. Carbon monoxide blocks oxidative stress-induced hepatocyte apoptosis via inhibition of the p54 JNK isoform. Free Radic. Biol. Med. 44, 1323–1333.
Beck H., Nähse V., Larsen M.S., Groth P., Clancy T., Lees M., Jørgensen M., Helleday T., Syljuåsen R.G., Sørensen C.S. 2010. Regulators of cyclin dependent kinases are crucial for maintaining genome integrity in S phase. J. Cell Biol. 188, 629–638.
Cerqueira A., Santamaría D., Martínez-Pastor B., Cuadrado M., Fernández-Capetillo O., Barbacid M. 2009. Overall Cdk activity modulates the DNA damage response in mammalian cells. J. Cell Biol. 187, 773–780.
Zaridze D.G. (2004) Kantserogenez (Carcinogenesis). Moscow: Meditsina.
Brown E.J., Baltimore D. 2003. Essential and dispensable roles of ATR in cell cycle arrest and genome maintenance. Genes Dev. 17, 615–628.
Morgan D. 2007. The Cell Cycle: Principles of Control. London: New Science Press.
Reinhardt H.C., Yaffe M.B. 2009. Kinases that control the cell cycle in response to DNA damage: Chk1, Chk2, and MK2. Curr. Opin. Cell Biol. 21, 245–255.
Chandler J.M., Cohen G.M., MacFarlane M. 1998. Different subcellular distribution of caspase-3 and caspase-7 following Fas-induced apoptosis in mouse liver. J. Biol. Chem. 273, 10815–10818.
Li F., Huang Q., Chen J., Peng, Y., Roop D.R., Bedford J.S., Li C.Y. 2010. Apoptotic cells activate the “phoenix rising” pathway to promote wound healing and tissue regeneration. Sci. Signal. 3, ra13.
Kropotov A.V., Chelomin V.P., Slobodskova V.V., Solodova E.E., Mikhailov A.O. 2013. Evaluation of genotoxicity of tetrachloromethane and protective action of silibinin and haurantin on the rat liver using the comet assay. Tikhookean. Med. Zh. 2, 63–66
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated by A. Levina
Abbreviations: CCl4, carbon tetrachloride; СО, carbon monoxide; ATM (Ataxia-Telangiectasia Mutated) and ATR (ATM Related), serine/threonine protein kinases.
Rights and permissions
About this article
Cite this article
Mukhammadiyeva, G.F., Karimov, D.O., Kutlina, T.G. et al. Expression of Cell Cycle, Oxidative Stress, and Apoptosis Related Genes Chek1, Hmox1, Casp7 in Rat Liver Exposed to Carbon Tetrachloride. Mol Biol 53, 64–69 (2019). https://doi.org/10.1134/S0026893319010102
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0026893319010102