Abstract
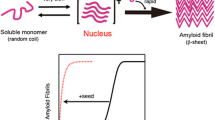
Meisl et al. have recently observed an anomalous dependence of the amyloid formation rate on the protein concentration. A novel mechanism of fibril growth has been proposed by Meisl et al. to explain the abnormality; it consists in the fibril-catalyzed initiation of fibril formation with saturation of catalytic sites at high concentrations of substrates. Our article describes an alternative explanation of the anomalous kinetics, assuming that the formation of metastable oligomers competes with fibril formation by decreasing the concentration of free monomers. Oligomers are indeed observed in the course of amyloid formation, but are usually considered as seeds of amyloid fibrils rather as their competitors. However, the oligomers visually detectable by electron microscopy were shown to be close in size to those that can be derived from the anomalous dependence of the amyloid growth rate on the protein concentration, given that the anomaly results from competition between oligomer formation and amyloidogenesis.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Irvine G.B., El-Agnaf O.M., Shankar G.M., et al. 2008. Protein aggregation in the brain: The molecular basis for Alzheimer′s and Parkinson′s diseases. Mol. Med. 14, 451–464.
Dobson C.M. 2003. Protein folding and misfolding. Nature. 426, 884–890.
Buxbaum J.N., Linke R.P. 2012. A molecular history of the amyloidoses. J. Mol. Biol. 421, 142–159.
Oosawa F., Asakura S., Hotta K., et al. 1959. Transformation of actin as a fibrous condensation. J. Polym. Sci. 37, 323–336.
Ferrone F.A., Hofrichter J., Eaton W.A. 1985. Kinetics of sickle hemoglobin polymerization: 2. A double nucleation mechanism. J. Mol. Biol. 183, 611–631.
Ferrone F. 1999. Analysis of protein aggregation kinetics. Methods Enzymol. 309, 256–274.
Xue W. F., Homans S.W., Radford S.E. 2008. Systematic analysis of nucleation-dependent polymerization reveals new insights into the mechanism of amyloid selfassembly. Proc. Natl. Acad. Sci. U. S. A. 105, 8926–8931.
Andersen C.B., Yagi H., Manno M., et al. 2009. Branching in amyloid fibril growth. Biophys. J. 96, 1529–1536.
Enqvist S., Sletten K., Westermark P. 2009. Fibril protein fragmentation pattern in systemic AL-amyloidosis. J. Pathol. 219, 473–480.
Knowles T.P., Waudby C.A., Devlin G.L., et al. 2009. An analytical solution to the kinetics of breakable filament assembly. Science. 326, 1533–1537.
Cohen S.I.A., Vendruscolo M., Welland M.E. et al. 2011. Nucleated polymerization with secondary pathways. I. Time evolution of the principal moments. J. Chem. Phys. 135, 065105.
Dovidchenko N.V., Finkelstein A.V., Galzitskaya O.V. 2014. How to determine the size of folding nuclei of protofibrils from the concentration dependence of the rate and lag-time of aggregation: 1. Modeling the amyloid protofibril formation. J. Phys. Chem. B. 118, 1189–1197.
Meisl G., Yang X., Hellstrand E., et al. 2014. Differences in nucleation behavior underlie the contrasting aggregation kinetics of the Aβ40 and Aβ42 peptides. Proc. Natl. Acad. Sci. U. S. A. 111, 9384–9389.
Lashuel H.A., Hartley D., Petre B.M., et al. 2002. Neurodegenerative disease: Amyloid pores from pathogenic mutations. Nature. 418, 291–291.
Zhang M., Zhao J., Zheng J. 2014. Molecular understanding of a potential functional link between antimicrobial and amyloid peptides. Soft Matter. 10, 7425–7451.
Suvorina M.Y., Selivanova O.M., Grigorashvili E.I., et al. 2015. Studies of polymorphism of amyloid-β42 peptide from different suppliers. J. Alzheimers Dis. 47, 583–593.
Collins S.R., Douglass A., Vale R.D., Weissman J.S. 2004. Mechanism of prion propagation: Amyloid growth occurs by monomer addition. PLoS Biol. 2 (10), e321.
Finkelstein A.V., Dovidchenko N.V., Selivanova O.M., et al. 2015. Determination of the size of the primary and secondary folding nuclei of protofibrils from the concentration dependence of the rate and the lag-time of their formation. In: Physical Biology of Proteins and Peptides: Theory, Simulation and Experiment. Eds. Olivares-Quiroz L., Guzmán-López O., Jardón-Valadez H.E. New York: Springer, pp. 47–66.
Pauling L. 1970. General Chemistry, New York: Freeman, Chapter 11.
Glenner G.G., Keiser H.R., Bladen H.A., et al. 1968. Amyloid: 6. A comparison of two morphologic components of human amyloid deposits. J. Histochem. Cytochem. 16, 633–644.
Selivanova O.M., Surin A.K., Marchenkov V.V., et al. 2016. The mechanism underlying amyloid polymorphism is opened for Alzheimer′s disease amyloid-β peptide. J. Alzheimer′s Disease. 54, 821–830.
Dovidchenko N.V., Glyakina A.V., Selivanova O.M., et al. 2016. One of the possible mechanisms of amyloid fibrils formation based on the sizes of primary and secondary folding nuclei of Aβ40 and Aβ42. J. Struct. Biol. 194, 404–414.
Grigorashvili E.I., Selivanova O.M., Dovidchenko N.V., et al. 2016. Determination of size of folding nuclei of fibrils formed from recombinant Aβ(1–40) peptide. Biochemistry (Moscow). 81, 538–547
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.V. Finkelstein, N.V. Dovidchenko, O.V. Galzitskaya, 2018, published in Molekulyarnaya Biologiya, 2018, Vol. 52, No. 1, pp. 73–81.
Rights and permissions
Open access. This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Finkelstein, A.V., Dovidchenko, N.V. & Galzitskaya, O.V. Anomalous Kinetics of Amyloidogenesis Suggest a Competition between Oligomers and Fibrils. Mol Biol 52, 62–68 (2018). https://doi.org/10.1134/S002689331801003X
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S002689331801003X